| |
| Names | |
|---|---|
|
IUPAC name
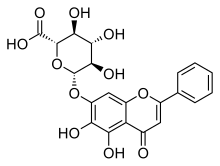
5,6-Dihydroxy-4-oxoflav-2-en-7-yl β-D-glucopyranosiduronic acid
| |
|
Systematic IUPAC name
(2S,3S,4S,5R,6S)-6-[(5,6-Dihydroxy-4-oxo-2-phenyl-4H-1-benzopyran-7-yl)oxy]-3,4,5-trihydroxyoxane-2-carboxylic acid | |
| Other names
Baicalein 7-O-
glucuronide; 5,6-Dihydroxy-4-oxygen-2-phenyl-4H-1-benzopyran-7-β-D-glucopyranose acid
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 70480 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.133.557 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| UNII | |
| |
| |
| Properties | |
| C21H18O11 | |
| Molar mass | 446.364 g·mol−1 |
| Melting point | 202 to 205 °C (396 to 401 °F; 475 to 478 K) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
As baicalin is a flavone glycoside, it is a flavonoid. It is the glucuronide of baicalein.
Natural occurrences
Baicalin is found in several species in the genus Scutellaria, including Scutellaria baicalensis, [1] and Scutellaria lateriflora. There are 10 mg/g baicalin in Scutellaria galericulata leaves. [2] It is also present in the bark isolate of the Oroxylum indicum tree.
Medical uses
Baicalin is one of the chemical ingredients of at least two herbal supplements: Shuanghuanglian [1] and Sho-Saiko-To, which is a Chinese classic herbal formula, and listed in Japan as Kampo medicine.[ citation needed]
Baicalin, along with its aglycone baicalein, is a positive allosteric modulator of the benzodiazepine site and/or a non-benzodiazepine site of the GABAA receptor. [3] [4] [5] In mice, baicalin produces anxiolytic effects without sedative or myorelaxant effects. [6] [7] It is thought that baicalin, along with other flavonoids, may underlie the anxiolytic effects of S. baicalensis and S. lateriflora. [8] [9]
Baicalin is a known prolyl endopeptidase inhibitor. [10] It induces apoptosis in pancreatic cancer cells. [11]
References
- ^ a b Su Hx, Yao S, Zhao WF, Li Mj, Liu J, Shang WJ, Xie H, Ke CQ, Hu HC, Gao Mn, Yu KQ, Liu H, Shen JS, Tang W, Zhang Lk, Xiao Gf, Ni L, Wang Dw, Zuo JP, Jiang HL, Bai F, Wu Y, Ye Y, Xu YC (2020). "Anti-SARS-CoV-2 activities in vitro of Shuanghuanglian preparations and bioactive ingredients". Acta Pharmacologica Sinica. 41 (9): 1167–1177. doi: 10.1038/s41401-020-0483-6. PMC 7393338. PMID 32737471.
- ^ P.H. and Horhammer, L., Hager's Handbuch der Pharmazeutischen Praxis, Vols. 2-6, Springer-Verlag, Berlin, 1969-1979
- ^ Wang H, Hui KM, Xu S, Chen Y, Wong JT, Xue H (2002). "Two flavones from Scutellaria baicalensis Georgi and their binding affinities to the benzodiazepine site of the GABAA receptor complex". Pharmazie. 57 (12): 857–8. PMID 12561253.
- ^ Hui KM, Wang XH, Xue H (2000). "Interaction of flavones from the roots of Scutellaria baicalensis with the benzodiazepine site". Planta Med. 66 (1): 91–3. doi: 10.1055/s-0029-1243121. PMID 10705749. S2CID 260249283.
- ^ Edwin Lowell Cooper, Nobuo Yamaguchi (1 January 2004). Complementary and Alternative Approaches to Biomedicine. Springer Science & Business Media. pp. 188–. ISBN 978-0-306-48288-5.
- ^ Xu Z, Wang F, Tsang SY, Ho KH, Zheng H, Yuen CT, Chow CY, Xue H (2006). "Anxiolytic-Like Effect of baicalin and its additivity with other anxiolytics". Planta Med. 72 (2): 189–92. doi: 10.1055/s-2005-873193. PMID 16491459. S2CID 2398014.
- ^ Liao JF, Hung WY, Chen CF (2003). "Anxiolytic-like effects of baicalein and baicalin in the Vogel conflict test in mice". Eur. J. Pharmacol. 464 (2–3): 141–6. doi: 10.1016/s0014-2999(03)01422-5. PMID 12620506.
- ^ Awad R, Arnason JT, Trudeau V, Bergeron C, Budzinski JW, Foster BC, Merali Z (2003). "Phytochemical and biological analysis of skullcap (Scutellaria lateriflora L.): a medicinal plant with anxiolytic properties". Phytomedicine. 10 (8): 640–9. doi: 10.1078/0944-7113-00374. PMID 14692724.
- ^ Stefanie Schwartz (9 January 2008). Psychoactive Herbs in Veterinary Behavior Medicine. John Wiley & Sons. pp. 139–. ISBN 978-0-470-34434-7.
- ^ Tarragó T, Kichik N, Claasen B, Prades R, Teixidó M, Giralt E (2008). "Baicalin, a prodrug able to reach the CNS, is a prolyl oligopeptidase inhibitor". Bioorganic & Medicinal Chemistry. 16 (15): 7516–24. doi: 10.1016/j.bmc.2008.04.067. PMID 18650094.
- ^ Takahashi H, Chen MC, Pham H, Angst E, King JC, Park J, Brovman EY, Ishiguro H, Harris DM, Reber HA, Hines OJ, Gukovskaya AS, Go VL, Eibl G (2011). "Baicalein, a component of Scutellaria baicalensis, induces apoptosis by Mcl-1 down-regulation in human pancreatic cancer cells". Biochim Biophys Acta. 1813 (8): 1465–1474. doi: 10.1016/j.bbamcr.2011.05.003. PMC 3123440. PMID 21596068.