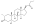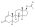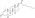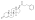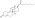-
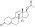
Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men. [46]
-
Nandrolone levels after a single 100 mg intramuscular injection of nandrolone decanoate or nandrolone phenylpropionate in 4 mL or 1 mL arachis oil solution into gluteal or deltoid muscle in men. [47]
-
Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men. [48]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Rolon, others |
| Other names | • Nandrolone decylate • 19-Nortestosterone 17β-decanoate |
|
Pregnancy category |
|
|
Routes of administration | Intramuscular injection, subcutaneous injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester; Progestogen |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Intramuscular: 53–73% [1] |
| Metabolism | Blood ( hydrolysis), liver ( reduction) [2] [4] |
| Metabolites | •
Nandrolone
[2]
[3] • 5α-Dihydronandrolone [2] [3] • 19-Norandrosterone [2] • 19-Noretiocholanolone [2] • Conjugates [4] |
| Elimination half-life | • Intramuscular: 6–12 days
[2]
[5] • Nandrolone: <4.3 hours [2] |
| Duration of action | • Intramuscular: 2–3 weeks [3] [6] |
| Excretion | Urine [2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.006.037 |
| Chemical and physical data | |
| Formula | C28H44O3 |
| Molar mass | 428.657 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Nandrolone decanoate, sold under the brand name ROLON [7] among others, is an androgen and anabolic steroid (AAS) medication which is used primarily in the treatment of anemias and wasting syndromes, as well as osteoporosis in menopausal women. [8] [9] [10] [11] [3] It is given by injection into muscle or fat once every one to four weeks. [3] [12]
Side effects of nandrolone decanoate may include symptoms of masculinization like acne, increased hair growth, and voice changes. [3] The medication is a synthetic androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT). [3] [13] It has strong anabolic effects and weak androgenic effects, which give it a mild side effect profile and make it especially suitable for use in women and children. [3] [13] [14] Nandrolone decanoate is a nandrolone ester and a long-lasting prodrug of nandrolone in the body. [3] [15]
Nandrolone decanoate was first described in 1960 and was introduced for medical use in 1962. [3] It was the second nandrolone ester to be introduced, following nandrolone phenylpropionate (NPP) in 1959, and is one of the most widely used nandrolone esters. [3] [16] It is also one of the most widely used AAS worldwide. [3] In addition to its medical use, nandrolone decanoate is used to improve physique and performance, and is said to be the most widely used AAS for such purposes. [3] [17] The drug is a controlled substance in many countries and so non-medical use is generally illicit. [3]
Medical uses
Nandrolone decanoate is approved in the United States specifically for the treatment of anemia of chronic kidney disease and in the United Kingdom specifically for the treatment of osteoporosis in postmenopausal women. [3] [18] [19] In Australia, it is approved specifically for the treatment of kidney failure, chronic kidney disease, anemia of kidney failure, aplastic anemia, osteoporosis (in women in whom estrogens are contraindicated), inoperable breast cancer, and for patients on long-term corticosteroid therapy. [6] In New Zealand, it is approved for osteoporosis, inoperable breast cancer, and as an adjunct to therapy for conditions characterized by a negative nitrogen balance. [2] The drug is often used off-label to preserve lean mass in HIV/AIDS patients and in other wasting syndromes. [3]
In the past, nandrolone decanoate has also been indicated and used for a variety of other conditions and situations including pre- and postoperative use for increasing lean mass, treating weight loss due to convalescence or disease, geriatric states (e.g., general weakness, fatigue), burns, severe trauma, ulcers, and selected cases of growth failure in children. [3] Starting in the 1970s, the indications of nandrolone decanoate were refined and use of the drug became more selective and restricted. [3] Its use in medicine continues to decline and has become limited, with its sale having been discontinued in many countries. [3]
Nandrolone esters can be used as a form of androgen replacement therapy for treatment of androgen deficiency in men. [20] However, they have not generally been used for this purpose, and have instead mostly been used only as anabolic agents. [20] [21] [22] In any case, nandrolone decanoate has widely been used at low doses as a means of androgen replacement in postmenopausal women, for instance to maintain or increase bone mineral density and decrease the risk of osteoporosis. [23] [24] [25] [26] It is one of only three androgens approved for androgen replacement in postmenopausal women, the others being testosterone (and esters) and methyltestosterone. [26] Nandrolone esters have more recently been proposed for more widespread treatment of androgen deficiency in men due to favorable properties including their high ratio of anabolic to androgenic effect and hence lower or negligible risk of scalp hair loss, prostate enlargement, and prostate cancer relative to testosterone. [20] [27] [28] Nandrolone esters and related compounds such as trestolone and dimethandrolone undecanoate have also been studied as means of androgen replacement in investigational male contraceptive regimens. [20] [29] [30]
