| |
 | |
| Clinical data | |
|---|---|
| Trade names | Erleada, others |
| Other names | ARN-509; JNJ-56021927; JNJ-927; A52 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a618018 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth [2] |
| Drug class | Nonsteroidal antiandrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 100% [2] |
| Protein binding | Apalutamide: 96%
[2] NDMA: 95% [2] |
| Metabolism | Liver ( CYP2C8, CYP3A4) [2] |
| Metabolites | • NDMA [2] |
| Elimination half-life | Apalutamide: 3–4 days (at steady-state) [7] [2] |
| Excretion |
Urine: 65%
[2] Feces: 24% [2] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.235.115 |
| Chemical and physical data | |
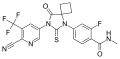
| Formula | C21H15F4N5O2S |
| Molar mass | 477.44 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Apalutamide, sold under the brand name Erleada among others, is a nonsteroidal antiandrogen (NSAA) medication which is used in the treatment of prostate cancer. [2] [8] [9] [10] [11] It is specifically indicated for use in conjunction with castration in the treatment of non- metastatic castration-resistant prostate cancer (NM-CRPC). [2] [12] [13] It is taken by mouth. [2] [8]
Side effects of apalutamide when added to castration include fatigue, nausea, abdominal pain, diarrhea, high blood pressure, rash, falls, bone fractures, and an underactive thyroid. [2] [14] [15] [8] [10] Rarely, it can cause seizures. [2] [8] The medication has a high potential for drug interactions. [2] [8] Apalutamide is an antiandrogen, and acts as an antagonist of the androgen receptor, the biological target of androgens like testosterone and dihydrotestosterone. [2] [8] [11] In doing so, it prevents the effects of these hormones in the prostate gland and elsewhere in the body. [2] [8] [11]
Apalutamide was first described in 2007, and was approved for the treatment of prostate cancer in February 2018. [12] [13] [8] [16] It was the first medication to be approved specifically for the treatment of NM-CRPC. [2] [8] [13]
Medical uses
Apalutamide is used in conjunction with castration, either via bilateral orchiectomy or gonadotropin-releasing hormone analogue (GnRH analogue) therapy, as a method of androgen deprivation therapy in the treatment of NM-CRPC. [2] [17] [18] [19] It is also a promising potential treatment for metastatic castration-resistant prostate cancer (mCRPC), which the NSAA enzalutamide and the androgen synthesis inhibitor abiraterone acetate are used to treat. [10]
Available forms
Apalutamide is provided in the form of 60 mg oral tablets. [2] It is taken at a dosage of 240 mg once per day (four tablets) when used in the treatment of NM-CRPC. [2]
Contraindications
Contraindications of apalutamide include pregnancy and a history of or susceptibility to seizures. [2]
Side effects
Apalutamide has been found to be well tolerated in clinical trials, [20] [17] with the most common side effects reported when added to surgical or medical castration including fatigue, nausea, abdominal pain, and diarrhea. [14] [15] [21] Other side effects have included rash, falls and bone fractures, and hypothyroidism, as well as seizures (in 0.2%), among others. [2] [8] [13] Apalutamide is an expected teratogen and has a theoretical risk of birth defects in male infants if taken by women during pregnancy. [2] It may impair male fertility. [2] When used as a monotherapy (i.e., without surgical or medical castration) in men, NSAAs are known to produce additional, estrogenic side effects like breast tenderness, gynecomastia, and feminization in general by increasing estradiol levels. [22] Similarly to the related second-generation NSAA enzalutamide but unlike first-generation NSAAs like flutamide and bicalutamide, elevated liver enzymes and hepatotoxicity have not been reported with apalutamide. [2] Case reports of rare interstitial lung disease with apalutamide exist similarly to with first-generation NSAAs however. [23] [24] [25]
Overdose
There is no known antidote for overdose of apalutamide. [2] General supportive measures should be undertaken until clinical toxicity, if any, diminishes or resolves. [2]
Interactions
