From Wikipedia, the free encyclopedia
Chemical compound
Quingestanol acetate
Trade names Demovis, Pilomin, others Other names W-4540; Norethisterone acetate 3-cyclopentyl enol ether; 17α-Ethynyl-19-nortestosterone acetate 3-cyclopentyl enol ether; ENTACP; (17β)-3-(Cyclopentyloxy)-17-ethynylestra-3,5-dien-17-yl acetate
Routes of
By mouth
Drug class
Progestogen ;
Progestin ;
Progestogen ester
ATC code
Legal status
In general: ℞ (Prescription only)
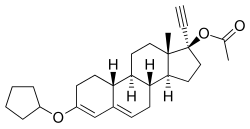
[(8R ,9S ,10R ,13S ,14S ,17R )-3-cyclopentyloxy-17-ethynyl-13-methyl-2,7,8,9,10,11,12,14,15,16-decahydro-1H -cyclopenta[a ]phenanthren-17-yl] acetate
CAS Number
PubChem
CID
DrugBank
ChemSpider
UNII
ChEBI
ChEMBL
CompTox Dashboard (
EPA )
ECHA InfoCard
100.019.163
Formula C 27 H 36 O 3
Molar mass −1 3D model (
JSmol )
O=C(O[C@@]5(C#C)CC[C@@H]4[C@]5(C)CC[C@@H]3[C@@H]2C(\C=C(\OC1CCCC1)CC2)=C/C[C@H]34)C
InChI=1S/C27H36O3/c1-4-27(30-18(2)28)16-14-25-24-11-9-19-17-21(29-20-7-5-6-8-20)10-12-22(19)23(24)13-15-26(25,27)3/h1,9,17,20,22-25H,5-8,10-16H2,2-3H3/t22-,23+,24+,25-,26-,27-/m0/s1
Key:FLGJKPPXEKYCBY-AKCFYGDASA-N
Quingestanol acetate , sold under the brand names Demovis and Pilomin among others, is a
progestin medication which was used in
birth control pills but is no longer marketed.
[1] It is taken
by mouth .
[2]
[3]
[4]
Quingestanol acetate is a progestin, or a
synthetic
progestogen , and hence is an
agonist of the
progesterone receptor , the
biological target of progestogens like
progesterone .
[2]
[3]
[4] It has weak
androgenic and
estrogenic activity and no other important
hormonal activity.
[2]
[3]
[4] The medication is a
prodrug of
norethisterone in the body, with
quingestanol and
norethisterone acetate occurring as
intermediates .
[5]
[6]
Quingestanol acetate was
patented in 1963 and was introduced for medical use in 1972.
[7]
[8] It was marketed in
Italy .
[8]
Medical uses Quingestanol acetate was used as an
oral , once-a-month, or
postcoital
hormonal contraceptive .
[2]
[3]
[4]
Side effects Pharmacology Quingestanol acetate is a
progestogen , and also has weak
androgenic and
estrogenic activity.
[2]
[3]
[4] It is a
prodrug of
norethisterone , with both
quingestanol and
norethisterone acetate serving as
intermediates in the
transformation .
[5]
[6] Unlike
penmesterol (
methyltestosterone 3-cyclopentyl enol ether) and
quinestrol (
ethinylestradiol 3-cyclopentyl ether), quingestanol acetate is not stored in
fat and does not have a prolonged
duration of action .
[2]
Chemistry Quingestanol acetate, also known as norethisterone 17β-acetate 3-cyclopentyl enol ether or as 17α-ethynyl-19-nortestosterone 17β-acetate 3-cyclopentyl enol ether (ENTACP), as well as 3-(cyclopentyloxy)-17α-ethynylestra-3,5-dien-17β-yl acetate, is a
synthetic
estrane
steroid and a
derivative of
testosterone .
[1] It is specifically a derivative of
19-nortestosterone and
17α-ethynyltestosterone , or of
norethisterone (17α-ethynyl-19-nortestosterone), in which a
cyclopentyl
enol ether
group has been attached at the C3 position and an
acetate
ester has been attached at the C17β position.
[1] Quingestanol acetate is the C17β
acetate
ester of
quingestanol (norethisterone 3-cyclopentyl enol ether).
[1]
History Quingestanol acetate was patented in 1963
[7] and marketed in
Italy in 1972.
[8]
[9]
Society and culture Generic names Quingestanol acetate is the
generic name of the drug and its
INN Tooltip International Nonproprietary Name and
USAN Tooltip United States Adopted Name .
