| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
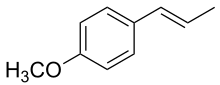
1-Methoxy-4-[(E)-prop-1-enyl]benzene
[1] | |
| Other names
(E)-1-Methoxy-4-(prop-1-en-1-yl)benzene
(E)-1-Methoxy-4-(1-propenyl)benzene para-Methoxyphenylpropene p-Propenylanisole Isoestragole trans-1-Methoxy-4-(prop-1-enyl)benzene | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.914 |
| KEGG | |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C10H12O | |
| Molar mass | 148.205 g/mol |
| Density | 0.998 g/cm3 |
| Melting point | 20 to 21 °C (68 to 70 °F; 293 to 294 K) |
| Boiling point | 234 °C (453 °F; 507 K) 81 °C (178 °F; 354 K) at 2 mmHg |
| −9.60×10−5 cm3/mol | |
| Hazards | |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Related compounds
|
anisole estragole |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Anethole (also known as anise camphor) [2] is an organic compound that is widely used as a flavoring substance. It is a derivative of the aromatic compound allylbenzene and occurs widely in plants in essential oils. It is in the class of phenylpropanoid organic compounds. It contributes a large component of the odor and flavor of anise and fennel (both in the botanical family Apiaceae), anise myrtle ( Myrtaceae), liquorice ( Fabaceae), magnolia blossoms, and star anise ( Schisandraceae). Closely related to anethole is its isomer estragole, which is abundant in tarragon ( Asteraceae) and basil ( Lamiaceae), and has a flavor reminiscent of anise. It is a colorless, fragrant, mildly volatile liquid.[ clarification needed] [3] Anethole is only slightly soluble in water but exhibits high solubility in ethanol. This trait causes certain anise-flavored liqueurs to become opaque when diluted with water; this is called the ouzo effect.
Structure and production
Anethole is an aromatic, unsaturated ether related to lignols. It exists as both cis–trans isomers (see also E–Z notation), involving the double bond outside the ring. The more abundant isomer, and the one preferred for use, is the trans or E isomer. [4]
Like related compounds, anethole is poorly soluble in water. Historically, this property was used to detect adulteration in samples. [5]
Most anethole is obtained from turpentine-like extracts from trees. [3] [6] Of only minor commercial significance, anethole can also be isolated from essential oils. [7] [8] [9]
| Essential oil | World production | Trans-anethole |
|---|---|---|
| Anise | 8 tonnes (1999) | 95% |
| Star anise | 400 tonnes (1999), mostly from China | 87% |
| Fennel | 25 tonnes (1999), mostly from Spain | 70% |
Currently Banwari Chemicals Pvt Ltd situated in Bhiwadi, Rajasthan, India is the leading manufacturer of anethole. It is prepared commercially from 4-methoxy propiophenone, [4] [10] which is prepared from anisole. [3]
Uses
Flavoring
Anethole is distinctly sweet, measuring 13 times sweeter than sugar. It is perceived as being pleasant to the taste even at higher concentrations. It is used in alcoholic drinks ouzo, rakı, anisette and absinthe, among others. It is also used in seasoning and confectionery applications, such as German Lebkuchen, oral hygiene products, and in small quantities in natural berry flavors. [8]
Precursor to other compounds
Because they metabolize anethole into several aromatic chemical compounds, some bacteria are candidates for use in commercial bioconversion of anethole to more valuable materials. [11] Bacterial strains capable of using trans-anethole as the sole carbon source include JYR-1 ( Pseudomonas putida) [12] and TA13 ( Arthrobacter aurescens). [11]
Research
Antimicrobial and antifungal activity
Anethole has potent antimicrobial properties, against bacteria, yeasts, and fungi. [13] Reported antibacterial properties include both bacteriostatic and bactericidal action against Salmonella enterica [14] but not when used against Salmonella via a fumigation method. [15] Antifungal activity includes increasing the effectiveness of some other phytochemicals (such as polygodial) against Saccharomyces cerevisiae and Candida albicans; [16]
In vitro, anethole has antihelmintic action on eggs and larvae of the sheep gastrointestinal nematode Haemonchus contortus. [17] Anethole also has nematicidal activity against the plant nematode Meloidogyne javanica in vitro and in pots of cucumber seedlings. [18]
Insecticidal activity
