-
Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men. [38]
-
Nandrolone levels after a single 100 mg intramuscular injection of nandrolone decanoate or nandrolone phenylpropionate in 4 mL or 1 mL arachis oil solution into gluteal or deltoid muscle in men. [39]
-
Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men. [40]
-
Dose-normalized nandrolone exposure (serum level divided by dose administered) with nandrolone decanoate in oil solution by intramuscular or subcutaneous injection in men. [41] [42]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈnændrəloʊn/ [1] |
| Trade names | • Deca-Durabolin (as
ND) • Durabolin (as NPP) • Many others (see here) |
| Other names | • 19-Nortestosterone
[2]
[3] • 10-Nortestosterone • Estr-4-en-17β-ol-3-one • Estrenolone • Oestrenolone • 19-Norandrost-4-en-17β-ol-3-one • Norandrostenolone [2] • Nortestrionate [2] • Nortestonate [2] • Norandroone • SG-4341 [2] [3] |
|
Pregnancy category |
|
|
Routes of administration | •
IM injection (
esters) • SC injection ( esters) • Eye drops ( NS) |
| Drug class | Androgen; Anabolic steroid; Progestogen |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | •
Oral: <3% (pigs)
[5] • Intramuscular: high [6] |
| Metabolism | Liver ( reduction) [7] [9] |
| Metabolites | •
5α-Dihydronandrolone
[7]
[8] • 19-Norandrosterone [7] • 19-Noretiocholanolone [7] • Conjugates [9] |
| Elimination half-life | • Nandrolone: <4.3 hours
[7] • ND ( IM): 6–12 days [7] [8] [10] • NPP: 2.7 days [10] |
| Duration of action | •
ND (
IM): 2–3 weeks
[8]
[11] • NPP ( IM): 5–7 days [8] [10] |
| Excretion | Urine [7] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.006.457 |
| Chemical and physical data | |
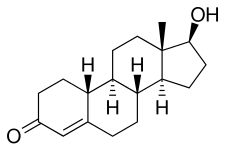
| Formula | C18H26O2 |
| Molar mass | 274.404 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Nandrolone, also known as 19-nortestosterone, is an endogenous androgen which exists in the male body at a ratio of 1:50 compared to testosterone.[ citation needed] It is also an anabolic steroid (AAS) which is medically used in the form of esters such as nandrolone decanoate (brand name Deca-Durabolin) and nandrolone phenylpropionate (brand name Durabolin). [2] [12] [8] [13] Nandrolone esters are used in the treatment of anemias, cachexia ( muscle wasting syndrome), osteoporosis, breast cancer, and for other indications. [8] They are now used by oral administration or instead are given by injection into muscle or fat. [8] [13] [14]
Side effects of nandrolone esters include symptoms of masculinization like acne, increased hair growth, and voice changes. [8] They are synthetic androgens and anabolic steroids and hence are agonists of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT). [8] [15] Nandrolone has strong anabolic effects and weak androgenic effects, which give them a mild side effect profile and make them especially suitable for use in women and children. [8] [15] [16] There are metabolites of Nandrolone that act as long-lasting prodrugs in the body, [8] such as 5α-Dihydronandrolone.
Nandrolone esters were first described and introduced for medical use in the late 1950s. [8] They are among the most widely used anabolic steroid worldwide. [8] In addition to their medical use, nandrolone esters are used to improve physique and performance, and are said to be the most widely used anabolic steroid for such purposes. [8] [17] The drugs are controlled substances in many countries and so non-medical use is generally illicit. [8]
Medical uses
Nandrolone esters are used clinically, although increasingly rarely, for people in catabolic states with major burns, cancer, and AIDS, and an ophthalmological formulation was available to support cornea healing. [18]: 134
The positive effects of nandrolone esters include muscle growth, appetite stimulation and increased red blood cell production,[ medical citation needed] and bone density. [19] Clinical studies have shown them to be effective in treating anemia, osteoporosis, and breast cancer.
