 | |
| Clinical data | |
|---|---|
| Other names | MENT; MENTR; RU-27333; 7α-Methylnandrolone; 7α-Methyl-19-nortestosterone; 7α-Methylestr-4-en-17β-ol-3-one |
|
Routes of administration | Subcutaneous implant, intramuscular injection (as trestolone acetate) |
| Drug class | Androgen; Anabolic steroid; Progestogen; Antigonadotropin |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.184.887 |
| Chemical and physical data | |
| Formula | C19H28O2 |
| Molar mass | 288.431 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Trestolone, also known as 7α-methyl-19-nortestosterone (MENT), is an experimental androgen/ anabolic steroid (AAS) and progestogen medication which has been under development for potential use as a form of hormonal birth control for men and in androgen replacement therapy for low testosterone levels in men but has never been marketed for medical use. [1] [2] [3] [4] [5] It is given as an implant that is placed into fat. [3] As trestolone acetate, an androgen ester and prodrug of trestolone, the medication can also be given by injection into muscle. [1] [5]
Side effects of trestolone use in men include low estrogen levels and associated symptoms such as reduced sexual function and decreased bone mineral density among others. [5] [3] [6] Trestolone is an AAS, and hence is an agonist of the androgen receptor, the biological target of androgens like testosterone. [3] [7] It is also a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone. [3] [7] Due to its androgenic and progestogenic activity, trestolone has antigonadotropic effects. [3] [7] These effects result in reversible suppression of sperm production and are responsible for the contraceptive effects of trestolone in men. [3]
Trestolone was first described in 1963. [8] Subsequently, it was not studied again until 1990. [9] Development of trestolone for potential clinical use started by 1993 and continued thereafter. [4] [10] No additional development appears to have been conducted since 2013. [3] The medication was developed by the Population Council, a non-profit, non-governmental organization dedicated to reproductive health. [3] [11]
Medical uses
Trestolone is an experimental medication and is not currently approved for medical use. [2] [3] It has been under development for potential use as a male hormonal contraceptive and in androgen replacement therapy for low testosterone levels. [2] [3] [4] [10] [5] The medication has been studied and developed for use as a subcutaneous implant. [3] An androgen ester and prodrug of trestolone, trestolone acetate, has also been developed, for use via intramuscular injection. [1] [5]
Side effects
Trestolone may cause sexual dysfunction (e.g., decreased sex drive, reduced erectile function) and decreased bone mineral density due to estrogen deficiency. [5] [3] [6]
Pharmacology
Pharmacodynamics
As an AAS, trestolone is an agonist of the androgen receptor (AR), similarly to androgens like testosterone and dihydrotestosterone (DHT). [4] [3] Trestolone is not a substrate for 5α-reductase and hence is not potentiated or inactivated in so-called "androgenic" tissues like the skin, hair follicles, and prostate gland. [12] As such, it has a high ratio of anabolic to androgenic activity, similarly to other nandrolone derivatives. [4] [3] Trestolone is a substrate for aromatase and hence produces the estrogen 7α-methylestradiol as a metabolite. [7] [13] However, trestolone has only weak estrogenic activity and an amount that would appear to be insufficient for replacement purposes, as evidenced by decreased bone mineral density in men treated with it for hypogonadism. [5] [3] Trestolone also has potent progestogenic activity. [7] [3] Both the androgenic and progestogenic activity of trestolone are thought to be involved in its antigonadotropic activity. [7] [3]
| Compound | PR | AR | ER | GR | MR | SHBG | CBG | |
|---|---|---|---|---|---|---|---|---|
| Nandrolone | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1 | ? | |
| Trestolone | 50–75 | 100–125 | ? | <1 | ? | ? | ? | |
| 7α-Methylestradiol | 1–3 | 15–25 | 101 | <1 | <1 | ? | ? | |
| Values are percentages (%). Reference ligands (100%) were progesterone for the PR, testosterone for the AR, E2 for the ER, DEXA for the GR, aldosterone for the MR, DHT for SHBG, and cortisol for CBG. | ||||||||
Mechanism of action
Spermatozoa are produced in the testes of males in a process called spermatogenesis. In order to render a man infertile, a hormone-based male contraceptive method must stop spermatogenesis by interrupting the release of gonadotropins from the pituitary gland. Even in low concentrations, trestolone is a potent inhibitor of the release of the gonadotropins, luteinizing hormone (LH) and follicle stimulating hormone (FSH). [4] [3] In order for spermatogenesis to occur in the testes, both FSH and testosterone must be present. By inhibiting release of FSH, trestolone creates an endocrine environment in which conditions for spermatogenesis are not ideal. [4] [3] Manufacture of sperm is further impaired by the suppression of LH, which in turn drastically curtails the production of testosterone. [4] [3] Sufficient regular doses of trestolone cause severe oligozoospermia or azoospermia, and therefore infertility, in most men. [4] [3] Trestolone-induced infertility has been found to be quickly reversible upon discontinuation. [4] [3]