Anethole also is a promising insecticide. Several essential oils consisting mostly of anethole have insecticidal action against larvae of the mosquito Ochlerotatus caspius [19] and Aedes aegypti. [20] [21] In a similar manner, anethole itself is effective against the fungus gnat Lycoriella ingenua ( Sciaridae) [22] and the mold mite Tyrophagus putrescentiae. [23] Against the mite, anethole is a slightly more effective pesticide than DEET, but anisaldehyde, a related natural compound that occurs with anethole in many essential oils, is 14 times more effective. [23] The insecticidal action of anethole is greater as a fumigant than as a contact agent. trans-Anethole is highly effective as a fumigant against the cockroach Blattella germanica [24] and against adults of the weevils Sitophilus oryzae, Callosobruchus chinensis and beetle Lasioderma serricorne. [25]
As well as an insect pesticide, anethole is an effective insect repellent against mosquitos. [26]
Ouzo effect

Anethole is responsible for the " ouzo effect" (also "louche effect"), the spontaneous formation of a microemulsion [27] [28] that gives many alcoholic beverages containing anethole and water their cloudy appearance. [29] Such a spontaneous microemulsion has many potential commercial applications in the food and pharmaceutical industries. [30]
Precursor to illicit drugs
Anethole is an inexpensive chemical precursor for paramethoxyamphetamine (PMA), [31] and is used in its clandestine manufacture. [32] Anethole is present in the essential oil from guarana, which has psychoactive effects typically attributed to its caffeine content. The absence of PMA or any other known psychoactive derivative of anethole in human urine after ingestion of guarana leads to the conclusion that any psychoactive effect of guarana is not due to aminated anethole metabolites. [33]
Anethole is also present in absinthe, a liquor with a reputation for psychoactive effects; these effects, however, are attributed to ethanol. [34] (See also thujone, anethole dithione (ADT), and anethole trithione (ATT).)
Estrogen and prolactin
Anethole has estrogenic activity. [35] [36] [37] It has been found to significantly increase uterine weight in immature female rats. [38]
Fennel, which contains anethole, has been found to have a galactagogue effect in animals. Anethole bears a structural resemblance to catecholamines like dopamine and may displace dopamine from its receptors and thereby disinhibit prolactin secretion, which in turn may be responsible for the galactagogue effects. [39]
Safety
In the USA, anethole is generally recognized as safe (GRAS). After a hiatus due to safety concerns, anethole was reaffirmed by Flavor and Extract Manufacturers Association (FEMA) as GRAS. [40] The concerns related to liver toxicity and possible carcinogenic activity reported in rats. [41] Anethole is associated with a slight increase in liver cancer in rats, [41] although the evidence is scant and generally regarded as evidence that anethole is not a carcinogen. [41] [42] An evaluation of anethole by the Joint FAO/WHO Expert Committee on Food Additives (JECFA) found its notable pharmacologic properties to be reduction in motor activity, lowering of body temperature, and hypnotic, analgesic, and anticonvulsant effects. [43] A subsequent evaluation by JECFA found some reason for concern regarding carcinogenicity, but there is currently insufficient data to support this. [44] At this time, the JECFA summary of these evaluations is that anethole has "no safety concern at current levels of intake when used as a flavoring agent". [45]
In large quantities, anethole is slightly toxic and may act as an irritant. [46]
History
That an oil could be extracted from anise and fennel had been known since the Renaissance by the German alchemist Hieronymus Brunschwig ( c. 1450 – c. 1512), the German botanist Adam Lonicer (1528–1586), and the German physician Valerius Cordus (1515–1544), among others. [47] Anethole was first investigated chemically by the Swiss chemist Nicolas-Théodore de Saussure in 1820. [48] In 1832, the French chemist Jean Baptiste Dumas determined that the crystallizable components of anise oil and fennel oil were identical, and he determined anethole's empirical formula. [49] In 1845, the French chemist Charles Gerhardt coined the term anethol – from the Latin anethum (anise) + oleum (oil) – for the fundamental compound from which a family of related compounds was derived. [50] Although the German chemist Emil Erlenmeyer proposed the correct molecular structure for anethole in 1866, [51] it was not until 1872, that the structure was accepted as correct. [47]
See also
- Category:Anise liqueurs and spirits
- List of liqueurs § Anise-flavored liqueurs
- Anol
- Chavicol
- Dianethole
- Fenchone
- Pseudoisoeugenol
- Safrole
References
- ^ "Anethole".
- ^ "Anise camphor definition and meaning | Collins English Dictionary".