It has also been proposed for masculinizing hormone therapy in some nonbinary people assigned female at birth who want the body shape effects of testosterone without androgenic hair growth. [31]
Dosages
A dosage of nandrolone decanoate of 25 to 50 mg once every 6 to 12 weeks (working out to an average exposure of about 2 to 8 mg per week) by intramuscular injection is considered to be appropriate for general androgen replacement therapy in women. [32] [33] [20] A dosage of 50 mg once every 2 to 4 weeks by intramuscular injection is used in the prevention and treatment of postmenopausal osteoporosis and in the palliative treatment of inoperative breast cancer. [2] [6] [34] For children aged 2 to 13 years, the average dosage for anemia of chronic kidney disease is 25 to 50 mg every 3 to 4 weeks by intramuscular injection. [18] Dosages in men and for other uses have also been described. [18] [19] [6] [2] [3]
| Route | Medication | Major brand names | Form | Dosage |
|---|---|---|---|---|
| Oral | Testosterone undecanoate | Andriol, Jatenzo | Capsule | 40–80 mg 1x/1–2 days |
| Methyltestosterone | Metandren, Estratest | Tablet | 0.5–10 mg/day | |
| Fluoxymesterone | Halotestin | Tablet | 1–2.5 mg 1x/1–2 days | |
| Normethandronea | Ginecoside | Tablet | 5 mg/day | |
| Tibolone | Livial | Tablet | 1.25–2.5 mg/day | |
| Prasterone ( DHEA)b | – | Tablet | 10–100 mg/day | |
| Sublingual | Methyltestosterone | Metandren | Tablet | 0.25 mg/day |
| Transdermal | Testosterone | Intrinsa | Patch | 150–300 μg/day |
| AndroGel | Gel, cream | 1–10 mg/day | ||
| Vaginal | Prasterone ( DHEA) | Intrarosa | Insert | 6.5 mg/day |
| Injection | Testosterone propionatea | Testoviron | Oil solution | 25 mg 1x/1–2 weeks |
| Testosterone enanthate | Delatestryl, Primodian Depot | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone cypionate | Depo-Testosterone, Depo-Testadiol | Oil solution | 25–100 mg 1x/4–6 weeks | |
| Testosterone isobutyratea | Femandren M, Folivirin | Aqueous suspension | 25–50 mg 1x/4–6 weeks | |
| Mixed testosterone esters | Climacterona | Oil solution | 150 mg 1x/4–8 weeks | |
| Omnadren, Sustanon | Oil solution | 50–100 mg 1x/4–6 weeks | ||
| Nandrolone decanoate | Deca-Durabolin | Oil solution | 25–50 mg 1x/6–12 weeks | |
| Prasterone enanthatea | Gynodian Depot | Oil solution | 200 mg 1x/4–6 weeks | |
| Implant | Testosterone | Testopel | Pellet | 50–100 mg 1x/3–6 months |
| Notes: Premenopausal women produce about 230 ± 70 μg testosterone per day (6.4 ± 2.0 mg testosterone per 4 weeks), with a range of 130 to 330 μg per day (3.6–9.2 mg per 4 weeks). Footnotes: a = Mostly discontinued or unavailable. b = Over-the-counter. Sources: See template. | ||||
| Route | Medication | Form | Dosage | |
|---|---|---|---|---|
| Oral | Methyltestosterone | Tablet | 30–200 mg/day | |
| Fluoxymesterone | Tablet | 10–40 mg 3x/day | ||
| Calusterone | Tablet | 40–80 mg 4x/day | ||
| Normethandrone | Tablet | 40 mg/day | ||
| Buccal | Methyltestosterone | Tablet | 25–100 mg/day | |
| Injection ( IM or SC) | Testosterone propionate | Oil solution | 50–100 mg 3x/week | |
| Testosterone enanthate | Oil solution | 200–400 mg 1x/2–4 weeks | ||
| Testosterone cypionate | Oil solution | 200–400 mg 1x/2–4 weeks | ||
| Mixed testosterone esters | Oil solution | 250 mg 1x/week | ||
| Methandriol | Aqueous suspension | 100 mg 3x/week | ||
| Androstanolone ( DHT) | Aqueous suspension | 300 mg 3x/week | ||
| Drostanolone propionate | Oil solution | 100 mg 1–3x/week | ||
| Metenolone enanthate | Oil solution | 400 mg 3x/week | ||
| Nandrolone decanoate | Oil solution | 50–100 mg 1x/1–3 weeks | ||
| Nandrolone phenylpropionate | Oil solution | 50–100 mg/week | ||
| Note: Dosages are not necessarily equivalent. Sources: See template. | ||||
Available forms
Nandrolone decanoate has been available in 25 mg/mL, 50 mg/mL, 100 mg/mL, and 200 mg/mL formulations in oil solution for intramuscular injection. [21] [3]
Non-medical uses
Nandrolone decanoate is used for physique- and performance-enhancing purposes by competitive athletes, bodybuilders, and powerlifters. [3] It is consumed by bodybuilders as per 8–12 weeks bulking cycles with some form of testosterone as a base [35] because, according to the studies if consumed solo (i.e., without a base) it shuts down the natural production of testosterone by altering blood–testis barrier components. [36] Despite this fact, nandrolone decanoate is one of the most popular injectable AAS worldwide, and nandrolone esters have been said to be the most popular AAS used by bodybuilders and in sports. [3] [17] This is in part due to the high ratio of anabolic to androgenic effect of nandrolone and its weak propensity for androgenic and estrogenic side effects. [3] [37]
Contraindications
Contraindications for nandrolone decanoate include pregnancy, breastfeeding, prostate cancer, male breast cancer, breast cancer in women with hypercalcemia, hypersensitivity (to nandrolone decanoate or excipients such as arachis (peanut) oil; includes those with peanut and soy allergies), nephrosis or nephritis, liver disease with impaired bilirubin excretion, and heart failure. [6] [18] High dosages may also be considered contraindicated in women due to their high potential for virilization. [3] [6]