Nandrolone sulfate has been used in an eye drop formulation as an ophthalmic medication. [2] [12]
Non-medical uses
Nandrolone esters are used for physique- and performance-enhancing purposes by competitive athletes, bodybuilders, and powerlifters. [8]
Side effects
Side effects of nandrolone esters include masculinization among others. [8] In women, nandrolone and nandrolone esters have been reported to produce increased libido, acne, facial and body hair growth, voice changes, and clitoral enlargement. [20] However, the masculinizing effects of nandrolone and its esters are reported to be slighter than those of testosterone. [20] Nandrolone has also been found to produce penile growth in prepubertal boys. [20] Amenorrhea and menorrhagia have been reported as side effects of nandrolone cypionate. [20]
Nandrolone theoretically may produce erectile dysfunction as a side effect, although there is no clinical evidence to support this notion at present. [21] Side effects of high doses of nandrolone may include cardiovascular toxicity as well as hypogonadism and infertility.[ citation needed] Nandrolone may not produce scalp hair loss, although this is also theoretical. [21]
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone ( DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Nandrolone is an agonist of the AR, the biological target of androgens like testosterone and DHT. Unlike testosterone and certain other anabolic steroids, nandrolone is not potentiated in androgenic tissues like the scalp, skin, and prostate, hence deleterious effects in these tissues are lessened. [22] This is because nandrolone is metabolized by 5α-reductase to the much weaker AR ligand 5α-dihydronandrolone (DHN), which has both reduced affinity for the androgen receptor (AR) relative to nandrolone in vitro and weaker AR agonistic potency in vivo. [22] The lack of alkylation on the 17α-carbon drastically reduces the hepatotoxic potential of nandrolone.[ medical citation needed] Estrogen effects resulting from reaction with aromatase are also reduced due to lessened enzyme interaction, [23] but effects such as gynecomastia and reduced libido may still occur at sufficiently high doses.[ citation needed]
In addition to its AR agonistic activity, unlike many other anabolic steroids, nandrolone is also a potent progestogen. [24] It binds to the progesterone receptor with approximately 22% of the affinity of progesterone. [24] The progestogenic activity of nandrolone serves to augment its antigonadotropic effects, [25] [8] as antigonadotropic action is a known property of progestogens. [26] [27]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG |
|---|---|---|---|---|---|---|---|
| Nandrolone | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1–16 | 0.1 |
| Testosterone | 1.0–1.2 | 100 | <0.1 | 0.17 | 0.9 | 19–82 | 3–8 |
| Estradiol | 2.6 | 7.9 | 100 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PR, testosterone for the AR, estradiol for the ER, dexamethasone for the GR, aldosterone for the MR, dihydrotestosterone for SHBG, and cortisol for CBG. Sources: See template. | |||||||
Anabolic and androgenic activity
Nandrolone has a very high ratio of anabolic to androgenic activity. [15] In fact, many nandrolone-like anabolic steroids and even nandrolone itself are said to have among the highest ratio of anabolic to androgenic effect of all anabolic steroids. [25] This is attributed to the fact that whereas testosterone is potentiated via conversion into dihydrotestosterone (DHT) in androgenic tissues, the opposite is true with nandrolone and similar anabolic steroids (i.e., other 19-nortestosterone derivatives). [15] As such, nandrolone-like anabolic steroids, namely nandrolone esters, are the most frequently used anabolic steroids in clinical settings in which anabolic effects are desired; for instance, in the treatment of AIDS-associated cachexia, severe burns, and chronic obstructive pulmonary disease. [25] However, anabolic steroids with a very high ratio of anabolic to androgenic action like nandrolone still have significant androgenic effects and can produce symptoms of masculinization like hirsutism and voice deepening in women and children with extended use. [15]
| Compound | rAR (%) | hAR (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Testosterone | 38 | 38 | ||||||
| 5α-Dihydrotestosterone | 77 | 100 | ||||||
| Nandrolone | 75 | 92 | ||||||
| 5α-Dihydronandrolone | 35 | 50 | ||||||
| Ethylestrenol | ND | 2 | ||||||
| Norethandrolone | ND | 22 | ||||||
| 5α-Dihydronorethandrolone | ND | 14 | ||||||
| Metribolone | 100 | 110 | ||||||
| Sources: See template. | ||||||||