- ^ a b c Fahlbusch, Karl-Georg; Hammerschmidt, Franz-Josef; Panten, Johannes; Pickenhagen, Wilhelm; Schatkowski, Dietmar; Bauer, Kurt; Garbe, Dorothea; Surburg, Horst. "Flavors and Fragrances". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi: 10.1002/14356007.a11_141. ISBN 978-3527306732.
- ^ a b Zhang, Hongwei; Lim, Candy Li-Fen; Zaki, Muhammad; Jaenicke, Stephan; Chuah, Gaik Khuan (2018). "A Dual-Functional Catalyst for Cascade Meerwein–Pondorf–Verley Reduction and Dehydration of 4′-Methoxypropiophenone to Anethole". ChemSusChem. 11 (17): 3007–3017. doi: 10.1002/cssc.201801340. ISSN 1864-564X. PMID 29927044. S2CID 49342669.
- ^ Waldbott, S. (1920). "Essential oils". Chemical Abstracts. 14 (17): 3753–3755.
- ^ US 4902850, Davis, Curry B., "Purification of anethole by crystallization", issued 1990-02-20
- ^ Chopra, R. N.; Chopra, I. C.; Handa, K. L.; Kapur, L. D. (1958). Chopra's Indigenous Drugs of India (2nd ed.). Academic Publishers. pp. 178–179. ISBN 978-81-85086-80-4.
- ^ a b Ashurst, Philip R. (1999). Food Flavorings. Springer. p. 460. ISBN 978-0-8342-1621-1.[ permanent dead link]
- ^ Bodsgard, B. R.; Lien, N. R.; Waulters, Q. T. (2016). "Liquid CO2 Extraction and NMR Characterization of Anethole from Fennel Seed: A General Chemistry Laboratory". Journal of Chemical Education. 93 (2): 397–400. Bibcode: 2016JChEd..93..397B. doi: 10.1021/acs.jchemed.5b00689.
- ^ Zhang, Hongwei; Quek, Zhan Jiang; Jaenicke, Stephan; Chuah, Gaik-Khuan (2021-08-01). "Hydrophobicity and co-solvent effects on Meerwein-Ponndorf-Verley reduction/dehydration cascade reactions over Zr-zeolite catalysts". Journal of Catalysis. 400: 50–61. doi: 10.1016/j.jcat.2021.05.011. ISSN 0021-9517. S2CID 236342527.
- ^ a b Shimoni, E; Baasov, T.; Ravid, U.; Shoham, Y. (2002). "The trans-anethole degradation pathway in an Arthrobacter sp". Journal of Biological Chemistry. 277 (14): 11866–11872. doi: 10.1074/jbc.M109593200. PMID 11805095.
- ^ Ryu, J.; Seo, J.; Lee, Y.; Lim, Y.; Ahn, J. H.; Hur, H. G. (2005). "Identification of syn- and anti-anethole-2,3-epoxides in the metabolism of trans-anethole by the newly isolated bacterium Pseudomonas putida JYR-1". Journal of Agricultural and Food Chemistry. 53 (15): 5954–5958. doi: 10.1021/jf040445x. PMID 16028980.
- ^ De, M.; De, A. K.; Sen, P.; Banerjee, A. B. (2002). "Antimicrobial properties of star anise (Illicium verum Hook. f.)". Phytotherapy Research. 16 (1): 94–95. doi: 10.1002/ptr.989. PMID 11807977. S2CID 27196549.
- ^ Kubo, I.; Fujita, K. (2001). "Naturally occurring anti-Salmonella agents". Journal of Agricultural and Food Chemistry. 49 (12): 5750–5754. doi: 10.1021/jf010728e. PMID 11743758.
- ^ Weissinger, W. R.; McWatters, K. H.; Beuchat, L. R. (April 2001). "Evaluation of volatile chemical treatments for lethality to Salmonella on alfalfa seeds and sprouts". Journal of Food Protection. 64 (4): 442–450. doi: 10.4315/0362-028X-64.4.442. PMID 11307877.
- ^ Fujita, K.; Fujita, T.; Kubo, I. (2007). "Anethole, a potential antimicrobial synergist, converts a fungistatic dodecanol to a fungicidal agent". Phytotherapy Research. 21 (1): 47–51. doi: 10.1002/ptr.2016. PMID 17078111. S2CID 9666596.
- ^ Camurça-Vasconcelos, A. L.; Bevilaqua, C. M.; Morais, S. M.; Maciel, M. V.; Costa, C. T.; Macedo, I. T.; Oliveira, L. M.; Braga, R. R.; Silva, R. A.; Vieira, L. S. (2007). "Anthelmintic activity of Croton zehntneri and Lippia sidoides essential oils". Veterinary Parasitology. 148 (3–4): 288–294. doi: 10.1016/j.vetpar.2007.06.012. PMID 17629623.
