From Wikipedia, the free encyclopedia
17α-Epiestriol
Names
IUPAC name
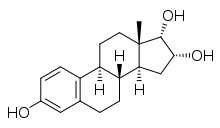
Estra-1,3,5(10)-triene-3,16α,17α-triol
Systematic IUPAC name
(1S ,2R ,3aS ,3bR ,9bS ,11aS )-11a-Methyl-2,3,3a,3b,4,5,9b,10,11,11a-decahydro-1H -cyclopenta[a ]phenanthrene-1,2,7-triol
Other names
17-Epiestriol; 16α-Hydroxy-17α-estradiol; 3,16α,17α-Trihydroxy-1,3,5(10)-estratriene
Identifiers
ChEBI
ChEMBL
ChemSpider
DrugBank
UNII
InChI=1S/C18H24O3/c1-18-7-6-13-12-5-3-11(19)8-10(12)2-4-14(13)15(18)9-16(20)17(18)21/h3,5,8,13-17,19-21H,2,4,6-7,9H2,1H3/t13-,14-,15+,16-,17-,18+/m1/s1
Key: PROQIPRRNZUXQM-PNVOZDDCSA-N
CC12CCC3C(C1CC(C2O)O)CCC4=C3C=CC(=C4)O
Properties
C 18 H 24 O 3
Molar mass
288.38136 g/mol
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
17α-Epiestriol , or simply 17-epiestriol , also known as 16α-hydroxy-17α-estradiol or estra-1,3,5(10)-triene-3,16α,17α-triol , is a minor and weak
endogenous
estrogen , and the 17α-
epimer of
estriol (which is 16α-hydroxy-17β-estradiol).
[1]
[2]
[3] It is formed from
16α-hydroxyestrone .
[4]
[5] In contrast to other endogenous estrogens like
estradiol , 17α-epiestriol is a selective
agonist of the
ERβ .
[6] It is described as a relatively weak estrogen, which is in accordance with its relatively low
affinity for the
ERα .
[7] 17α-Epiestriol has been found to be approximately 400-fold more potent than estradiol in inhibiting
tumor necrosis factor α (TNFα)-induced
vascular cell adhesion molecule 1 (VCAM-1)
expression
in vitro .
[8]
Relative affinities (%) of 17α-epiestriol and related steroids
[9]
[10]
[11]
[12]
Compound
PR Tooltip Progesterone receptor
AR Tooltip Androgen receptor
ER Tooltip Estrogen receptor
GR Tooltip Glucocorticoid receptor
MR Tooltip Mineralocorticoid receptor
SHBG Tooltip Sex hormone-binding globulin
CBG Tooltip Corticosteroid binding globulin
Estradiol
2.6
7.9
100
0.6
0.13
8.7
<0.1
Alfatradiol
<1
<1
15
<1
<1
?
?
Estriol
<1
<1
15
<1
<1
?
?
16β-Epiestriol
<1
<1
20
<1
<1
?
?
17α-Epiestriol
<1
<1
31
<1
<1
?
?
Values are percentages (%). Reference
ligands (100%) were
progesterone for the
PR Tooltip progesterone receptor ,
testosterone for the
AR Tooltip androgen receptor ,
E2 for the
ER Tooltip estrogen receptor ,
DEXA Tooltip dexamethasone for the
GR Tooltip glucocorticoid receptor ,
aldosterone for the
MR Tooltip mineralocorticoid receptor ,
DHT Tooltip dihydrotestosterone for
SHBG Tooltip sex hormone-binding globulin , and
cortisol for
CBG Tooltip Corticosteroid-binding globulin .
See also References
^ Tewari AK (5 April 2013).
Prostate Cancer: A Comprehensive Perspective ISBN
978-1-4471-2864-9
^ Labhart A (6 December 2012).
Clinical Endocrinology: Theory and Practice ISBN
978-3-642-96158-8
^ Assali NS (3 September 2013).
The Maternal Organism ISBN
978-1-4832-6380-9
^ Von Euler US (2 December 2012).
Comparative Endocrinology ISBN
978-0-323-14609-8
^ Tietz NW (1 August 1976).
Fundamentals of clinical chemistry ISBN
978-0-7216-8866-4
^ Sherbet GV (26 July 2013).
Therapeutic Strategies in Cancer Biology and Pathology ISBN
978-0-12-416590-8
^ Dorfman RI (22 October 2013).
Steroidal Activity in Experimental Animals and Man ISBN
978-1-4832-7299-3
^ Mukherjee TK, Nathan L, Dinh H, Reddy ST, Chaudhuri G (April 2003).
"17-epiestriol, an estrogen metabolite, is more potent than estradiol in inhibiting vascular cell adhesion molecule 1 (VCAM-1) mRNA expression" . The Journal of Biological Chemistry . 278 (14): 11746–52.
doi :
10.1074/jbc.M207800200 .
PMID
12547825 .
^ Raynaud, J.P.; Ojasoo, T.; Bouton, M.M.; Philibert, D. (1979).
"Receptor Binding as a Tool in the Development of New Bioactive Steroids" . Drug Design . pp. 169–214.
doi :
10.1016/B978-0-12-060308-4.50010-X .
ISBN
9780120603084
^ Ojasoo T, Raynaud JP (November 1978).
"Unique steroid congeners for receptor studies" . Cancer Research . 38 (11 Pt 2): 4186–98.
PMID
359134 .
^ Ojasoo T, Delettré J, Mornon JP, Turpin-VanDycke C, Raynaud JP (1987). "Towards the mapping of the progesterone and androgen receptors". Journal of Steroid Biochemistry . 27 (1–3): 255–69.
doi :
10.1016/0022-4731(87)90317-7 .
PMID
3695484 .
