 | |
| Clinical data | |
|---|---|
| Other names | 11β-MNTDC; CDB-4754 |
|
Routes of administration | By mouth, intramuscular injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester; Progestogen |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C32H52O4 |
| Molar mass | 500.764 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
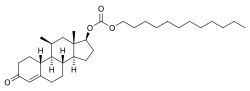
11β-Methyl-19-nortestosterone 17β-dodecylcarbonate (11β-MNTDC) (developmental code name CDB-4754) is a synthetic and orally active anabolic–androgenic steroid (AAS) and a derivative of nandrolone (19-nortestosterone) which was developed by the Contraceptive Development Branch (CDB) of the National Institute of Child Health and Human Development (NICHD) and has not been marketed for medical use at this time. [1] [2] [3] [4] It is an androgen ester – specifically, the C17β dodecyl carbonate ester of 11β-methyl-19-nortestosterone (11β-MNT) – and acts as a prodrug of 11β-MNT in the body. [1] [2] [3] [4]
11β-MNTDC is under development as a birth control pill for men. [5] [6] [7]\ It has been described as the "sister compound" of dimethandrolone undecanoate, which is also under development as a birth control pill for men. [7] [5]
Because 11β-MNTDC is not 5α-reduced, in contrast to testosterone, it may have less risk of scalp hair loss. [8]
See also
- List of androgen esters § Esters of other synthetic AAS
- List of investigational sex-hormonal agents § Androgenics
References
- ^ a b Attardi BJ, Marck BT, Matsumoto AM, Koduri S, Hild SA (2011). "Long-term effects of dimethandrolone 17β-undecanoate and 11β-methyl-19-nortestosterone 17β-dodecylcarbonate on body composition, bone mineral density, serum gonadotropins, and androgenic/anabolic activity in castrated male rats". Journal of Andrology. 32 (2): 183–192. doi: 10.2164/jandrol.110.010371. PMID 20798389.
- ^ a b Attardi BJ, Hild SA, Koduri S, Pham T, Pessaint L, Engbring J, et al. (October 2010). "The potent synthetic androgens, dimethandrolone (7α,11β-dimethyl-19-nortestosterone) and 11β-methyl-19-nortestosterone, do not require 5α-reduction to exert their maximal androgenic effects". The Journal of Steroid Biochemistry and Molecular Biology. 122 (4): 212–218. doi: 10.1016/j.jsbmb.2010.06.009. PMC 2949447. PMID 20599615.
- ^ a b Hild SA, Attardi BJ, Koduri S, Till BA, Reel JR (2010). "Effects of synthetic androgens on liver function using the rabbit as a model". Journal of Andrology. 31 (5): 472–481. doi: 10.2164/jandrol.109.009365. PMC 2943539. PMID 20378929.
- ^ a b US 7820642, Blye RP, Kim HK, "Nandrolone 17β-carbonates", issued 26 October 2010, assigned to U.S. Department of Health and Human Services
- ^ a b Thirumalai A, Page ST (January 2019). "Recent Developments in Male Contraception". Drugs. 79 (1): 11–20. doi: 10.1007/s40265-018-1038-8. PMID 30588563. S2CID 56895132.
- ^ "Second potential male birth control pill passes human safety tests".
- ^ a b Yuen F, Thirumalai A, Pham C, Swerdloff RS, Anawalt BD, Liu PY, Amory JK, Bremner WJ, Dart C, Wu H, Hull L, Blithe DL, Long J, Wang C, Page ST (March 2020). "Daily Oral Administration of the Novel Androgen 11β-MNTDC Markedly Suppresses Serum Gonadotropins in Healthy Men". The Journal of Clinical Endocrinology and Metabolism. 105 (3): e835–e847. doi: 10.1210/clinem/dgaa032. PMC 7049261. PMID 31976519.
- ^ Shapiro LJ, Shapiro DB (2018). Low Anabolic Profile in Assessing a Patient's Overall Hair Loss. pp. 687–698. doi: 10.1007/978-4-431-56547-5_72. ISBN 978-4-431-56545-1.