[1]
Brand names Quingestanol acetate was marketed under the brand names Demovis, Pilomin, Riglovis, and Unovis.
[1]
[7]
References
^
a b c d e f J. Elks (14 November 2014).
The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies ISBN
978-1-4757-2085-3 ^
a b c d e f Giannina T, Steinetz BG, Rassaert CL, McDougall EA, Meli A (July 1969). "Biological profile of quingestanol acetate". Proceedings of the Society for Experimental Biology and Medicine . 131 (3): 781–9.
doi :
10.3181/00379727-131-33977 .
PMID
5815452 .
S2CID
12433167 . ^
a b c d e Mischler TW, Rubio B, Larranaga A, Guiloff E, Moggia AV (March 1974). "Further experience with quingestanol acetate as a postcoital oral contraceptive". Contraception . 9 (3): 221–5.
doi :
10.1016/0010-7824(74)90013-4 .
PMID
4613534 . ^
a b c d e Donde UM, Virkar KD (June 1975). "Biochemical studies with once-a-month contraceptive pill containing quinestrol-quingestanol acetate". Contraception . 11 (6): 681–8.
doi :
10.1016/0010-7824(75)90065-7 .
PMID
1137940 . ^
a b Raynaud JP, Ojasoo T (1986). "The design and use of sex-steroid antagonists". J. Steroid Biochem . 25 (5B): 811–33.
doi :
10.1016/0022-4731(86)90313-4 .
PMID
3543501 . Similar androgenic potential is inherent to norethisterone and its prodrugs (norethisterone acetate, ethynodiol diacetate, lynestrenol, norethynodrel, quingestanol). ^
a b Di Carlo FJ, Loo JC, Aceto T, Zuleski FR, Barr WH (1974). "Quingestanol acetate metabolism in women". Pharmacology . 11 (5): 287–303.
doi :
10.1159/000136501 .
PMID
4853997 . ^
a b c Lara Marks (2010).
Sexual Chemistry: A History of the Contraceptive Pill ISBN
978-0-300-16791-7 ^
a b c
Population Reports: Oral contraceptives
^ Janne S. Kowalski (1 August 1988).
Drug companies & products world guide ISBN
9780800242398
Androgens
AAS Tooltip anabolic–androgenic steroid )
Antiandrogens
AR Tooltip Androgen receptor
antagonists
Steroidogenesis
Antigonadotropins
D2 receptor
antagonists (
prolactin releasers ) (e.g.,
domperidone ,
metoclopramide ,
risperidone ,
haloperidol ,
chlorpromazine ,
sulpiride )
Estrogens (e.g.,
bifluranol ,
diethylstilbestrol ,
estradiol ,
estradiol esters ,
ethinylestradiol ,
ethinylestradiol sulfonate ,
paroxypropione )
GnRH agonists (e.g.,
leuprorelin )
GnRH antagonists (e.g.,
cetrorelix )
Progestogens (incl.,
chlormadinone acetate ,
cyproterone acetate ,
hydroxyprogesterone caproate ,
gestonorone caproate ,
medroxyprogesterone acetate ,
megestrol acetate ) Others
Estrogens
ER Tooltip Estrogen receptor agonists
Steroidal:
Alfatradiol Certain
androgens /
anabolic steroids (e.g.,
testosterone ,
testosterone esters ,
methyltestosterone ,
metandienone ,
nandrolone esters ) (via estrogenic metabolites)
Certain
progestins (e.g.,
norethisterone ,
noretynodrel ,
etynodiol diacetate ,
tibolone )
Clomestrone
Cloxestradiol acetate
Conjugated estriol
Conjugated estrogens
Epiestriol
Epimestrol
Esterified estrogens
Estetrol †
Estradiol
Estradiol esters (e.g.,
estradiol acetate ,
estradiol benzoate ,
estradiol cypionate ,
estradiol enanthate ,
estradiol undecylate ,
estradiol valerate ,
polyestradiol phosphate ,
estradiol ester mixtures (
Climacteron ))
Estramustine phosphate
Estriol
Estriol esters (e.g.,
estriol succinate ,
polyestriol phosphate )
Estrogenic substances
Estrone
Estrone esters
Ethinylestradiol #
Hydroxyestrone diacetate
Mestranol
Methylestradiol