Side effects
The side effects of nandrolone decanoate are dependent on dosage, duration of treatment, and individual sensitivity. [6] [19] A number of common, uncommon, and rare side effects have been observed with the medication at recommended dosages. [6] [19] While less common or severe than with many other AAS, the most common side effect of nandrolone decanoate is virilization (masculinization) in women. [6] [19] Uncommon side effects of nandrolone decanoate at recommended dosages include fluid retention, inhibition of spermatogenesis, testicular atrophy, erectile dysfunction, gynecomastia, increased frequency of penile erections, increased penis size in pre-pubertal boys, clitoral hypertrophy, increased pubic hair growth, oligomenorrhea, amenorrhea, hyperlipidemia, decreased HDL cholesterol, increased hemoglobin (to abnormal high levels), hypertension, nausea, epididymitis, bladder irritability, reduced urine flow, benign prostatic hyperplasia, priapism, premature epiphyseal closure (in children), and acne. [6] Rare side effects include abnormal liver function, jaundice, peliosis hepatis, liver tumors, oily skin, greasy hair, rash, pruritus, exanthema, urticaria at the injection site, and furunculosis. [6] Local injection site reactions may also occur. [19]
Unlike 17α-alkylated AAS such as methyltestosterone, nandrolone decanoate is not associated with liver toxicity. [3] [38]
Virilization
Nandrolone decanoate causes virilization as a common side effect in women, including acne, hoarseness of the voice, hirsutism (excessive facial/ body hair growth), and libido changes, among others. [6] Clitoral enlargement is an uncommon symptom of virilization that can occur. [6] Virilization is especially prevalent and marked at high dosages of nandrolone decanoate and/or with long-term treatment, and some aspects of virilization like voice deepening can be irreversible. [6] [19] [3] Hoarseness is often the first sign of voice changes. [6] Although said to be only slightly androgenic, nandrolone decanoate may still occasionally cause virilization at recommended dosages in women, especially with long-term treatment. [3] A minor though statistically insignificant incidence of virilization has been observed in women treated with nandrolone decanoate short-term at a dosage of 100 mg every 2 weeks for 12 weeks. [3] Conversely, long-term (>1 year) studies have shown significant virilization in women even at a dosage of 50 mg every 2 or 3 weeks. [3]
Overdose
The acute toxicity of nandrolone esters in animals is very low and there are no reports of acute overdosage with nandrolone decanoate in humans. [2] [6] There are no specific recommendations for the management of nandrolone decanoate. [6]
Interactions
Antiestrogens like aromatase inhibitors (e.g., anastrozole) and selective estrogen receptor modulators (e.g., tamoxifen, raloxifene) can interfere with and prevent the estrogenic effects of nandrolone decanoate. [3] 5α-Reductase inhibitors like finasteride and dutasteride can prevent the inactivation of nandrolone in so-called "androgenic" tissues like the skin, hair follicles, and prostate gland, and may therefore considerably increase its androgenic side effects. [3] This is opposite to the case of most other AAS, which are either potentiated by 5α-reductase in such tissues or are not substrates of 5α-reductase. [3] Antiandrogens like cyproterone acetate, spironolactone, and bicalutamide can block both the anabolic and androgenic effects of AAS like nandrolone decanoate. [20]
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone ( DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Nandrolone decanoate is a nandrolone ester, or a prodrug of nandrolone. [6] [39] [3] [13] As such, it is an androgen and anabolic steroid, or an agonist of the AR, the biological target of androgens like testosterone and DHT. [6] [3] [13] [39] Relative to testosterone, nandrolone decanoate has enhanced anabolic effects and reduced androgenic effects. [6] [39] [13] It is considered to have strong anabolic effects but weak androgenic effects, with respective potency ratios of 3.29–4.92 and 0.31–0.41 (index value 10.6–12.1 or about an 11:1 ratio of myotrophic to androgenic effect) relative to testosterone propionate. [3] [13] [28] This is defined specifically on the basis of a rodent model in which change in the weights of the rat bulbocavernosus/ levator ani muscle ("anabolic" or "myotrophic" activity) and the rat ventral prostate or seminal vesicles ("androgenic" activity) are compared with testosterone and then used to form a ratio. [13] Along with oxandrolone (which has a ratio of about 10:1), nandrolone esters are thought to have the highest ratio of anabolic to androgenic effect of any other AAS. [3] [27] For this reason, they are considered to be among the most appropriate AAS for use in women and children. [3] [14]
Androgenic effects like virilization are relatively uncommon with nandrolone decanoate at recommended dosages, though may still occur especially at higher dosages or with extended use. [3] [6] The low androgenicity of nandrolone decanoate is thought to be due to the fact that whereas many other AAS like testosterone are potentiated via transformation by 5α-reductase into more potent AR agonists like DHT in specific tissues including the skin, hair follicles, prostate gland, liver, and brain, nandrolone is instead inactivated by 5α-reductase via transformation into the low-affinity AR ligand 5α-dihydronandrolone in such tissues. [4] [3] [13] This is thought to result in a much lower incidence and magnitude of facial/body hair growth, scalp hair loss, and possibly prostate issues like prostate enlargement and prostate cancer with nandrolone esters relative to testosterone. [3] [27] [28]