Pharmacokinetics
The oral activity of nandrolone has been studied. [28] [29] [30] [31] [5] [32] With oral administration of nandrolone in rodents, it had about one-tenth of the potency of subcutaneous injection of nandrolone. [28] [33] [20]
Nandrolone has very low affinity for human serum sex hormone-binding globulin (SHBG), about 5% of that of testosterone and 1% of that of DHT. [34] It is metabolized by the enzyme 5α-reductase, among others. [35][ additional citation(s) needed] Nandrolone is less susceptible to metabolism by 5α-reductase and 17β-hydroxysteroid dehydrogenase than testosterone. [35] This results in it being transformed less in so-called "androgenic" tissues like the skin, hair follicles, and prostate gland and in the kidneys, respectively. [35] Metabolites of nandrolone include 5α-dihydronandrolone, 19-norandrosterone, and 19-noretiocholanolone, and these metabolites may be detected in urine. [36]
Single intramuscular injections of 100 mg nandrolone phenylpropionate or nandrolone decanoate have been found to produce an anabolic effect for 10 to 14 days and 20 to 25 days, respectively. [37] Conversely, unesterified nandrolone has been used by intramuscular injection once daily. [20] [33]
Chemistry

Nandrolone, also known as 19-nortestosterone (19-NT) or as estrenolone, as well as estra-4-en-17β-ol-3-one or 19-norandrost-4-en-17β-ol-3-one, [43] is a naturally occurring estrane (19-norandrostane) steroid and a derivative of testosterone (androst-4-en-17β-ol-3-one). [2] [12] It is specifically the C19 demethylated ( nor) analogue of testosterone. [2] [12] Nandrolone is an endogenous intermediate in the production of estradiol from testosterone via aromatase in mammals including humans and is present in the body naturally in trace amounts. [44] It can be detected during pregnancy in women. [45] Nandrolone esters have an ester such as decanoate or phenylpropionate attached at the C17β position. [2] [12]
Derivatives
Esters
A variety of esters of nandrolone have been marketed and used medically. [2] [12] The most commonly used esters are nandrolone decanoate and to a lesser extent nandrolone phenylpropionate. Examples of other nandrolone esters that have been marketed and used medically include nandrolone cyclohexylpropionate, nandrolone cypionate, nandrolone hexyloxyphenylpropionate, nandrolone laurate, nandrolone sulfate, and nandrolone undecanoate. [2] [12] [8]
Anabolic steroids
Nandrolone is the parent compound of a large group of anabolic steroids. Notable examples include the non-17α-alkylated trenbolone and the 17α-alkylated ethylestrenol (ethylnandrol) and metribolone (R-1881), as well as the 17α-alkylated designer steroids norboletone and tetrahydrogestrinone (THG). The following is list of derivatives of nandrolone that have been developed as anabolic steroids: [8]
|
|
Progestins
Nandrolone, together with ethisterone (17α-ethynyltestosterone), is also the parent compound of a large group of progestins, the norethisterone (17α-ethynyl-19-nortestosterone) derivatives. [46] [47] This family is subdivided into two groups: the estranes and the gonanes. [46] The estranes include norethisterone (norethindrone), norethisterone acetate, norethisterone enanthate, lynestrenol, etynodiol diacetate, and noretynodrel, while the gonanes include norgestrel, levonorgestrel, desogestrel, etonogestrel, gestodene, norgestimate, dienogest (actually a 17α-cyanomethyl-19-nortestosterone derivative), and norelgestromin. [46]
Synthesis
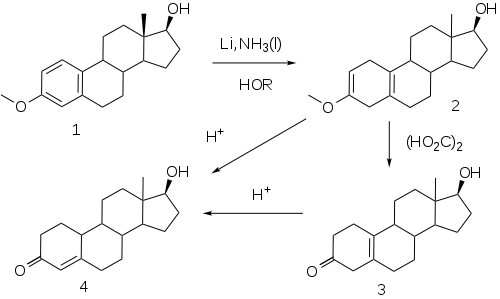
The elaboration of a method for the reduction of aromatic rings to the corresponding dihydrobenzenes under controlled conditions by A. J. Birch opened a convenient route to compounds related to the putative 19-norprogesterone.
This reaction, now known as the Birch reduction, [51] is typified by the treatment of the monomethyl ether of estradiol (1) with a solution of lithium metal in liquid ammonia in the presence of alcohol as a proton source. Initial reaction constituents of 1,4-dimetalation of the most electron deficient positions of the aromatic ring–in the case of an estrogen, the 1 and 4-positions. Rxn of the intermediate with the proton source leads to a dihydrobenzene; a special virtue of this sequence in steroids is the fact that the double bind at 2 is in effect becomes an enol ether moiety. Treatment of this product (2) with weak acid, oxalic acid for e.g., leads to the hydrolysis of the enol ether, producing β,γ-unconjugated ketone 3. Hydrolysis under more strenuous conditions ( mineral acids) results in migration/conjugation of the olefin to yield nandrolone (4).