Moxestrol
Nilestriol
Prasterone (dehydroepiandrosterone; DHEA)
Promestriene
Quinestradol
Quinestrol
Progonadotropins
Antiestrogens
ER Tooltip Estrogen receptor antagonists
SERMs Tooltip selective estrogen receptor modulators /
SERDs Tooltip selective estrogen receptor downregulators )
Aromatase inhibitors
Antigonadotropins
Androgens /
anabolic steroids (e.g.,
testosterone ,
testosterone esters ,
nandrolone esters ,
oxandrolone ,
fluoxymesterone )
D2 receptor
antagonists (
prolactin releasers) (e.g.,
domperidone ,
metoclopramide ,
risperidone ,
haloperidol ,
chlorpromazine ,
sulpiride )
GnRH agonists (e.g.,
leuprorelin ,
goserelin )
GnRH antagonists (e.g.,
cetrorelix ,
elagolix )
Progestogens (e.g.,
chlormadinone acetate ,
cyproterone acetate ,
gestonorone caproate ,
hydroxyprogesterone caproate ,
medroxyprogesterone acetate ,
megestrol acetate ) Others
AR Tooltip Androgen receptor
Agonists
SARMs Tooltip Selective androgen receptor modulator Antagonists
GPRC6A
ER Tooltip Estrogen receptor
Agonists
Steroidal:
2-Hydroxyestradiol
2-Hydroxyestrone
3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol
3α-Androstanediol
3α,5α-Dihydrolevonorgestrel
3β,5α-Dihydrolevonorgestrel
3α-Hydroxytibolone
3β-Hydroxytibolone
3β-Androstanediol
4-Androstenediol
4-Androstenedione
4-Fluoroestradiol
4-Hydroxyestradiol
4-Hydroxyestrone
4-Methoxyestradiol
4-Methoxyestrone
5-Androstenediol
7-Oxo-DHEA
7α-Hydroxy-DHEA
7α-Methylestradiol
7β-Hydroxyepiandrosterone
8,9-Dehydroestradiol
8,9-Dehydroestrone
8β-VE2
10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED)
11β-Chloromethylestradiol
11β-Methoxyestradiol
15α-Hydroxyestradiol
16-Ketoestradiol
16-Ketoestrone
16α-Fluoroestradiol
16α-Hydroxy-DHEA
16α-Hydroxyestrone
16α-Iodoestradiol
16α-LE2
16β-Hydroxyestrone
16β,17α-Epiestriol (16β-hydroxy-17α-estradiol)
17α-Estradiol (
alfatradiol )
17α-Dihydroequilenin
17α-Dihydroequilin
17α-Epiestriol (16α-hydroxy-17α-estradiol)
17α-Ethynyl-3α-androstanediol
17α-Ethynyl-3β-androstanediol
17β-Dihydroequilenin
17β-Dihydroequilin
17β-Methyl-17α-dihydroequilenin
Abiraterone
Abiraterone acetate
Alestramustine
Almestrone
Anabolic steroids (e.g.,
testosterone and
esters ,
methyltestosterone ,
metandienone (methandrostenolone) ,
nandrolone and
esters , many others; via estrogenic metabolites)
Atrimustine
Bolandiol
Bolandiol dipropionate
Butolame
Clomestrone
Cloxestradiol
Conjugated estriol
Conjugated estrogens
Cyclodiol
Cyclotriol
DHEA
DHEA-S
ent -Estradiol
Epiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol)
Epimestrol
Equilenin
Equilin
ERA-63 (ORG-37663)
Esterified estrogens
Estetrol
Estradiol
Estramustine
Estramustine phosphate
Estrapronicate
Estrazinol
Estriol
Estrofurate
Estrogenic substances
Estromustine
Estrone
Etamestrol (eptamestrol)
Ethinylandrostenediol
Ethinylestradiol
Ethinylestriol
Ethylestradiol
Etynodiol
Etynodiol diacetate
Hexolame
Hippulin
Hydroxyestrone diacetate
Lynestrenol
Lynestrenol phenylpropionate
Mestranol
Methylestradiol
Moxestrol
Mytatrienediol
Nilestriol
Norethisterone
Noretynodrel
Orestrate
Pentolame
Prodiame
Prolame
Promestriene
RU-16117
Quinestradol
Quinestrol
Tibolone Xenoestrogens:
Anise -related (e.g.,
anethole ,
anol ,
dianethole ,
dianol ,
photoanethole )
Chalconoids (e.g.,
isoliquiritigenin ,
phloretin ,
phlorizin (phloridzin) ,
wedelolactone )
Coumestans (e.g.,
coumestrol ,
psoralidin )
Flavonoids (incl.