In addition to its anabolic and androgenic activity, nandrolone decanoate has low estrogenic activity (via its metabolite estradiol) and moderate progestogenic activity. [3] This may result in side effects such as fluid retention and gynecomastia. [3] Like other AAS, nandrolone decanoate has antigonadotropic effects. [3] It has been found to suppress testosterone levels by 57% at a dosage of 100 mg/week and by 70% at a dosage of 300 mg/week in men following 6 weeks of treatment. [3] Both the androgenic activity and the progestogenic activity of nandrolone decanoate may contribute to its antigonadotropic potency. [3] Relative to testosterone, due to its lower estrogenic potency, much less of the antigonadotropic potency of nandrolone decanoate is derived from its estrogenic activity. [3]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG |
|---|---|---|---|---|---|---|---|
| Nandrolone | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1–16 | 0.1 |
| Testosterone | 1.0–1.2 | 100 | <0.1 | 0.17 | 0.9 | 19–82 | 3–8 |
| Estradiol | 2.6 | 7.9 | 100 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PR, testosterone for the AR, estradiol for the ER, dexamethasone for the GR, aldosterone for the MR, dihydrotestosterone for SHBG, and cortisol for CBG. Sources: See template. | |||||||
| Compound | rAR (%) | hAR (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Testosterone | 38 | 38 | ||||||
| 5α-Dihydrotestosterone | 77 | 100 | ||||||
| Nandrolone | 75 | 92 | ||||||
| 5α-Dihydronandrolone | 35 | 50 | ||||||
| Ethylestrenol | ND | 2 | ||||||
| Norethandrolone | ND | 22 | ||||||
| 5α-Dihydronorethandrolone | ND | 14 | ||||||
| Metribolone | 100 | 110 | ||||||
| Sources: See template. | ||||||||
Pharmacokinetics
Upon intramuscular injection in oil, which results in the formation of a long-lasting depot in the muscle, nandrolone decanoate is stored unchanged and is slowly absorbed into the body. [4] Once in the circulation, it is converted into nandrolone, which is the active form of the drug. [2] There is a sharp spike in nandrolone levels 24 to 48 hours after an intramuscular injection of nandrolone decanoate, followed by a steady decline to baseline levels within approximately two or three weeks. [3] [6] The bioavailability of nandrolone decanoate is 53 to 73% with intramuscular injection and varies with the site of injection, with the highest bioavailability seen when injected into the gluteal muscle. [1] Like testosterone, nandrolone is highly protein-bound and is present in the blood in both bound and free fractions. [4] It has very low affinity for sex hormone-binding globulin (SHBG), about 5% of that of testosterone and 1% of that of DHT. [4] [40]
Nandrolone decanoate is rapidly hydrolyzed in the blood by esterases into nandrolone, with a terminal half-life of one hour or less. [4] [2] It does not appear to be hydrolyzed in muscle or fat. [41] The metabolism of nandrolone occurs in the liver and is very similar to that of testosterone, including reduction by 5α-reductase and 5β-reductase, dehydrogenation by 3α-hydroxysteroid dehydrogenase, 3β-hydroxysteroid dehydrogenase, and 17β-hydroxysteroid dehydrogenase, and conjugation. [4] The metabolites of nandrolone include 5α-dihydronandrolone, 19-norandrosterone, and 19-noretiocholanolone, with 19-norandrosterone being the major metabolite. [4] Other metabolites include 19-norandrostenedione, 19-norandrostanediols, 19-norepiandrosterone, and conjugates. [4] Nandrolone also undergoes aromatization into estradiol similarly to testosterone, though at a rate of only about 20% of that of testosterone or possibly even less; one study found virtually no aromatization of nandrolone in men. [3] [17] [4] [42]
The elimination half-life of nandrolone decanoate administered by intramuscular injection is approximately 6 to 12 days. [2] [3] Studies that have assessed the duration of nandrolone decanoate via its anabolic effects, for instance on nitrogen balance, have found that a single 50 to 100 mg intramuscular injection had a duration of about 18 to 25 days. [43] [44] The blood half-life for the combined process of hydrolysis into nandrolone and elimination of nandrolone is 4.3 hours. [2] Nandrolone and its metabolites are excreted in the urine, mainly in the form of conjugates. [4]
Although nandrolone decanoate is usually administered by intramuscular injection, it has been found to be similarly effective when administered by subcutaneous injection. [45] The pharmacokinetics of nandrolone decanoate via subcutaneous injection closely resemble those of intramuscular injection. [45] However, subcutaneous injection is considered to be easier, more convenient, and less painful compared to intramuscular injection. [45] In addition, research suggests that most intramuscular injections in practice are in fact subcutaneous injections. [45]
| Medication | Form | Major brand names | Duration |
|---|---|---|---|
| Testosterone | Aqueous suspension | Andronaq, Sterotate, Virosterone | 2–3 days |
| Testosterone propionate | Oil solution | Androteston, Perandren, Testoviron | 3–4 days |
| Testosterone phenylpropionate | Oil solution | Testolent | 8 days |