Esters
- Treatment of 4 with decanoic anhydride and pyridine affords nandrolone decanoate. [52]
- Acylation of 4 with phenylpropionyl chloride yields nandrolone phenpropionate. [53]
Detection in body fluids
Nandrolone use is directly detectable in hair or indirectly detectable in urine by testing for the presence of 19-norandrosterone, a metabolite. The International Olympic Committee has set a limit of 2.0 μg/L of 19-norandrosterone in urine as the upper limit, [54] beyond which an athlete is suspected of doping. In the largest nandrolone study performed on 621 athletes at the 1998 Nagano Olympic Games, no athlete tested over 0.4 μg/L. 19-Norandrosterone was identified as a trace contaminant in commercial preparations of androstenedione, which until 2004 was available without a prescription as a dietary supplement in the U.S. [55] [56] [57] [58]
A number of nandrolone cases in athletics occurred in 1999, which included high-profile athletes such as Merlene Ottey, Dieter Baumann, and Linford Christie. [59] However, the following year the detection method for nandrolone at the time was proved to be faulty. Mark Richardson, a British Olympic relay runner who tested positive for the substance, gave a significant amount of urine samples in a controlled environment and delivered a positive test for the drug, demonstrating that false positives could occur, which led to an overhaul of his competitive ban. [60]
Heavy consumption of the essential amino acid lysine (as indicated in the treatment of cold sores) has allegedly shown false positives in some and was cited by American shotputter C. J. Hunter as the reason for his positive test, though in 2004 he admitted to a federal grand jury that he had injected nandrolone. [61] A possible cause of incorrect urine test results is the presence of metabolites from other anabolic steroids, though modern urinalysis can usually determine the exact anabolic steroid used by analyzing the ratio of the two remaining nandrolone metabolites. As a result of the numerous overturned verdicts, the testing procedure was reviewed by UK Sport. In October 2007, three-time Olympic gold medalist for track and field Marion Jones admitted to use of the drug, and was sentenced to six months in jail for lying to a federal grand jury in 2000. [62]
Mass spectrometry is also used to detect small amounts of nandrolone in urine samples. [63]
History

Nandrolone was first synthesized in 1950. [2] [43] [18]: 130 [64] It was first introduced, as nandrolone phenylpropionate, in 1959, and then as nandrolone decanoate in 1962, followed by additional esters. [65]
Society and culture
Generic names
Nandrolone is the generic name of the drug and its INN, BAN, DCF, and DCIT. [2] [12] [3] [66] The formal generic names of nandrolone esters include nandrolone cyclohexylpropionate ( BANM), nandrolone cyclotate ( USAN), nandrolone decanoate ( USAN, USP, BANM, JAN), nandrolone laurate ( BANM), nandrolone phenpropionate ( USP), and nandrolone phenylpropionate ( BANM, JAN). [2] [12] [3] [66]
Doping in sports
Nandrolone was probably among the first anabolic steroids to be used as a doping agent in sports in the 1960s.[ citation needed] It has been banned at the Olympics since 1974. [18]: 128 There are many known cases of doping in sports with nandrolone esters by professional athletes.
Research
Nandrolone esters have been studied in several indications. They were intensively studied for osteoporosis, and increased calcium uptake and decreased bone loss, but caused virilization in about half of the women who took them and were mostly abandoned for this use when better drugs like the bisphosphonates became available. [21] They have also been studied in clinical trials for chronic kidney failure, aplastic anemia, and as male contraceptives. [18]: 134
References
- ^ "Nandrolone Meaning in the Cambridge English Dictionary".
- ^ a b c d e f g h i j k l m n o Elks J, Ganellin CR, eds. (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. New York City: Springer. pp. 660–. ISBN 978-1-4757-2085-3. OCLC 1079003025.
- ^ a b c d Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ^ a b McEvoy JD, McVeigh CE, McCaughey WJ (December 1998). "Residues of nortestosterone esters at injection sites. Part 1. Oral bioavailability". The Analyst. 123 (12): 2475–8. doi: 10.1039/a804919j. PMID 10435281.
- ^ Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1185–. ISBN 978-0-7817-1750-2.
- ^ a b c d e f g "Deca-Durabolin" (PDF). Schering-Plough. Archived from the original (PDF) on 18 December 2010.
- ^ a b c d e f g h i j k l m n o p q r s t Llewellyn W (2011). Anabolics. Molecular Nutrition Llc. pp. 402–412, 460–467, 193–194. ISBN 978-0-9828280-1-4.
- ^ a b Thomas JA (6 December 2012). Drugs, Athletes, and Physical Performance. Springer Science & Business Media. pp. 27–29. ISBN 978-1-4684-5499-4.