7,8-DHF ,
8-prenylnaringenin ,
apigenin ,
baicalein ,
baicalin ,
biochanin A ,
calycosin ,
catechin ,
daidzein ,
daidzin ,
ECG ,
EGCG ,
epicatechin ,
equol ,
formononetin ,
glabrene ,
glabridin ,
genistein ,
genistin ,
glycitein ,
kaempferol ,
liquiritigenin ,
mirificin ,
myricetin ,
naringenin ,
penduletin ,
pinocembrin ,
prunetin ,
puerarin ,
quercetin ,
tectoridin ,
tectorigenin )
Lavender oil
Lignans (e.g.,
enterodiol ,
enterolactone ,
nyasol (cis -hinokiresinol) )
Metalloestrogens (e.g.,
cadmium )
Pesticides (e.g.,
alternariol ,
dieldrin ,
endosulfan ,
fenarimol ,
HPTE ,
methiocarb ,
methoxychlor ,
triclocarban ,
triclosan )
Phytosteroids (e.g.,
digitoxin (
digitalis ),
diosgenin ,
guggulsterone )
Phytosterols (e.g.,
β-sitosterol ,
campesterol ,
stigmasterol )
Resorcylic acid lactones (e.g.,
zearalanone ,
α-zearalenol ,
β-zearalenol ,
zearalenone ,
zeranol (α-zearalanol) ,
taleranol (teranol, β-zearalanol) )
Steroid -like (e.g.,
deoxymiroestrol ,
miroestrol )
Stilbenoids (e.g.,
resveratrol ,
rhaponticin )
Synthetic xenoestrogens (e.g.,
alkylphenols ,
bisphenols (e.g.,
BPA ,
BPF ,
BPS ),
DDT ,
parabens ,
PBBs ,
PHBA ,
phthalates ,
PCBs )
Others (e.g.,
agnuside ,
rotundifuran ) Mixed
SERMs Tooltip Selective estrogen receptor modulators ) Antagonists
Coregulator-binding modulators:
ERX-11
GPER Tooltip G protein-coupled estrogen receptor
Agonists Antagonists Unknown
PR Tooltip Progesterone receptor
Agonists
Testosterone derivatives: Progestins:
6,6-Difluoronorethisterone
6,6-Difluoronorethisterone acetate
17α-Allyl-19-nortestosterone
Allylestrenol
Altrenogest
Chloroethynylnorgestrel
Cingestol
Danazol
Desogestrel
Dienogest
Ethinylandrostenediol
Ethisterone
Ethynerone
Etonogestrel
Etynodiol
Etynodiol diacetate
Gestodene
Gestrinone
Levonorgestrel
Levonorgestrel esters (e.g.,
levonorgestrel butanoate )
Lynestrenol
Lynestrenol phenylpropionate
Metynodiol
Metynodiol diacetate
Norelgestromin
Norethisterone (norethindrone)
Norethisterone esters (e.g.,
norethisterone acetate ,
norethisterone enanthate )
Noretynodrel
Norgesterone
Norgestimate
Norgestrel
Norgestrienone
Norvinisterone
Oxendolone
Quingestanol
Quingestanol acetate
Tibolone
Tigestol
Tosagestin ; Anabolic–androgenic steroids:
11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone dodecylcarbonate
19-Nor-5-androstenediol
19-Nor-5-androstenedione
19-Nordehydroepiandrosterone
Bolandiol
Bolandiol dipropionate
Bolandione
Dimethisterone
Dienedione
Dienolone
Dimethandrolone
Dimethandrolone buciclate
Dimethandrolone dodecylcarbonate
Dimethandrolone undecanoate
Dimethyldienolone
Dimethyltrienolone
Ethyldienolone
Ethylestrenol (ethylnandrol)
Methyldienolone
Metribolone (R-1881)
Methoxydienone (methoxygonadiene)
Mibolerone
Nandrolone
Nandrolone esters (e.g.,
nandrolone decanoate ,
nandrolone phenylpropionate )
Norethandrolone
Normethandrone (methylestrenolone, normethandrolone, normethisterone)
RU-2309
Tetrahydrogestrinone
Trenbolone (trienolone)
Trenbolone esters (e.g.,
trenbolone acetate ,
trenbolone enanthate )
Trendione
Trestolone
Trestolone acetate Mixed
SPRMs Tooltip Selective progesterone receptor modulators ) Antagonists
mPR Tooltip Membrane progesterone receptor
PAQR Tooltip Progestin and adipoQ receptor )