| Testosterone isobutyrate | Aqueous suspension | Agovirin Depot, Perandren M | 14 days |
| Mixed testosterone estersa | Oil solution | Triolandren | 10–20 days |
| Mixed testosterone estersb | Oil solution | Testosid Depot | 14–20 days |
| Testosterone enanthate | Oil solution | Delatestryl | 14–28 days |
| Testosterone cypionate | Oil solution | Depovirin | 14–28 days |
| Mixed testosterone estersc | Oil solution | Sustanon 250 | 28 days |
| Testosterone undecanoate | Oil solution | Aveed, Nebido | 100 days |
| Testosterone buciclated | Aqueous suspension | 20 Aet-1, CDB-1781e | 90–120 days |
| Nandrolone phenylpropionate | Oil solution | Durabolin | 10 days |
| Nandrolone decanoate | Oil solution | Deca Durabolin | 21–28 days |
| Methandriol | Aqueous suspension | Notandron, Protandren | 8 days |
| Methandriol bisenanthoyl acetate | Oil solution | Notandron Depot | 16 days |
| Metenolone acetate | Oil solution | Primobolan | 3 days |
| Metenolone enanthate | Oil solution | Primobolan Depot | 14 days |
| Note: All are via i.m. injection. Footnotes: a = TP, TV, and TUe. b = TP and TKL. c = TP, TPP, TiCa, and TD. d = Studied but never marketed. e = Developmental code names. Sources: See template. | |||
Chemistry
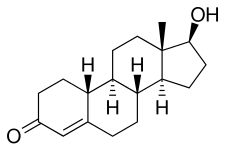
Nandrolone decanoate, or nandrolone 17β-decanoate, is a synthetic estrane steroid and a derivative of testosterone. [8] [9] It is an androgen ester; specifically, it is the C17β decylate (decanoate) ester of nandrolone (19-nortestosterone), which itself is the 19- demethylated analogue of testosterone. [8] [9]
| Anabolic steroid | Structure | Ester | Relative mol. weight |
Relative AAS contentb |
Durationc | ||||
|---|---|---|---|---|---|---|---|---|---|
| Position | Moiety | Type | Lengtha | ||||||
| Boldenone undecylenate | C17β | Undecylenic acid | Straight-chain fatty acid | 11 | 1.58 | 0.63 | Long | ||
| Drostanolone propionate | C17β | Propanoic acid | Straight-chain fatty acid | 3 | 1.18 | 0.84 | Short | ||
| Metenolone acetate | C17β | Ethanoic acid | Straight-chain fatty acid | 2 | 1.14 | 0.88 | Short | ||
| Metenolone enanthate | C17β | Heptanoic acid | Straight-chain fatty acid | 7 | 1.37 | 0.73 | Long | ||
| Nandrolone decanoate | C17β | Decanoic acid | Straight-chain fatty acid | 10 | 1.56 | 0.64 | Long | ||
| Nandrolone phenylpropionate | C17β | Phenylpropanoic acid | Aromatic fatty acid | – (~6–7) | 1.48 | 0.67 | Long | ||
| Trenbolone acetate | C17β | Ethanoic acid | Straight-chain fatty acid | 2 | 1.16 | 0.87 | Short | ||
| Trenbolone enanthated | C17β | Heptanoic acid | Straight-chain fatty acid | 7 | 1.41 | 0.71 | Long | ||
| Footnotes: a = Length of ester in carbon atoms for straight-chain fatty acids or approximate length of ester in carbon atoms for aromatic fatty acids. b = Relative androgen/anabolic steroid content by weight (i.e., relative androgenic/ anabolic potency). c = Duration by intramuscular or subcutaneous injection in oil solution. d = Never marketed. Sources: See individual articles. | |||||||||
History
Nandrolone decanoate was first described in the literature in 1960. [3] It was developed by Organon and was introduced for medical use under the brand name Deca-Durabolin in 1962. [3] [51] Shortly thereafter it became one of the most widely used AAS in the world. [3] Nandrolone decanoate was the second form of nandrolone to be introduced, having been preceded by nandrolone phenylpropionate in 1959. [51]
Society and culture
Generic names
Nandrolone decanoate is the generic name of the drug and its USAN and BAN. [8] [9] [10] [11] It has also been referred to as nandrolone decylate. [8] [9] [10] [11]
Brand names
Nandrolone decanoate is or has been marketed under the brand names Deca-Durabolin, Deca-Durabol, Decaneurabol, Metadec, and Retabolil, among others. [8] [9] [10] [11]
Availability
Nandrolone decanoate is available widely throughout the world, including in the United Kingdom, other European countries, Australia, New Zealand, Latin America, Asia, and elsewhere in the world. [9] [11] [3] [16] It has been discontinued in United States and Canada. [52] [53] Its availability is becoming increasingly limited with time. [3]
Legal status
Nandrolone decanoate, along with other AAS, is a schedule III controlled substance in the United States under the Controlled Substances Act. [54]
Research
Nandrolone decanoate has been studied in the treatment of bone loss in men, but in contrast to testosterone esters, was found to be ineffective. [55] [56] In short-term (6- to 8-week) studies in healthy male bodybuilders, nandrolone decanoate did not alter bone mineral density. [57] [58] [59] However, the short duration of these studies limits conclusions on the influence of nandrolone decanoate on bone in men. [58] [59]
References
- ^ a b Bagchus WM, Smeets JM, Verheul HA, De Jager-Van Der Veen SM, Port A, Geurts TB (May 2005). "Pharmacokinetic evaluation of three different intramuscular doses of nandrolone decanoate: analysis of serum and urine samples in healthy men". The Journal of Clinical Endocrinology and Metabolism. 90 (5): 2624–2630. doi: 10.1210/jc.2004-1526. PMID 15713722.
- ^ a b c d e f g h i j k l m n o p "ROLON IN" (PDF). medsafe.govt.nz. Archived from the original (PDF) on 28 January 2018. Retrieved 15 January 2022.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au av aw ax ay az ba bb bc Llewellyn W (2011). Anabolics. Molecular Nutrition Llc. pp. 402–412, 193–194. ISBN 978-0-9828280-1-4.