- ^ a b c Minto CF, Howe C, Wishart S, Conway AJ, Handelsman DJ (April 1997). "Pharmacokinetics and pharmacodynamics of nandrolone esters in oil vehicle: effects of ester, injection site and injection volume". The Journal of Pharmacology and Experimental Therapeutics. 281 (1): 93–102. PMID 9103484.
- ^ "Deca-Durabolin" (PDF). Merck Sharp & Dohme (Australia).
- ^ a b c d e f g h i Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 716–. ISBN 978-3-88763-075-1.
- ^ a b Sneader W (23 June 2005). Drug Discovery: A History. John Wiley & Sons. pp. 206–. ISBN 978-0-471-89979-2.
- ^ Singh GK, Turner L, Desai R, Jimenez M, Handelsman DJ (July 2014). "Pharmacokinetic-pharmacodynamic study of subcutaneous injection of depot nandrolone decanoate using dried blood spots sampling coupled with ultrapressure liquid chromatography tandem mass spectrometry assays". The Journal of Clinical Endocrinology and Metabolism. 99 (7): 2592–8. doi: 10.1210/jc.2014-1243. PMID 24684468.
- ^ a b c d e Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. doi: 10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ^ Kochakian CD (6 December 2012). Anabolic-Androgenic Steroids. Springer Science & Business Media. pp. 401–. ISBN 978-3-642-66353-6.
- ^ Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. pp. 2388–. ISBN 978-0-323-32195-2.
- ^ a b c d Hemmersbach P, Große J (2009). "Nandrolone: A Multi-Faceted Doping Agent". In Thieme D, Hemmersbach P (eds.). Doping in sports. Berlin: Springer. pp. 127–154. ISBN 978-3-540-79088-4.
-
^ Handelsman DJ (2013).
"Androgen Physiology, Pharmacology and Abuse". In De Groot LJ (ed.). Endotext. MDText.com. 4.1.2 Pharmacologic Androgen Therapy.
PMID
25905160 – via NCBI Bookshelf.
Both testosterone and its non-aromatizable derivative nandrolone, produce increased bone density in men with glucocorticoid-induced osteoporosis with minimal short-term side-effects....
- ^
a
b
c
d
e
f Camerino B, Sala G (1960). "Anabolic Steroids". Fortschritte der Arzneimittelforschung / Progress in Drug Research / Progrès des recherches pharmaceutiques. Vol. 2. pp. 71–134.
doi:
10.1007/978-3-0348-7038-2_2.
ISBN
978-3-0348-7040-5.
PMID
14448579.
{{ cite book}}:|journal=ignored ( help) - ^ a b c Pan MM, Kovac JR (April 2016). "Beyond testosterone cypionate: evidence behind the use of nandrolone in male health and wellness". Translational Andrology and Urology. 5 (2): 213–9. doi: 10.21037/tau.2016.03.03. PMC 4837307. PMID 27141449.
- ^ a b Bergink EW, Janssen PS, Turpijn EW, van der Vies J (June 1985). "Comparison of the receptor binding properties of nandrolone and testosterone under in vitro and in vivo conditions". Journal of Steroid Biochemistry. 22 (6): 831–6. doi: 10.1016/0022-4731(85)90293-6. PMID 4021486.
- ^ Brueggemeier RW (September 16, 2006). "Sex Hormones (Male): Analogs and Antagonists". In Meyers RA (ed.). Encyclopedia of Molecular Cell Biology and Molecular Medicine (abstract). John Wiley & Sons. doi: 10.1002/3527600906.mcb.200500066. ISBN 978-3527600908.
- ^ a b Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration". Climacteric. 8 (Suppl 1): 3–63. doi: 10.1080/13697130500148875. PMID 16112947. S2CID 24616324.
- ^ a b c de Souza GL, Hallak J (December 2011). "Anabolic steroids and male infertility: a comprehensive review". BJU International. 108 (11): 1860–5. doi: 10.1111/j.1464-410X.2011.10131.x. PMID 21682835. S2CID 29035729.
- ^ Couzinet B, Young J, Brailly S, Chanson P, Thomas JL, Schaison G (December 1996). "The antigonadotropic activity of progestins (19-nortestosterone and 19-norprogesterone derivatives) is not mediated through the androgen receptor". The Journal of Clinical Endocrinology and Metabolism. 81 (12): 4218–23. doi: 10.1210/jcem.81.12.8954018. PMID 8954018.