When LH release is inhibited, the amount of testosterone made in the testes declines dramatically. [4] [3] As a result of trestolone's gonadotropin-suppressing qualities, levels of serum testosterone fall sharply in men treated with sufficient amounts of the medication. [4] [3] Testosterone is the main hormone responsible for maintenance of male secondary sex characteristics. Normally, an inadequate testosterone level causes undesirable effects such as fatigue, loss of skeletal muscle mass, reduced libido, and weight gain. However, the androgenic and anabolic properties of trestolone largely ameliorate this problem [4] [3] — essentially, trestolone replaces testosterone's role as the primary male hormone in the body. [4] [3]
Pharmacokinetics
The pharmacokinetic properties of trestolone, such as poor oral bioavailability and short elimination half-life, make it unsuitable for oral administration or long-term intramuscular injection. [19] [20] As such, trestolone must be administered parenterally via a different and more practical route such as subcutaneous implant, transdermal patch, or topical gel. [19] Trestolone acetate, a prodrug of trestolone, can be administered via intramuscular injection. [5]
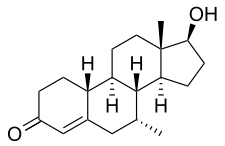
Chemistry
Trestolone, also known as 7α-methyl-19-nortestosterone (MENT) or as 7α-methylestr-4-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of nandrolone (19-nortestosterone). [1] It is a modification of nandrolone with a methyl group at the C7α position. [1] Closely related AAS include 7α-methyl-19-norandrostenedione (MENT dione, trestione) (an androgen prohormone of trestolone) and dimethandrolone (7α,11β-dimethyl-19-nortestosterone) (the C11β methylated derivative of trestolone), as well as mibolerone (7α,17α-dimethyl-19-nortestosterone) and dimethyltrienolone (7α,17α-dimethyl-δ9,11-19-nortestosterone). [1] The progestin tibolone (7α-methyl-17α-ethynyl-δ5(10)-19-nortestosterone) is also closely related to trestolone. [1]
History
Trestolone was first described in 1963. [8] However, it was not subsequently studied again until 1990. [9] [21] Development of trestolone for potential use in male hormonal contraception and androgen replacement therapy was started by 1993, and continued thereafter. [4] [10] [3] No additional development appears to have been conducted since 2013. [3] Trestolone was developed by the Population Council, a non-profit, non-governmental organization dedicated to reproductive health.. [3] [11]
Society and culture
Generic names
Trestolone is the generic name of the drug and its INN. [1] It is also commonly known as 7α-methyl-19-nortestosterone (MENT). [1] [2] [3]
References
- ^ a b c d e f g h i J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 888–. ISBN 978-1-4757-2085-3.
- ^ a b c d "7-alpha-methyl-19-nortestosterone - AdisInsight".
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae Nieschlag E, Kumar N, Sitruk-Ware R (2013). "7α-methyl-19-nortestosterone (MENTR): the population council's contribution to research on male contraception and treatment of hypogonadism". Contraception. 87 (3): 288–95. doi: 10.1016/j.contraception.2012.08.036. PMID 23063338.
- ^ a b c d e f g h i j k l m n o Sundaram K, Kumar N, Bardin CW (April 1993). "7 alpha-methyl-nortestosterone (MENT): the optimal androgen for male contraception". Ann. Med. 25 (2): 199–205. doi: 10.3109/07853899309164168. PMID 8489761.
- ^ a b c d e f g h Corona G, Rastrelli G, Vignozzi L, Maggi M (2012). "Emerging medication for the treatment of male hypogonadism". Expert Opin Emerg Drugs. 17 (2): 239–59. doi: 10.1517/14728214.2012.683411. PMID 22612692. S2CID 22068249.
- ^ a b Anderson RA, Wallace AM, Sattar N, Kumar N, Sundaram K (June 2003). "Evidence for tissue selectivity of the synthetic androgen 7 alpha-methyl-19-nortestosterone in hypogonadal men". J. Clin. Endocrinol. Metab. 88 (6): 2784–93. doi: 10.1210/jc.2002-021960. PMID 12788888.
- ^ a b c d e f García-Becerra R, Ordaz-Rosado D, Noé G, Chávez B, Cooney AJ, Larrea F (2012). "Comparison of 7α-methyl-19-nortestosterone effectiveness alone or combined with progestins on androgen receptor mediated-transactivation". Reproduction. 143 (2): 211–9. doi: 10.1530/REP-11-0171. PMID 22065861.
- ^ a b Lyster SC, Duncan GW (July 1963). "Anabolic, androgenic and myotropic activities of derivatives of 7alpha-methyl-19-nortestosterone". Acta Endocrinol. 43 (3): 399–411. doi: 10.1530/acta.0.0430399. PMID 13931986.