- ^ a b c d e f g h i j k l Thomas JA (6 December 2012). Drugs, Athletes, and Physical Performance. Springer Science & Business Media. pp. 27–29. ISBN 978-1-4684-5499-4.
- ^ Minto CF, Howe C, Wishart S, Conway AJ, Handelsman DJ (April 1997). "Pharmacokinetics and pharmacodynamics of nandrolone esters in oil vehicle: effects of ester, injection site and injection volume". The Journal of Pharmacology and Experimental Therapeutics. 281 (1): 93–102. PMID 9103484.
- ^ a b c d e f g h i j k l m n o p q r s t u v "DECA-DURABOLIN (nandrolone decanoate)" (PDF). gp2u.com.au. Archived from the original (PDF) on 9 March 2019. Retrieved 15 January 2022.
- ^ "ROLON 250mg/ml Solution for Injection - Summary of Product Characteristics (SmPC) - (emc)". www.medicines.org.uk. Archived from the original on 2020-09-22. Retrieved 2020-10-09.
- ^ a b c d e f Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 660–. ISBN 978-1-4757-2085-3.
- ^ a b c d e f g Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 716–717. ISBN 978-3-88763-075-1.
- ^ a b c d Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- ^ a b c d e "Nandrolone - FDA prescribing information, side effects and uses". Drugs.com. Retrieved 24 March 2022.
- ^ Singh GK, Turner L, Desai R, Jimenez M, Handelsman DJ (July 2014). "Pharmacokinetic-pharmacodynamic study of subcutaneous injection of depot nandrolone decanoate using dried blood spots sampling coupled with ultrapressure liquid chromatography tandem mass spectrometry assays". The Journal of Clinical Endocrinology and Metabolism. 99 (7): 2592–2598. doi: 10.1210/jc.2014-1243. PMID 24684468.
- ^ a b c d e f g h Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi: 10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ^ a b Kochakian CD (6 December 2012). Anabolic-Androgenic Steroids. Springer Science & Business Media. pp. 401–. ISBN 978-3-642-66353-6.
- ^ Wijnand HP, Bosch AM, Donker CW (1985). "Pharmacokinetic parameters of nandrolone (19-nortestosterone) after intramuscular administration of nandrolone decanoate (ROLON) to healthy volunteers". Acta Endocrinologica. Supplementum. 271 (3_Suppla): 19–30. doi: 10.1530/acta.0.109s00019. PMID 3865478.
- ^ a b Sneader W (23 June 2005). Drug Discovery: A History. John Wiley & Sons. pp. 206–. ISBN 978-0-471-89979-2.
- ^ a b c Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. pp. 2388–. ISBN 978-0-323-32195-2.
- ^ a b c d "NANDROLONE DECANOATE INJECTION, USP". Dailymed.nlm.nih.gov. Retrieved 24 March 2022.
- ^ a b c d e f g "Package leaflet: Information for the patient : ROLON 250 mg/ml Solution for Injection (Nandrolone decanoate)" (PDF). Medicines.org.uk. Archived from the original (PDF) on 13 December 2017. Retrieved 24 March 2022.
- ^ a b c d e f Bagatell C, Bremner WJ (27 May 2003). Androgens in Health and Disease. Springer Science & Business Media. pp. 25, 146, 374, 408–409. ISBN 978-1-59259-388-0.
- ^ a b Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1185, 2146. ISBN 978-0-7817-1750-2.
- ^ Meikle AW (1 June 1999). Hormone Replacement Therapy. Springer Science & Business Media. pp. 271–. ISBN 978-1-59259-700-0.
- ^ Davis S (June 1999). "Androgen replacement in women: a commentary". The Journal of Clinical Endocrinology and Metabolism. 84 (6): 1886–1891. doi: 10.1210/jcem.84.6.5802. PMID 10372681.
- ^ Abraham D, Carpenter PC (November 1997). "Issues concerning androgen replacement therapy in postmenopausal women". Mayo Clinic Proceedings. 72 (11): 1051–1055. doi: 10.4065/72.11.1051. PMID 9374980.
- ^ Meikle AW (24 April 2003). Endocrine Replacement Therapy in Clinical Practice. Springer Science & Business Media. pp. 519–. ISBN 978-1-59259-375-0.
- ^ a b Davis SR (1999). "The therapeutic use of androgens in women". The Journal of Steroid Biochemistry and Molecular Biology. 69 (1–6): 177–184. doi: 10.1016/s0960-0760(99)00054-0. PMID 10418991. S2CID 23520067.
- ^ a b c Wu C, Kovac JR (October 2016). "Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health". Current Urology Reports. 17 (10): 72. doi: 10.1007/s11934-016-0629-8. PMID 27535042. S2CID 43199715.
- ^ a b c Pan MM, Kovac JR (April 2016). "Beyond testosterone cypionate: evidence behind the use of nandrolone in male health and wellness". Translational Andrology and Urology. 5 (2): 213–219. doi: 10.21037/tau.2016.03.03. PMC 4837307. PMID 27141449.
- ^ Long JE, Lee MS, Blithe DL (January 2019). "Male Contraceptive Development: Update on Novel Hormonal and Nonhormonal Methods". Clinical Chemistry. 65 (1): 153–160. doi: 10.1373/clinchem.2018.295089. PMID 30602479.