Apalutamide has a high potential for drug interactions. [2] In terms of effects of apalutamide on other drugs, the exposure of substrates of CYP3A4, CYP2C19, CYP2C9, UDP-glucuronosyltransferase, P-glycoprotein, ABCG2, or OATP1B1 may be reduced to varying extents. [2] In terms of effects of other drugs on apalutamide, strong CYP2C8 or CYP3A4 inhibitors may increase levels of apalutamide or its major active metabolite N-desmethylapalutamide, while mild to moderate CYP2C8 or CYP3A4 inhibitors are not expected to affect their exposure. [2]
Pharmacology
Pharmacodynamics
Antiandrogenic activity
Apalutamide acts as a selective competitive silent antagonist of the androgen receptor (AR), via the ligand-binding domain, and hence is an antiandrogen. [8] [11] [14] [17] It is similar both structurally and pharmacologically to the second-generation NSAA enzalutamide, [20] [26] but shows some advantages, including higher antiandrogenic activity as well as several-fold reduced central nervous system distribution. [11] [14] [17] The latter difference may reduce its comparative risk of seizures and other central side effects. [11] [14] [17] Apalutamide has 5- to 10-fold greater affinity for the AR than bicalutamide, a first-generation NSAA. [19] [18]
The acquired F876L mutation of the AR identified in advanced prostate cancer cells has been found to confer resistance to both enzalutamide and apalutamide. [27] [28] A newer NSAA, darolutamide, is not affected by this mutation, nor has it been found to be affected by any other tested/well-known AR mutations. [29] Apalutamide may be effective in a subset of prostate cancer patients with acquired resistance to abiraterone acetate. [20]
Other activities
Apalutamide shows potent induction potential of cytochrome P450 enzymes similarly to enzalutamide. [2] [30] [31] It is a strong inducer of CYP3A4 and CYP2C19 and a weak inducer of CYP2C9, as well as an inducer of UDP-glucuronosyltransferase. [2] In addition, apalutamide is an inducer of P-glycoprotein, ABCG2, and OATP1B1. [2]
Apalutamide binds weakly to and inhibits the GABAA receptor in vitro similarly to enzalutamide ( IC50 = 3.0 and 2.7 μM, respectively), [32] but due to its relatively lower central concentrations, may have a lower risk of seizures in comparison. [11] [14] [21]
Apalutamide has been found to significantly and concentration-dependently increase QT interval. [2]
Pharmacokinetics
The mean absolute oral bioavailability of apalutamide is 100%. [2] Mean peak levels of apalutamide occur 2 hours following administration, with a range of 1 to 5 hours. [2] Food delays the median time to peak levels of apalutamide by approximately 2 hours, with no significant changes in the peak levels themselves or in area-under-curve levels. [2] Steady-state levels of apalutamide are achieved following 4 weeks of administration, with an approximate 5-fold accumulation. [2] Peak concentrations for 160 mg/day apalutamide at steady-state are 6.0 μg/mL (12.5 μmol/L), [2] relative to peak levels of 16.6 μg/mL (35.7 μmol/L) for 160 mg/day enzalutamide and mean (R)-bicalutamide levels of 21.6 μg/mL (50.2 μmol/L) for 150 mg/day bicalutamide. [33] [34] The mean volume of distribution of apalutamide at steady-state is approximately 276 L. [2] The plasma protein binding of apalutamide is 96%, while that of its major metabolite N-desmethylapalutamide is 95%, both irrespective of concentration. [2]
Apalutamide is metabolized in the liver by CYP2C8 and CYP3A4. [2] A major active metabolite, N-desmethylapalutamide, is formed by these enzymes, with similar contribution of each of these enzymes to its formation at steady-state. [2] Following a single oral dose of 200 mg apalutamide, apalutamide represented 45% and N-desmethylapalutamide 44% of total area-under-curve levels. [2] The mean elimination half-life of apalutamide at steady-state is 3 to 4 days. [2] [7] Fluctuations in apalutamide exposure are low and levels are stable throughout the day, with mean peak-to-trough ratios of 1.63 for apalutamide and 1.27–1.3 for N-desmethylapalutamide. [2] After a single dose of apalutamide, its clearance rate (CL/F) was 1.3 L/h, while its clearance rate increased to 2.0 L/h at steady-state. [8] This change is considered to be likely due to CYP3A4 auto-induction. [8] Approximately 65% of apalutamide is excreted in urine (1.2% as unchanged apalutamide and 2.7% as N-desmethylapalutamide) while 24% is excreted in feces (1.5% as unchanged apalutamide and 2% as N-desmethylapalutamide). [2]
Chemistry