- ^ Oka, Y.; Nacar, S.; Putievsky, E.; Ravid, U.; Yaniv, Z.; Spiegel, Y. (2000). "Nematicidal activity of essential oils and their components against the root-knot nematode". Phytopathology. 90 (7): 710–715. doi: 10.1094/PHYTO.2000.90.7.710. PMID 18944489.
- ^ Knio, K. M.; Usta, J.; Dagher, S.; Zournajian, H.; Kreydiyyeh, S. (2008). "Larvicidal activity of essential oils extracted from commonly used herbs in Lebanon against the seaside mosquito, Ochlerotatus caspius". Bioresource Technology. 99 (4): 763–768. Bibcode: 2008BiTec..99..763K. doi: 10.1016/j.biortech.2007.01.026. PMID 17368893.
- ^ Cheng, S. S.; Liu, J. Y.; Tsai, K. H.; Chen, W. J.; Chang, S. T. (2004). "Chemical composition and mosquito larvicidal activity of essential oils from leaves of different Cinnamomum osmophloeum provenances". Journal of Agricultural and Food Chemistry. 52 (14): 4395–4400. doi: 10.1021/jf0497152. PMID 15237942.
- ^ Morais, S. M.; Cavalcanti, E. S.; Bertini, L. M.; Oliveira, C. L.; Rodrigues, J. R.; Cardoso, J. H. (2006). "Larvicidal activity of essential oils from Brazilian Croton species against Aedes aegypti L.". Journal of the American Mosquito Control Association. 22 (1): 161–164. doi: 10.2987/8756-971X(2006)22[161:LAOEOF]2.0.CO;2. PMID 16646345. S2CID 33429927.
- ^ Park, I. K.; Choi, K. S.; Kim, D. H.; Choi, I. H.; Kim, L. S.; Bak, W. C.; Choi, J. W.; Shin, S. C. (2006). "Fumigant activity of plant essential oils and components from horseradish (Armoracia rusticana), anise (Pimpinella anisum) and garlic (Allium sativum) oils against Lycoriella ingenua (Diptera: Sciaridae)". Pest Management Science. 62 (8): 723–728. doi: 10.1002/ps.1228. PMID 16786497.
- ^ a b Lee, H. S. (2005). "Food protective effect of acaricidal components isolated from anise seeds against the stored food mite, Tyrophagus putrescentiae (Schrank)". Journal of Food Protection. 68 (6): 1208–1210. doi: 10.4315/0362-028X-68.6.1208. PMID 15954709.
- ^ Chang, K. S.; Ahn, Y. J. (2002). "Fumigant activity of (E)-anethole identified in Illicium verum fruit against Blattella germanica". Pest Management Science. 58 (2): 161–166. doi: 10.1002/ps.435. PMID 11852640.
- ^ Kim, D. H.; Ahn, Y. J. (2001). "Contact and fumigant activities of constituents of Foeniculum vulgare fruit against three coleopteran stored-product insects". Pest Management Science. 57 (3): 301–306. doi: 10.1002/ps.274. PMID 11455661.
- ^ Padilha de Paula, J.; Gomes-Carneiro, M. R.; Paumgartten, F. J. (2003). "Chemical composition, toxicity and mosquito repellency of Ocimum selloi oil". Journal of Ethnopharmacology. 88 (2–3): 253–260. doi: 10.1016/s0378-8741(03)00233-2. PMID 12963152.
- ^ Sitnikova, Natalia L.; Sprik, Rudolf; Wegdam, Gerard; Eiser, Erika (2005). "Spontaneously formed trans-anethol/water/alcohol emulsions: Mechanism of formation and stability" (PDF). Langmuir. 21 (16): 7083–7089. doi: 10.1021/la046816l. PMID 16042427. Archived from the original (PDF) on 2009-03-18. Retrieved 2009-03-15.
- ^ Carteau, David; Bassani, Dario; Pianet, Isabelle (2008). "The "Ouzo effect": Following the spontaneous emulsification of trans-anethole in water by NMR". Comptes Rendus Chimie. 11 (4–5): 493–498. doi: 10.1016/j.crci.2007.11.003.
-
^ Sánchez Domínguez, M.; Rodríguez Abreu, C. (2016).
Nanocolloids: A Meeting Point for Scientists and Technologists. Elsevier Science. p. 369.