- ^ Mauvais-Jarvis, P. "Progesterone and progestins: a general overview." (1983): 1-16.
- ^ a b Camerino B, Sciaky R (1975). "Structure and effects of anabolic steroids". Pharmacology & Therapeutics B. 1 (2): 233–75. doi: 10.1016/0306-039X(75)90007-0. PMID 817322.
- ^ Holtkamp DE, Heming AE, Mansor LF (1955). "Comparison of Oral and Subcutaneous Administration in the Anabolic and Androgenic Effectiveness of 19-Nortestosterone and Testosterone". The Journal of Clinical Endocrinology & Metabolism. 15 (7): 848. doi: 10.1210/jcem-15-7-834. ISSN 0021-972X.
- ^ Furman RH, Howard RP, Smith CW, Norcia LN (January 1956). "Comparison of the effects of oral methyltestosterone, 19-nortestosterone, and 17-methyl-19-nortestosterone on serum lipids and lipoproteins". Journal of Laboratory and Clinical Medicine. 48 (5): 808–809.
- ^ Furman RH, Howard RP, Norcia LN, Keaty EC (January 1958). "The influence of androgens, estrogens and related steroids on serum lipids and lipoproteins". The American Journal of Medicine. 24 (1): 80–97. doi: 10.1016/0002-9343(58)90364-4. PMID 13498038.
- ^ Arnold A, Potts GO (January 1964). "Oral anabolic and androgenic effects of 19-nortestosterone and 17α-methyl-19-nortestosterone". Federation Proceedings. 23 (2): 412.
- ^ a b Baker WH, Henneman PH, Baggett B, Engel LL, Tibbetts D, Brown M (January 1955). "Metabolic effect of 19-nortestosterone". Journal of Clinical Endocrinology & Metabolism. 15 (7): 848–849. doi: 10.1210/jcem-15-7-834. ISSN 0021-972X.
- ^ Saartok T, Dahlberg E, Gustafsson JA (June 1984). "Relative binding affinity of anabolic-androgenic steroids: comparison of the binding to the androgen receptors in skeletal muscle and in prostate, as well as to sex hormone-binding globulin". Endocrinology. 114 (6): 2100–6. doi: 10.1210/endo-114-6-2100. PMID 6539197.
- ^ a b c Bergink EW, Geelen JA, Turpijn EW (1985). "Metabolism and receptor binding of nandrolone and testosterone under in vitro and in vivo conditions". Acta Endocrinologica. Supplementum. 271 (3_Suppla): 31–7. doi: 10.1530/acta.0.109S0031. PMID 3865479.
- ^ Mottram DR (12 November 2010). Drugs in Sport. Routledge. pp. 63–. ISBN 978-1-135-25825-2.
- ^ Dorfman RI (5 December 2016). Steroidal Activity in Experimental Animals and Man. Elsevier Science. pp. 68–. ISBN 978-1-4832-7300-6.
- ^ Bagchus WM, Smeets JM, Verheul HA, De Jager-Van Der Veen SM, Port A, Geurts TB (2005). "Pharmacokinetic evaluation of three different intramuscular doses of nandrolone decanoate: analysis of serum and urine samples in healthy men". J. Clin. Endocrinol. Metab. 90 (5): 2624–30. doi: 10.1210/jc.2004-1526. PMID 15713722.
- ^ Minto CF, Howe C, Wishart S, Conway AJ, Handelsman DJ (1997). "Pharmacokinetics and pharmacodynamics of nandrolone esters in oil vehicle: effects of ester, injection site and injection volume". J. Pharmacol. Exp. Ther. 281 (1): 93–102. PMID 9103484.
- ^ Belkien L, Schürmeyer T, Hano R, Gunnarsson PO, Nieschlag E (May 1985). "Pharmacokinetics of 19-nortestosterone esters in normal men". J. Steroid Biochem. 22 (5): 623–9. doi: 10.1016/0022-4731(85)90215-8. PMID 4010287.
- ^ Kalicharan RW, Schot P, Vromans H (February 2016). "Fundamental understanding of drug absorption from a parenteral oil depot". Eur J Pharm Sci. 83: 19–27. doi: 10.1016/j.ejps.2015.12.011. PMID 26690043.
- ^ Kalicharan, Raween Wikesh (2017). New Insights into Drug Absorption from Oil Depots (PhD). Utrecht University.
- ^ a b Schnitzer R (1 January 1967). Experimental Chemotherapy. Elsevier Science. pp. 165–. ISBN 978-0-323-14611-1.