- ^ a b Ma JB, Li ZS (1990). "[Synthesis of 4-substituted 17 beta-hydroxy-7 alpha-methyl-4-estren-3-one and their 17-acetates as antifertility compounds]". Yao Xue Xue Bao (in Chinese). 25 (1): 18–23. PMID 2363352.
- ^ a b c Sundaram K, Kumar N, Bardin CW (1994). "7 alpha-Methyl-19-nortestosterone: an ideal androgen for replacement therapy". Recent Prog. Horm. Res. 49: 373–6. doi: 10.1016/b978-0-12-571149-4.50027-1. ISBN 9780125711494. PMID 8146434.
- ^ a b MENT – project information from the Population Council
- ^ Attardi BJ, Hild SA, Koduri S, Pham T, Pessaint L, Engbring J, Till B, Gropp D, Semon A, Reel JR (October 2010). "The potent synthetic androgens, dimethandrolone (7α,11β-dimethyl-19-nortestosterone) and 11β-methyl-19-nortestosterone, do not require 5α-reduction to exert their maximal androgenic effects". The Journal of Steroid Biochemistry and Molecular Biology. 122 (4): 212–8. doi: 10.1016/j.jsbmb.2010.06.009. PMC 2949447. PMID 20599615.
- ^ Attardi BJ, Pham TC, Radler LC, Burgenson J, Hild SA, Reel JR (June 2008). "Dimethandrolone (7,11β-dimethyl-19-nortestosterone) and 11β-methyl-19-nortestosterone are not converted to aromatic A-ring products in the presence of recombinant human aromatase". The Journal of Steroid Biochemistry and Molecular Biology. 110 (3–5): 214–22. doi: 10.1016/j.jsbmb.2007.11.009. PMC 2575079. PMID 18555683.
- ^ Ojasoo T, Delettré J, Mornon JP, Turpin-VanDycke C, Raynaud JP (1987). "Towards the mapping of the progesterone and androgen receptors". J. Steroid Biochem. 27 (1–3): 255–69. doi: 10.1016/0022-4731(87)90317-7. PMID 3695484.
- ^ Delettré J, Mornon JP, Lepicard G, Ojasoo T, Raynaud JP (January 1980). "Steroid flexibility and receptor specificity". J. Steroid Biochem. 13 (1): 45–59. doi: 10.1016/0022-4731(80)90112-0. PMID 7382482.
- ^ Raynaud JP, Bouton MM, Moguilewsky M, Ojasoo T, Philibert D, Beck G, Labrie F, Mornon JP (January 1980). "Steroid hormone receptors and pharmacology". J. Steroid Biochem. 12: 143–57. doi: 10.1016/0022-4731(80)90264-2. PMID 7421203.
- ^ Ojasoo T, Raynaud JP, Doé JC (January 1994). "Affiliations among steroid receptors as revealed by multivariate analysis of steroid binding data". J. Steroid Biochem. Mol. Biol. 48 (1): 31–46. doi: 10.1016/0960-0760(94)90248-8. PMID 8136304. S2CID 21336380.
- ^ Raynaud J, Ojasoo T, Bouton M, Philibert D (1979). "Receptor Binding as a Tool in the Development of New Bioactive Steroids". Drug Design: Medicinal Chemistry: A Series of Monographs. Vol. 8. New York, Academic Press. pp. 169–214. doi: 10.1016/B978-0-12-060308-4.50010-X. ISBN 9780120603084.
- ^
a
b Attardi BJ, Hild SA, Reel JR (June 2006).
"Dimethandrolone undecanoate: a new potent orally active androgen with progestational activity". Endocrinology. 147 (6): 3016–26.
doi:
10.1210/en.2005-1524.
PMID
16497801.
The pharmacokinetic properties of MENT make it unsuitable for once-daily oral treatment or long-term injection; thus, administration by sc implant or by patch or gel is required (27). MENT showed a more rapid metabolic clearance rate than T in men and monkeys, probably due in part to its failure to bind SHBG (28). In monkeys, MENT acetate in subdermal implants was 10 times as potent as T in suppression of gonadotropin secretion and anabolic effects, but was only twice as potent in stimulating prostate growth (29).
- ^ Suvisaari J, Sundaram K, Noé G, Kumar N, Aguillaume C, Tsong YY, Lähteenmäki P, Bardin CW (May 1997). "Pharmacokinetics and pharmacodynamics of 7alpha-methyl-19-nortestosterone after intramuscular administration in healthy men". Hum. Reprod. 12 (5): 967–73. doi: 10.1093/humrep/12.5.967. PMID 9194649.
- ^ Kumar N, Didolkar AK, Ladd A, Thau R, Monder C, Bardin CW, Sundaram K (November 1990). "Radioimmunoassay of 7 alpha-methyl-19-nortestosterone and investigation of its pharmacokinetics in animals". J. Steroid Biochem. Mol. Biol. 37 (4): 587–91. doi: 10.1016/0960-0760(90)90405-a. PMID 2278844. S2CID 37597215.