ISBN
978-0-12-801758-6. Retrieved 2018-08-02.
O/W and W/O nano-emulsions can also be formed without a surfactant by self-emulsification, using the so-called Ouzo effect. The major components of Ouzo (a Greek drink) are trans-anethole, ethanol, and water. Anethole is almost insoluble ...
- ^ Spernath, A.; Aserin, A. (2006). "Microemulsions as carriers for drugs and nutraceuticals". Advances in Colloid and Interface Science. 128–130: 47–64. doi: 10.1016/j.cis.2006.11.016. PMID 17229398.
- ^ Waumans, D.; Bruneel, N.; Tytgat, J. (2003). "Anise oil as para-methoxyamphetamine (PMA) precursor". Forensic Science International. 133 (1–2): 159–170. doi: 10.1016/S0379-0738(03)00063-X. PMID 12742705.
- ^ Waumans, D.; Hermans, B.; Bruneel, N.; Tytgat, J. (2004). "A neolignan-type impurity arising from the peracid oxidation reaction of anethole in the surreptitious synthesis of 4-methoxyamphetamine (PMA)". Forensic Science International. 143 (2–3): 133–139. doi: 10.1016/j.forsciint.2004.02.033. PMID 15240033.
- ^ Benoni, H.; Dallakian, P.; Taraz, K. (1996). "Studies on the essential oil from guarana". Zeitschrift für Lebensmittel-Untersuchung und -Forschung. 203 (1): 95–98. doi: 10.1007/BF01267777. PMID 8765992. S2CID 45636969.
- ^ Lachenmeier, D. W. (2008). "Thujon-Wirkungen von Absinth sind nur eine Legende—Toxikologie entlarvt Alkohol als eigentliche Absinthismus-Ursache" [Thujone-attributable effects of absinthe are only an urban legend—toxicology uncovers alcohol as real cause of absinthism]. Medizinische Monatsschrift für Pharmazeuten (in German). 31 (3): 101–106. PMID 18429531.
- ^ Jordan, Virgil Craig (1986). Estrogen/Antiestrogen Action and Breast Cancer Therapy. University of Wisconsin Press. pp. 21–22. ISBN 978-0-299-10480-1.
- ^ Howes, M.-J. R.; Houghton, P. J.; Barlow, D. J.; Pocock, V. J.; Milligan, S. R. (November 2002). "Assessment of estrogenic activity in some common essential oil constituents". The Journal of Pharmacy and Pharmacology. 54 (11): 1521–1528. doi: 10.1211/002235702216. ISSN 0022-3573. PMID 12495555. S2CID 28650422.
- ^ Albert-Puleo, M. (December 1980). "Fennel and anise as estrogenic agents". Journal of Ethnopharmacology. 2 (4): 337–344. doi: 10.1016/s0378-8741(80)81015-4. ISSN 0378-8741. PMID 6999244.
- ^ Tisserand, Robert; Young, Rodney (2013). Essential Oil Safety: A Guide for Health Care Professionals. Elsevier Health Sciences. p. 150. ISBN 978-0-7020-5434-1.
- ^ Bone, Kerry; Mills, Simon Y. (2013). Principles and Practice of Phytotherapy. Modern Herbal Medicine. Vol. 2. Elsevier Health Sciences. p. 559. ISBN 978-0-443-06992-5.
- ^ Newberne, P.; Smith, R. L.; Doull, J.; Goodman, J. I.; Munro, I. C.; Portoghese, P. S.; Wagner, B. M.; Weil, C. S.; Woods, L. A.; Adams, T. B.; Lucas, C. D.; Ford, R. A. (1999). "The FEMA GRAS assessment of trans-anethole used as a flavouring substance. Flavour and Extract Manufacturers' Association". Food and Chemical Toxicology. 37 (7): 789–811. doi: 10.1016/S0278-6915(99)00037-X. PMID 10496381.
- ^ a b c Newberne, P. M.; Carlton, W. W.; Brown, W. R. (1989). "Histopathological evaluation of proliferative liver lesions in rats fed trans-anethole in chronic studies". Food and Chemical Toxicology. 27 (1): 21–26. doi: 10.1016/0278-6915(89)90087-2. PMID 2467866.
- ^ Waddell, W. J. (2002). "Thresholds of carcinogenicity of flavors". Toxicological Sciences. 68 (2): 275–279. doi: 10.1093/toxsci/68.2.275. PMID 12151622.