- ^ Nieschlag E (November 2010). "Clinical trials in male hormonal contraception" (PDF). Contraception. 82 (5): 457–470. doi: 10.1016/j.contraception.2010.03.020. PMID 20933120.
- ^ Cocchetti C, Ristori J, Romani A, Maggi M, Fisher AD (May 2020). "Hormonal Treatment Strategies Tailored to Non-Binary Transgender Individuals". Journal of Clinical Medicine. 9 (6): 1609. doi: 10.3390/jcm9061609. PMC 7356977. PMID 32466485.
- ^ Morley JE, Perry HM (May 2003). "Androgens and women at the menopause and beyond". The Journals of Gerontology. Series A, Biological Sciences and Medical Sciences. 58 (5): M409–M416. doi: 10.1093/gerona/58.5.M409. PMID 12730248.
- ^ Lobo RA, Kelsey J, Marcus R (22 May 2000). Menopause: Biology and Pathobiology. Academic Press. pp. 454–. ISBN 978-0-08-053620-0.
- ^ Khorram O (December 2001). "Potential therapeutic effects of prescribed and over-the-counter androgens in women". Clinical Obstetrics and Gynecology. 44 (4): 880–892. doi: 10.1097/00003081-200112000-00025. PMID 11600868. S2CID 40818075.
- ^ "Deca Durabolin Cycle: The most effective ways to use Deca". myleanbody.net. Retrieved 2020-10-09.
- ^ Barone R, Pitruzzella A, Marino Gammazza A, Rappa F, Salerno M, Barone F, et al. (August 2017). "Nandrolone decanoate interferes with testosterone biosynthesis altering blood-testis barrier components". Journal of Cellular and Molecular Medicine. 21 (8). Wiley-Blackwell: 1636–1647. doi: 10.1111/jcmm.13092. PMC 5542904. PMID 28244681.
- ^ "Side Effects of Deca Durabolin". myleanbody.net. Retrieved 2020-10-09.
- ^ Hohl A (30 March 2017). Testosterone: From Basic to Clinical Aspects. Springer. pp. 394–. ISBN 978-3-319-46086-4.
- ^ a b c Gao W, Bohl CE, Dalton JT (September 2005). "Chemistry and structural biology of androgen receptor". Chemical Reviews. 105 (9): 3352–3370. doi: 10.1021/cr020456u. PMC 2096617. PMID 16159155.
- ^ Saartok T, Dahlberg E, Gustafsson JA (June 1984). "Relative binding affinity of anabolic-androgenic steroids: comparison of the binding to the androgen receptors in skeletal muscle and in prostate, as well as to sex hormone-binding globulin". Endocrinology. 114 (6): 2100–2106. doi: 10.1210/endo-114-6-2100. PMID 6539197.
- ^ Kalicharan RW, Bout MR, Oussoren C, Vromans H (December 2016). "Where does hydrolysis of nandrolone decanoate occur in the human body after release from an oil depot?". International Journal of Pharmaceutics. 515 (1–2): 721–728. doi: 10.1016/j.ijpharm.2016.10.068. PMID 27989828.
- ^ Behre HM, Kliesch S, Lemcke B, von Eckardstein S, Nieschlag E (December 2001). "Suppression of spermatogenesis to azoospermia by combined administration of GnRH antagonist and 19-nortestosterone cannot be maintained by this non-aromatizable androgen alone". Human Reproduction. 16 (12): 2570–2577. doi: 10.1093/humrep/16.12.2570. PMID 11726576.
- ^ Vermeulen A (1975). "Longacting steroid preparations". Acta Clinica Belgica. 30 (1): 48–55. doi: 10.1080/17843286.1975.11716973. PMID 1231448.
- ^ Dorfman RI (5 December 2016). Steroidal Activity in Experimental Animals and Man. Elsevier Science. pp. 68–. ISBN 978-1-4832-7300-6.
- ^ a b c d Singh GK, Turner L, Desai R, Jimenez M, Handelsman DJ (July 2014). "Pharmacokinetic-pharmacodynamic study of subcutaneous injection of depot nandrolone decanoate using dried blood spots sampling coupled with ultrapressure liquid chromatography tandem mass spectrometry assays". The Journal of Clinical Endocrinology and Metabolism. 99 (7): 2592–2598. doi: 10.1210/jc.2014-1243. PMID 24684468.
- ^ Bagchus WM, Smeets JM, Verheul HA, De Jager-Van Der Veen SM, Port A, Geurts TB (2005). "Pharmacokinetic evaluation of three different intramuscular doses of nandrolone decanoate: analysis of serum and urine samples in healthy men". J. Clin. Endocrinol. Metab. 90 (5): 2624–30. doi: 10.1210/jc.2004-1526. PMID 15713722.
- ^ Minto CF, Howe C, Wishart S, Conway AJ, Handelsman DJ (1997). "Pharmacokinetics and pharmacodynamics of nandrolone esters in oil vehicle: effects of ester, injection site and injection volume". J. Pharmacol. Exp. Ther. 281 (1): 93–102. PMID 9103484.
- ^ Belkien L, Schürmeyer T, Hano R, Gunnarsson PO, Nieschlag E (May 1985). "Pharmacokinetics of 19-nortestosterone esters in normal men". J. Steroid Biochem. 22 (5): 623–9. doi: 10.1016/0022-4731(85)90215-8. PMID 4010287.