Apalutamide is a structural analogue of enzalutamide and RD-162. [19] [35] It is a pyridyl variant of RD-162. Enzalutamide and RD-162 were derived from the nonsteroidal androgen RU-59063, which itself was derived from the first-generation NSAA nilutamide and by extension from flutamide. [36]
-
Apalutamide
History
Apalutamide was originated by the University of California system and was developed primarily by Janssen Research & Development, a division of Johnson & Johnson. [37] It was first described in the literature in a United States patent application that was published in November 2007 and in another that was submitted in July 2010. [16] [38] A March 2012 publication described the discovery and development of apalutamide. [11] A phase I clinical trial of apalutamide was completed by March 2012, and the results of this study were published in 2013. [11] [39] Information on phase III clinical studies, including ATLAS, SPARTAN, and TITAN, was published between 2014 and 2016. [40] [41] [42] Positive results for phase III trials were first described in 2017, and Janssen submitted a New Drug Application for apalutamide to the United States Food and Drug Administration on 11 October 2017. [43] Apalutamide was approved by the Food and Drug Administration in the United States, under the brand name Erleada, for the treatment of NM-CRPC on 14 February 2018. [12] [13] It was subsequently approved in Canada, the European Union, and Australia. [44] [6]
Society and culture
Generic names
Apalutamide is the generic name of the drug and its INN. [45] [44] It is also known by its developmental code names ARN-509 and JNJ-56021927. [37] [8]
Brand names
Apalutamide is marketed under the brand names Erleada and Erlyand. [2] [12] [13] [44]
Availability
Apalutamide is available in the United States, Canada, the European Union, and Australia. [2] [12] [13] [44] [6]
References
- ^ "Apalutamide (Erleada) Use During Pregnancy". Drugs.com. 20 July 2020. Retrieved 28 September 2020.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai aj ak al am an ao ap aq ar as at au av aw ax ay "Erleada- apalutamide tablet, film coated". DailyMed. 27 October 2020. Retrieved 8 November 2020.
- ^ "PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION" (PDF). hres.ca. Janssen Inc. 6 July 2021. Archived (PDF) from the original on 3 November 2022. Retrieved 3 November 2022.
- ^ "Regulatory Decision Summary for Erleada". Drug and Health Products Portal. 2 August 2023. Retrieved 2 April 2024.
- ^ "Drug and medical device highlights 2018: Helping you maintain and improve your health". Health Canada. 14 October 2020. Retrieved 17 April 2024.
- ^ a b c "Erleada EPAR". European Medicines Agency (EMA) (in Aragonese). 13 November 2018. Retrieved 9 November 2020.
- ^ a b Rathkopf DE, Morris MJ, Fox JJ, Danila DC, Slovin SF, Hager JH, et al. (October 2013). "Phase I study of ARN-509, a novel antiandrogen, in the treatment of castration-resistant prostate cancer". Journal of Clinical Oncology. 31 (28): 3525–30. doi: 10.1200/JCO.2013.50.1684. PMC 3782148. PMID 24002508.
- ^ a b c d e f g h i j k l m n Al-Salama ZT (April 2018). "Apalutamide: First Global Approval". Drugs. 78 (6): 699–705. doi: 10.1007/s40265-018-0900-z. PMID 29626324. S2CID 4653827.
- ^ Chong JT, Oh WK, Liaw BC (2018). "Profile of apalutamide in the treatment of metastatic castration-resistant prostate cancer: evidence to date". OncoTargets Ther. 11: 2141–2147. doi: 10.2147/OTT.S147168. PMC 5905496. PMID 29695920.
- ^ a b c Dellis AE, Papatsoris AG (June 2018). "Apalutamide: The established and emerging roles in the treatment of advanced prostate cancer". Expert Opin Investig Drugs. 27 (6): 553–559. doi: 10.1080/13543784.2018.1484107. PMID 29856649. S2CID 46925616.
- ^ a b c d e f g h i Clegg NJ, Wongvipat J, Joseph JD, Tran C, Ouk S, Dilhas A, et al. (March 2012). "ARN-509: a novel antiandrogen for prostate cancer treatment". Cancer Research. 72 (6): 1494–503. doi: 10.1158/0008-5472.CAN-11-3948. PMC 3306502. PMID 22266222.
- ^ a b c d e "FDA approves new treatment for a certain type of prostate cancer using novel clinical trial endpoint". Food and Drug Administration. 24 March 2020.
- ^ a b c d e f g "FDA Approves Apalutamide for Nonmetastatic Prostate Cancer".
- ^ a b c d e f Schweizer MT, Antonarakis ES (August 2012). "Abiraterone and other novel androgen-directed strategies for the treatment of prostate cancer: a new era of hormonal therapies is born". Therapeutic Advances in Urology. 4 (4): 167–78. doi: 10.1177/1756287212452196. PMC 3398601. PMID 22852027.