^ Raynaud JP, Bouton MM, Moguilewsky M, Ojasoo T, Philibert D, Beck G, Labrie F, Mornon JP (January 1980). "Steroid hormone receptors and pharmacology". Journal of Steroid Biochemistry . 12 : 143–57.
doi :
10.1016/0022-4731(80)90264-2 .
PMID
7421203 .
ER Tooltip Estrogen receptor
Agonists
Steroidal:
2-Hydroxyestradiol
2-Hydroxyestrone
3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol
3α-Androstanediol
3α,5α-Dihydrolevonorgestrel
3β,5α-Dihydrolevonorgestrel
3α-Hydroxytibolone
3β-Hydroxytibolone
3β-Androstanediol
4-Androstenediol
4-Androstenedione
4-Fluoroestradiol
4-Hydroxyestradiol
4-Hydroxyestrone
4-Methoxyestradiol
4-Methoxyestrone
5-Androstenediol
7-Oxo-DHEA
7α-Hydroxy-DHEA
7α-Methylestradiol
7β-Hydroxyepiandrosterone
8,9-Dehydroestradiol
8,9-Dehydroestrone
8β-VE2
10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED)
11β-Chloromethylestradiol
11β-Methoxyestradiol
15α-Hydroxyestradiol
16-Ketoestradiol
16-Ketoestrone
16α-Fluoroestradiol
16α-Hydroxy-DHEA
16α-Hydroxyestrone
16α-Iodoestradiol
16α-LE2
16β-Hydroxyestrone
16β,17α-Epiestriol (16β-hydroxy-17α-estradiol)
17α-Estradiol (
alfatradiol )
17α-Dihydroequilenin
17α-Dihydroequilin
17α-Epiestriol (16α-hydroxy-17α-estradiol)
17α-Ethynyl-3α-androstanediol
17α-Ethynyl-3β-androstanediol
17β-Dihydroequilenin
17β-Dihydroequilin
17β-Methyl-17α-dihydroequilenin
Abiraterone
Abiraterone acetate
Alestramustine
Almestrone
Anabolic steroids (e.g.,
testosterone and
esters ,
methyltestosterone ,
metandienone (methandrostenolone) ,
nandrolone and
esters , many others; via estrogenic metabolites)
Atrimustine
Bolandiol
Bolandiol dipropionate
Butolame
Clomestrone
Cloxestradiol
Conjugated estriol
Conjugated estrogens
Cyclodiol
Cyclotriol
DHEA
DHEA-S
ent -Estradiol
Epiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol)
Epimestrol
Equilenin
Equilin
ERA-63 (ORG-37663)
Esterified estrogens
Estetrol
Estradiol
Estramustine
Estramustine phosphate
Estrapronicate
Estrazinol
Estriol
Estrofurate
Estrogenic substances
Estromustine
Estrone
Etamestrol (eptamestrol)
Ethinylandrostenediol
Ethinylestradiol
Ethinylestriol
Ethylestradiol
Etynodiol
Etynodiol diacetate
Hexolame
Hippulin
Hydroxyestrone diacetate
Lynestrenol
Lynestrenol phenylpropionate
Mestranol
Methylestradiol
Moxestrol
Mytatrienediol
Nilestriol
Norethisterone
Noretynodrel
Orestrate
Pentolame
Prodiame
Prolame
Promestriene
RU-16117
Quinestradol
Quinestrol
Tibolone Xenoestrogens:
Anise -related (e.g.,
anethole ,
anol ,
dianethole ,
dianol ,
photoanethole )
Chalconoids (e.g.,
isoliquiritigenin ,
phloretin ,
phlorizin (phloridzin) ,
wedelolactone )
Coumestans (e.g.,
coumestrol ,
psoralidin )
Flavonoids (incl.
7,8-DHF ,
8-prenylnaringenin ,
apigenin ,
baicalein ,
baicalin ,
biochanin A ,
calycosin ,
catechin ,
daidzein ,
daidzin ,
ECG ,
EGCG ,
epicatechin ,
equol ,
formononetin ,
glabrene ,
glabridin ,
genistein ,
genistin ,
glycitein ,
kaempferol ,
liquiritigenin ,
mirificin ,
myricetin ,
naringenin ,
penduletin ,
pinocembrin ,
prunetin ,
puerarin ,
quercetin ,
tectoridin ,
tectorigenin )
Lavender oil
Lignans (e.g.,
enterodiol ,
enterolactone ,
nyasol (cis -hinokiresinol) )
Metalloestrogens (e.g.,
cadmium )
Pesticides (e.g.,
alternariol ,
dieldrin ,
endosulfan ,
fenarimol ,
HPTE ,
methiocarb ,
methoxychlor ,
triclocarban ,
triclosan )
Phytosteroids (e.g.,
digitoxin (
digitalis ),
diosgenin ,
guggulsterone )
Phytosterols (e.g.,
β-sitosterol ,
campesterol ,
stigmasterol )
Resorcylic acid lactones (e.g.,
zearalanone ,
α-zearalenol ,
β-zearalenol ,
zearalenone ,
zeranol (α-zearalanol) ,
taleranol (teranol, β-zearalanol) )
Steroid -like (e.g.,
deoxymiroestrol ,
miroestrol )
Stilbenoids (e.g.,
resveratrol ,
rhaponticin )
Synthetic xenoestrogens (e.g.,
alkylphenols ,
bisphenols (e.g.,
BPA ,
BPF ,
BPS ),
DDT ,
parabens ,
PBBs ,
PHBA ,
phthalates ,
PCBs )
Others (e.g.,
agnuside ,
rotundifuran ) Mixed
SERMs Tooltip Selective estrogen receptor modulators ) Antagonists
Coregulator-binding modulators:
ERX-11
GPER Tooltip G protein-coupled estrogen receptor
Agonists Antagonists Unknown