 | |
| Clinical data | |
|---|---|
| Trade names | Vestalin (with EE) |
| Other names | Norvinodrel; Vinylestrenolone; Vinilestrenolone; Vinylnoretynodrel; 17α-Vinylestr-5(10)-en-17-ol-3-one; 17α-Vinyl-δ5(10)-19-nortestosterone |
|
Routes of administration | By mouth |
| Drug class | Progestogen; Progestin |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
| Formula | C20H28O2 |
| Molar mass | 300.442 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Norgesterone, also known as norvinodrel or vinylestrenolone and sold under the brand name Vestalin, is a progestin medication which was formerly used in birth control pills for women but is now no longer marketed. [1] [2] [3] [4] It was used in combination with the estrogen ethinylestradiol. [2] [3] [4] It is taken by mouth. [5] [6]
Norgesterone is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone. [7] It has no androgenic activity. [7]
Norgesterone was first described in 1962. [8] [9] It is no longer available. [10]
Medical uses
Norgesterone was used in combination with ethinylestradiol in birth control pills to prevent pregnancy. [2] It is no longer available. [10]
Pharmacology
Pharmacodynamics
Norgesterone is a progestogen, and hence is an agonist of the progesterone receptor. [7] Unlike related progestins, it is virtually devoid of androgenic activity in animal assays. [7]
Chemistry
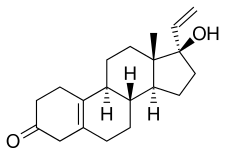
Norgesterone, also known as 17α-vinyl-δ5(10)-19-nortestosterone or as 17α-vinylestr-5(10)-en-17β-ol-3-one, is a synthetic estrane steroid and a derivative of testosterone and 19-nortestosterone. [1] Analogues of norgesterone include norvinisterone (17α-vinyl-19-nortestosterone) and vinyltestosterone (17α-vinyltestosterone). [1]
History
Norgesterone was first described in 1962. [8] [9]
Society and culture
Generic names
Norgesterone is the generic name of the drug and its INN. [1] It has also been referred to as norvinodrel, vinylestrenolone, and vinylnoretynodrel. [1] [11]
Brand names
Norgesterone was marketed in combination with ethinylestradiol, an estrogen, as a birth control pill under the brand name Vestalin. [2] [3] [4]
Availability
Norgesterone is no longer marketed and hence is no longer available in any country. [10]
References
- ^ a b c d e Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 887–. ISBN 978-1-4757-2085-3.
- ^ a b c d Wassef SA, Sami G, Hamid EA (June 1970). "Effect of switching with oral contraceptives". The Egyptian Population and Family Planning Review. 3 (1): 77–93. PMID 12254511.
- ^ a b c Bengtsson LP, Tausk M (September 1972). Pharmacology of the endocrine system and related drugs: progesterone, progestational drugs and antifertility agents. Pergamon Press. ISBN 9780080157450.
- ^ a b c Challener CA (1 December 2001). Chiral Drugs. Wiley. ISBN 978-0-566-08411-9.
- ^ Boris Rubio L (November 1966). "[Vinylestrenolone: a new progestational hormone. Results of its cyclic administration]". Minerva Ginecologica (in Italian). 18 (21): 1215–1217. PMID 5997085.
- ^ Samaja BA, Prandini B (March 1974). "The influence of estrogenic and-or progestogenic treatment on some parameters of lipid metabolism (a controlled clinical study)". Endokrinologie. 63 (1): 76–84. PMID 4140086. Archived from the original on 2018-02-28.
- ^ a b c d de Ruggieri P, Matscher R, Lupo C, Spazzoli G (1965). "Biological properties of 17α-vinyl-5(10)-estrene-17β-ol-3-one (norvinodrel) as a progestational and claudogenic compound". Steroids. 5 (1): 73–91. doi: 10.1016/0039-128X(65)90133-9. ISSN 0039-128X.
- ^ a b "Steroid hormone compositions and method of using same".
- ^ a b D'Incerti Bonini L, Pagani C (April 1962). "[Clinical investigations of the progestational activity of vinylestrenolone]". Annali di Ostetricia e Ginecologia (in Italian). 84: 279–285. PMID 13883015.
- ^ a b c http://www.micromedexsolutions.com/micromedex2/[ permanent dead link]
- ^ Greydanus DE (6 December 2012). "Contraception". In Lavery JP, Sanfilippo JS (eds.). Pediatric and Adolescent Obstetrics and Gynecology. Springer Science & Business Media. pp. 236–. ISBN 978-1-4612-5064-7.