- ^ Bricout V, Wright F (June 2004). "Update on nandrolone and norsteroids: how endogenous or xenobiotic are these substances?". European Journal of Applied Physiology. 92 (1–2): 1–12. doi: 10.1007/s00421-004-1051-3. PMID 15042372. S2CID 6472015.
- ^ Lippi G, Franchini M, Banfi G (May 2011). "Biochemistry and physiology of anabolic androgenic steroids doping". Mini Reviews in Medicinal Chemistry. 11 (5): 362–73. doi: 10.2174/138955711795445952. PMID 21443514. S2CID 3862299.
- ^ a b c Schindler AE, Campagnoli C, Druckmann R, Huber J, Pasqualini JR, Schweppe KW, Thijssen JH (December 2003). "Classification and pharmacology of progestins". Maturitas. 46 (Suppl 1): S7–S16. doi: 10.1016/j.maturitas.2003.09.014. PMID 14670641.
-
^ Meikle AW (24 April 2003).
Endocrine Replacement Therapy in Clinical Practice. Springer Science & Business Media. pp. 489–.
ISBN
978-1-59259-375-0.
Estranes. Estrane and gonane progestogens are derived from 19-nortestosterone, the progestogenic parent compound used in oral contraceptives in the United States. Estranes are characterized by the presence of an ethinyl group at position 17 and by the absence of a methyl group between the A and B rings (see Fig. 10). The estrane progestogens that are related structurally to norethindrone (norethynodrel, lynestrenol, norethindrone acetate, ethynodiol diacetate) are converted to this parent compound. Norethindrone is the second most commonly used progestogen in the United States for HRT. Gonanes. The gonanes share the structural modifications found in the estranes and also possess an ethinyl group at position 13 and a keto group at position 3 (see Fig. 11). Norgestrel was synthesized in 1963 and is a racemic mixture of dextro and levorotatory forms. The levorotatory form, levonorgestrel, provides the biologic activity. Third-generation gonanes (desogestrel, gestodene, and norgestimate) have been developed to reduce unwanted side effects of progestogens, [...]
- ^ Wilds AL, Nelson NA (1953). "The Facile Synthesis of 19-Nortestosterone and 19-Norandrostenedione from Estrone". Journal of the American Chemical Society. 75 (21): 5366–5369. doi: 10.1021/ja01117a065.
- ^ Ueberwasser H, Heusler K, Kalvoda J, Meystre CH, Wieland P, Anner G, Wettstein A (1963). "19-Norsteroide II. Ein einfaches Herstellungsverfahren für 19-Norandrostan-Derivate. Über Steroide, 193. Mitteilung". Helvetica Chimica Acta. 46: 344–352. doi: 10.1002/hlca.19630460135.
- ^ Shimizu I, Naito Y, Tsuji J (1980). "Synthesis of optically active (+)-19-nortestosterone by asymmetric bis-annulation reaction". Tetrahedron Letters. 21 (5): 487–490. doi: 10.1016/S0040-4039(00)71440-7.
- ^ Birch AJ (1950). "The reduction of organic compounds by metal-ammonia solutions". Quarterly Reviews, Chemical Society. 4: 69. doi: 10.1039/QR9500400069.
- ^ US 2998423, DeWytt ED, Overbeek O, Overbeek GA, issued 1961, assigned to Organon.
- ^ CH 206119, issued 1939, assigned to Gesellschaft für Chemische Industrie Basel
- ^ "Clarification about Nandrolone Testing". World Anti-Doping Agency. 2005-05-30. Archived from the original on 2012-09-15. Retrieved 2012-01-31.
- ^ Bresson M, Cirimele V, Villain M, Kintz P (May 2006). "Doping control for metandienone using hair analyzed by gas chromatography-tandem mass spectrometry". Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences. 836 (1–2): 124–8. doi: 10.1016/j.jchromb.2006.03.040. PMID 16597518.
- ^ Ueki M, Ikekita A, Takao Y (2000). "[Nandrolone metabolite in urine of Nagano Olympic athlete]". Jap. J. For. Tox. (in Japanese). 18: 198–199.
- ^ Catlin DH, Leder BZ, Ahrens B, Starcevic B, Hatton CK, Green GA, Finkelstein JS (2000). "Trace contamination of over-the-counter androstenedione and positive urine test results for a nandrolone metabolite". JAMA. 284 (20): 2618–21. doi: 10.1001/jama.284.20.2618. PMID 11086369.