- ^ a b Leibowitz-Amit R, Joshua AM (December 2012). "Targeting the androgen receptor in the management of castration-resistant prostate cancer: rationale, progress, and future directions". Current Oncology. 19 (Suppl 3): S22-31. doi: 10.3747/co.19.1281. PMC 3553559. PMID 23355790.
- ^ a b WO 2007126765, Jung ME, Sawyers CL, Ouk S, Tran C, Wongvipat J, "Androgen receptor modulator for the treatment of prostate cancer and androgen receptor-associated diseases", published 8 November 2007, assigned to The Regents Of The University Of California.
- ^ a b c d e Rathkopf D, Scher HI (2013). "Androgen receptor antagonists in castration-resistant prostate cancer". Cancer Journal. 19 (1): 43–9. doi: 10.1097/PPO.0b013e318282635a. PMC 3788593. PMID 23337756.
- ^ a b Kim W, Ryan CJ (February 2015). "Quo vadis: advanced prostate cancer-clinical care and clinical research in the era of multiple androgen receptor-directed therapies". Cancer. 121 (3): 361–71. doi: 10.1002/cncr.28929. PMID 25236176. S2CID 6309403.
- ^ a b c Kawahara T, Miyamoto H (June 2014). "Androgen Receptor Antagonists in the Treatment of Prostate Cancer". Clinical Immunology, Endocrine & Metabolic Drugs. 1 (1): 11–19. doi: 10.2174/22127070114019990002. ISSN 2212-7070.
- ^ a b c Patel JC, Maughan BL, Agarwal AM, Batten JA, Zhang TY, Agarwal N (2013). "Emerging molecularly targeted therapies in castration refractory prostate cancer". Prostate Cancer. 2013: 981684. doi: 10.1155/2013/981684. PMC 3684034. PMID 23819055.
- ^ a b Pinto Á (February 2014). "Beyond abiraterone: new hormonal therapies for metastatic castration-resistant prostate cancer". Cancer Biology & Therapy. 15 (2): 149–55. doi: 10.4161/cbt.26724. PMC 3928129. PMID 24100689.
- ^ Anderson J (March 2003). "The role of antiandrogen monotherapy in the treatment of prostate cancer". BJU Int. 91 (5): 455–61. doi: 10.1046/j.1464-410x.2003.04026.x. PMID 12603397. S2CID 8639102.
- ^ Kirishima F, Shigematsu Y, Kobayashi K (May 2022). "Interstitial lung disease induced by apalutamide therapy for castration-resistant prostate cancer: A report of a rare case". IJU Case Rep. 5 (3): 153–155. doi: 10.1002/iju5.12420. PMC 9057741. PMID 35509772.
- ^ Kobe H, Tachikawa R, Masuno Y, Matsunashi A, Murata S, Hagimoto H, Tomii K (September 2021). "Apalutamide-induced severe interstitial lung disease: A report of two cases from Japan". Respir Investig. 59 (5): 700–705. doi: 10.1016/j.resinv.2021.05.006. PMID 34144936. S2CID 235481675.
- ^ Wu B, Shen P, Yin X, Yu L, Wu F, Chen C, Li J, Xu T (March 2022). "Analysis of adverse event of interstitial lung disease in men with prostate cancer receiving hormone therapy using the Food and Drug Administration Adverse Event Reporting System". Br J Clin Pharmacol. 89 (2): 440–448. doi: 10.1111/bcp.15336. PMID 35349180. S2CID 247777754.
-
^ Tao YX (11 June 2014).
Pharmacology and Therapeutics of Constitutively Active Receptors. Elsevier Science. pp. 351–.
ISBN
978-0-12-417206-7.
ARN-509 is related structurally to enzalutamide with greater in vivo activity in CRPC xenograft models (Clegg et al., 2012).
- ^ Joseph JD, Lu N, Qian J, Sensintaffar J, Shao G, Brigham D, Moon M, Maneval EC, Chen I, Darimont B, Hager JH (September 2013). "A clinically relevant androgen receptor mutation confers resistance to second-generation antiandrogens enzalutamide and ARN-509". Cancer Discovery. 3 (9): 1020–9. doi: 10.1158/2159-8290.CD-13-0226. PMID 23779130.