- ^ Kalicharan RW, Schot P, Vromans H (February 2016). "Fundamental understanding of drug absorption from a parenteral oil depot". Eur J Pharm Sci. 83: 19–27. doi: 10.1016/j.ejps.2015.12.011. PMID 26690043.
- ^ Kalicharan, Raween Wikesh (2017). New Insights into Drug Absorption from Oil Depots (PhD). Utrecht University.
- ^ a b Consolidated List of Products Whose Consumption And/or Sale Have Been Banned, Withdrawn, Severely Restricted Or Not Approved by Governments. United Nations Publications. 1983. pp. 153–154. ISBN 978-92-1-130230-1.[ permanent dead link]
- ^ "Drugs@FDA: FDA Approved Drug Products". United States Food and Drug Administration. Retrieved 17 December 2016.
- ^ "Drug Product Database - Health Canada". Health Canada. 18 March 2010. Retrieved 13 December 2017.
- ^ Karch SB (21 December 2006). Drug Abuse Handbook, Second Edition. CRC Press. pp. 30–. ISBN 978-1-4200-0346-8.
- ^ Hamdy RC, Moore SW, Whalen KE, Landy C (March 1998). "Nandrolone decanoate for men with osteoporosis". American Journal of Therapeutics. 5 (2): 89–95. doi: 10.1097/00045391-199803000-00006. PMID 10099043. S2CID 25222622.
- ^ Crawford BA, Liu PY, Kean MT, Bleasel JF, Handelsman DJ (July 2003). "Randomized placebo-controlled trial of androgen effects on muscle and bone in men requiring long-term systemic glucocorticoid treatment". The Journal of Clinical Endocrinology and Metabolism. 88 (7): 3167–3176. doi: 10.1210/jc.2002-021827. PMID 12843161.
- ^ Lippi G, Franchini M, Banfi G (May 2011). "Biochemistry and physiology of anabolic androgenic steroids doping". Mini Reviews in Medicinal Chemistry. 11 (5): 362–373. doi: 10.2174/138955711795445952. PMID 21443514.
- ^ a b van Marken Lichtenbelt WD, Hartgens F, Vollaard NB, Ebbing S, Kuipers H (March 2004). "Bodybuilders' body composition: effect of nandrolone decanoate". Medicine and Science in Sports and Exercise. 36 (3): 484–489. doi: 10.1249/01.MSS.0000117157.06455.B0. PMID 15076791.
- ^ a b Hartgens F, Van Marken Lichtenbelt WD, Ebbing S, Vollaard N, Rietjens G, Kuipers H (April 2001). "Body composition and anthropometry in bodybuilders: regional changes due to nandrolone decanoate administration". International Journal of Sports Medicine. 22 (3): 235–241. doi: 10.1055/s-2001-18679. PMID 11354529. S2CID 260193226.
Further reading
- Geusens P (September 1995). "Nandrolone decanoate: pharmacological properties and therapeutic use in osteoporosis". Clinical Rheumatology. 14 (Suppl 3): 32–39. doi: 10.1007/bf02210686. PMID 8846659. S2CID 24365057.
- Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi: 10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- Hemmersbach P, Grosse J (2010). "Nandrolone: A Multi-Faceted Doping Agent". Doping in Sports. Handbook of Experimental Pharmacology. Vol. 195. Springer. pp. 127–154. doi: 10.1007/978-3-540-79088-4_6. ISBN 978-3-540-79087-7. PMID 20020363.
- Velema MS, Kwa BH, de Ronde W (March 2012). "Should androgenic anabolic steroids be considered in the treatment regime of selected chronic obstructive pulmonary disease patients?". Current Opinion in Pulmonary Medicine. 18 (2): 118–124. doi: 10.1097/MCP.0b013e32834e9001. PMID 22189453. S2CID 6155275.
- Busardò FP, Frati P, Sanzo MD, Napoletano S, Pinchi E, Zaami S, Fineschi V (January 2015). "The impact of nandrolone decanoate on the central nervous system". Current Neuropharmacology. 13 (1): 122–131. doi: 10.2174/1570159X13666141210225822. PMC 4462037. PMID 26074747.
- Wu C, Kovac JR (October 2016). "Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health". Current Urology Reports. 17 (10): 72. doi: 10.1007/s11934-016-0629-8. PMID 27535042. S2CID 43199715.
- Pan MM, Kovac JR (April 2016). "Beyond testosterone cypionate: evidence behind the use of nandrolone in male health and wellness". Translational Andrology and Urology. 5 (2): 213–219. doi: 10.21037/tau.2016.03.03. PMC 4837307. PMID 27141449.
![Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men.[46]](https://upload.wikimedia.org/wikipedia/commons/thumb/0/03/Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png/298px-Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png)
![Nandrolone levels after a single 100 mg intramuscular injection of nandrolone decanoate or nandrolone phenylpropionate in 4 mL or 1 mL arachis oil solution into gluteal or deltoid muscle in men.[47]](https://upload.wikimedia.org/wikipedia/commons/thumb/d/da/Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png/300px-Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png)
![Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men.[48]](https://upload.wikimedia.org/wikipedia/commons/thumb/e/e8/Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png/300px-Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png)
![Dose-normalized nandrolone exposure (serum level divided by dose administered) with nandrolone decanoate in oil solution by intramuscular or subcutaneous injection in men.[49][50]](https://upload.wikimedia.org/wikipedia/commons/thumb/5/50/Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png/300px-Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png)