- ^ Baselt RC (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1078–1080. ISBN 978-0-9626523-7-0. Archived from the original on 2020-12-04. Retrieved 2010-08-19.
- ^ Baron P (19 September 2000). "The Drugs Issue: Baumann to fight all the way". The Telegraph. Retrieved 2010-11-13.
- ^ Richardson M (2004-02-19). "Athletics: Mark Richardson explains how he felt at his disciplinary hearing". The Guardian. Retrieved 2010-11-13.
- ^ "Track Star Marion Jones to Admit Steroid Use". National Public Radio. 2007-10-05. Retrieved 2009-11-09.
- ^ UK Sport Anti-doping Directorate (January 2000). Nandrolone Review (PDF) (Report). UK Sport. Archived from the original (PDF) on 2005-04-04. Retrieved 2013-02-02.
- ^ Buiarelli F, Giannetti L, Jasionowska R, Cruciani C, Neri B (July 2010). "Determination of nandrolone metabolites in human urine: comparison between liquid chromatography/tandem mass spectrometry and gas chromatography/mass spectrometry". Rapid Communications in Mass Spectrometry. 24 (13): 1881–1894. Bibcode: 2010RCMS...24.1881B. doi: 10.1002/rcm.4583. PMID 20533318.
- ^ Birch AJ (1950). "80. Hydroaromatic steroid hormones. Part I. 10-Nortestosterone". Journal of the Chemical Society (Resumed): 367. doi: 10.1039/jr9500000367. ISSN 0368-1769.
- ^ Consolidated List of Products Whose Consumption And/or Sale Have Been Banned, Withdrawn, Severely Restricted Or Not Approved by Governments. United Nations Publications. 1983. pp. 154–. ISBN 978-92-1-130230-1.[ permanent dead link]
- ^ a b "Nandrolone".
Further reading
- Geusens P (September 1995). "Nandrolone decanoate: pharmacological properties and therapeutic use in osteoporosis". Clinical Rheumatology. 14 (Suppl 3): 32–9. doi: 10.1007/bf02210686. PMID 8846659. S2CID 24365057.
- Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. doi: 10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- Hemmersbach P, Grosse J (2010). "Nandrolone: A Multi-Faceted Doping Agent". Doping in Sports. Handbook of Experimental Pharmacology. Vol. 195. pp. 127–54. doi: 10.1007/978-3-540-79088-4_6. ISBN 978-3-540-79087-7. PMID 20020363.
- Velema MS, Kwa BH, de Ronde W (March 2012). "Should androgenic anabolic steroids be considered in the treatment regime of selected chronic obstructive pulmonary disease patients?". Current Opinion in Pulmonary Medicine. 18 (2): 118–24. doi: 10.1097/MCP.0b013e32834e9001. PMID 22189453. S2CID 6155275.
- Busardò FP, Frati P, Sanzo MD, Napoletano S, Pinchi E, Zaami S, Fineschi V (January 2015). "The impact of nandrolone decanoate on the central nervous system". Current Neuropharmacology. 13 (1): 122–31. doi: 10.2174/1570159X13666141210225822. PMC 4462037. PMID 26074747.
- Wu C, Kovac JR (October 2016). "Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health". Current Urology Reports. 17 (10): 72. doi: 10.1007/s11934-016-0629-8. PMID 27535042. S2CID 43199715.
- Pan MM, Kovac JR (April 2016). "Beyond testosterone cypionate: evidence behind the use of nandrolone in male health and wellness". Translational Andrology and Urology. 5 (2): 213–9. doi: 10.21037/tau.2016.03.03. PMC 4837307. PMID 27141449.
![Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men.[38]](https://upload.wikimedia.org/wikipedia/commons/thumb/0/03/Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png/298px-Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png)
![Nandrolone levels after a single 100 mg intramuscular injection of nandrolone decanoate or nandrolone phenylpropionate in 4 mL or 1 mL arachis oil solution into gluteal or deltoid muscle in men.[39]](https://upload.wikimedia.org/wikipedia/commons/thumb/d/da/Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png/300px-Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png)
![Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men.[40]](https://upload.wikimedia.org/wikipedia/commons/thumb/e/e8/Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png/300px-Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png)
![Dose-normalized nandrolone exposure (serum level divided by dose administered) with nandrolone decanoate in oil solution by intramuscular or subcutaneous injection in men.[41][42]](https://upload.wikimedia.org/wikipedia/commons/thumb/5/50/Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png/300px-Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png)