- ^ Joint FAO/WHO Expert Committee on Food Additives. "trans-Anethole". WHO Food Additives Series. International Program on Chemical Safety (IPCS).
- ^ Joint FAO/WHO Expert Committee on Food Additives (1998). "trans-Anethole". WHO Food Additives Series. International Program on Chemical Safety (IPCS).
- ^ "Summary of Evaluations Performed by the Joint FAO/WHO Expert Committee on Food Additives: trans-Anethole". International Program on Chemical Safety (IPCS). 2001-11-12. Archived from the original on 2009-03-11. Retrieved 2009-03-10.
- ^ "Safety data for anethole". Physical & Theoretical Chemistry Laboratory Safety, Oxford University. Archived from the original on 2008-06-15. Retrieved 2009-03-10.
- ^
a
b See:
- Semmler, F. W. (1907). Die ätherischen Öle nach ihren chemischen Bestandteilen unter Berücksichtigung der geschichtlichen Entwicklung [The volatile oils according to their chemical components with regard to their historical development] (in German). Vol. 4. Leipzig: Veit & Co. p. 76.
- Lippmann, Edmund Oskar von (1921). "§ 339. Anethol". Zeittafeln zur Geschichte der Organischen Chemie [Timeline of the history of organic chemistry] (in German). Berlin: Springer-Verlag. p. 1. ISBN 9783662246665.
- Gildemeister, Eduard; Hoffmann, Friedrich (1900). "§ 307. Oil of anise". The Volatile Oils. Translated by Kremers, Edward. Milwaukee, Wisconsin: Review Publishing Co. pp. s558–s563.
- ^ De Saussure, N.-T. (1820). "Observations sur la combinaison de l'essence de citron avec l'acide muriatique, et sur quelques substances huileuses" [Observations on the combination of lemon essence with muriatic acid, and on several oily substances]. Annales de Chimie et de Physique. Série 2 (in French). 13: 259–284. See especially pp. 280–284.
-
^ See:
- Dumas, J. (1832). "Mémoire sur les substances végétales qui se rapprochent du camphre, et sur quelques huiles essentielles" [Memoir on plant substances that resemble camphor, and on several essential oils]. Annales de Chimie et de Physique. Série 2 (in French). 50: 225–240. On p. 234, Dumas provides an empirical formula C10H6O1/2 for anethol. If the subscripts are doubled and if the subscript for carbon is then halved (because Dumas, like many of his contemporaries, used the wrong atomic mass for carbon, 6 instead of 12), then Dumas' empirical formula is correct.
- Dumas' finding that the crystallizable components of anise oil and fennel oil were identical was confirmed in 1833 by the team of Rodolphe Blanchet (1807–1864) and Ernst Sell (1808–1854). See: Blanchet, Sell (1833). "Ueber die Zusammensetzung einiger organischer Substanzen" [On the composition of some organic substances]. Annalen der Pharmacie (in German). 6 (3): 259–313. doi: 10.1002/jlac.18330060304. See especially pp. 287–288.
- Dumas' empirical formula for anethole was confirmed in 1841 by the French chemist Auguste Cahours. See: Cahours, A. A. T. (1841). "Sur les essences de fenouil, de badiane et d'anis" [On the essential oils of fennel, star anise, and anise]. Annales de Chimie et de Physique. Série 3 (in French). 2: 274–308. See pp. 278–279. Note that the subscripts of Cahours' empirical formula (C40H24O2) must be divided by 2 and then the subscript for carbon must be divided again by 2 (because, like many chemists of his time, Cahours used the wrong atomic mass for carbon, 6 instead of 12). If these changes are made, the resulting empirical formula is correct.
-
^ Gerhardt, Charles (1845).
"Ueber die Identität des Dragonöls und des Anisöls" [On the identity of tarragon oil and anise oil]. Journal für praktische Chemie (in German). 36: 267–276.
doi:
10.1002/prac.18450360159.
Ich werde keinen neuen Namen für jede einzelne Art der folgenden physisch verschiedenen Arten annehmen. In meinem Werke bezeichne ich sie als Varietäten der Gattung „Anethol".
[I will adopt no new name for any individual species of the following physically different species. In my work, I designate them as varieties of the genus anethol.] - ^ Erlenmeyer, Emil (1866). "Ueber die Constitution des Anisols (Anethols)" [On the constitution of anisol (anethol)]. Zeitschrift für Chemie. 2nd Series (in German). 2: 472–474.
External links
-
 Media related to
Anethole at Wikimedia Commons
Media related to
Anethole at Wikimedia Commons