- ^ Nelson WG, Yegnasubramanian S (September 2013). "Resistance emerges to second-generation antiandrogens in prostate cancer". Cancer Discovery. 3 (9): 971–4. doi: 10.1158/2159-8290.CD-13-0405. PMC 3800038. PMID 24019330.
- ^ Moilanen AM, Riikonen R, Oksala R, Ravanti L, Aho E, Wohlfahrt G, Nykänen PS, Törmäkangas OP, Palvimo JJ, Kallio PJ (July 2015). "Discovery of ODM-201, a new-generation androgen receptor inhibitor targeting resistance mechanisms to androgen signaling-directed prostate cancer therapies". Scientific Reports. 5: 12007. Bibcode: 2015NatSR...512007M. doi: 10.1038/srep12007. PMC 4490394. PMID 26137992.
- ^ Fizazi K, Albiges L, Loriot Y, Massard C (2015). "ODM-201: a new-generation androgen receptor inhibitor in castration-resistant prostate cancer". Expert Review of Anticancer Therapy. 15 (9): 1007–17. doi: 10.1586/14737140.2015.1081566. PMC 4673554. PMID 26313416.
- ^ Ivachtchenko AV, Mitkin OD, Kudan EV, Rjahovsky AA, Vorobiev AA, Trifelenkov AS, et al. (2014). "Preclinical Development of ONC1-13B, Novel Antiandrogen for Prostate Cancer Treatment". Journal of Cancer. 5 (2): 133–42. doi: 10.7150/jca.7773. PMC 3909768. PMID 24494031.
- ^ Clegg NJ, Wongvipat J, Joseph JD, Tran C, Ouk S, Dilhas A, et al. (March 2012). "ARN-509: a novel antiandrogen for prostate cancer treatment". Cancer Research. 72 (6): 1494–503. doi: 10.1158/0008-5472.CAN-11-3948. PMC 3306502. PMID 22266222.
- ^ "Reference at www.accessdata.fda.gov" (PDF).
- ^ Cockshott ID (2004). "Bicalutamide: clinical pharmacokinetics and metabolism". Clin Pharmacokinet. 43 (13): 855–78. doi: 10.2165/00003088-200443130-00003. PMID 15509184. S2CID 29912565.
- ^ Tran C, Ouk S, Clegg NJ, Chen Y, Watson PA, Arora V, Wongvipat J, Smith-Jones PM, Yoo D, Kwon A, Wasielewska T, Welsbie D, Chen CD, Higano CS, Beer TM, Hung DT, Scher HI, Jung ME, Sawyers CL (2009). "Development of a second-generation antiandrogen for treatment of advanced prostate cancer". Science. 324 (5928): 787–90. Bibcode: 2009Sci...324..787T. doi: 10.1126/science.1168175. PMC 2981508. PMID 19359544.
- ^ Liu B, Su L, Geng J, Liu J, Zhao G (2010). "Developments in nonsteroidal antiandrogens targeting the androgen receptor". ChemMedChem. 5 (10): 1651–61. doi: 10.1002/cmdc.201000259. PMID 20853390. S2CID 23228778.
- ^ a b "Apalutamide - Janssen Research and Development". AdisInsight. Springer Nature Switzerland AG.
- ^ US 20100190991, Ouerfelli O, Dilhas A, Yang G, Zhao H, "Synthesis of thiohydantoins", issued 11 June 2013, assigned to Sloan Kettering Institute for Cancer Research.
- ^ Rathkopf DE, Morris MJ, Fox JJ, Danila DC, Slovin SF, Hager JH, et al. (October 2013). "Phase I study of ARN-509, a novel antiandrogen, in the treatment of castration-resistant prostate cancer". Journal of Clinical Oncology. 31 (28): 3525–3530. doi: 10.1200/JCO.2013.50.1684. PMC 3782148. PMID 24002508.
- ^ Smith MR, Liu G, Shreeve SM, Matheny S, Sosa A, Kheoh TS, et al. A randomized double-blind, comparative study of ARN-509 plus androgen deprivation therapy (ADT) versus ADT alone in nonmetastatic castration-resistant prostate cancer (M0-CRPC): The SPARTAN trial. 2014 ASCO Annual Meeting. doi: 10.1200/jco.2014.32.15_suppl.tps5100.
- ^ Bossi A, Dearnaley D, McKenzie M, Baskin-Bey E, Tyler R, Tombal B, et al. (2016). "ATLAS: A phase 3 trial evaluating the efficacy of apalutamide (ARN-509) in patients with high-risk localized or locally advanced prostate cancer receiving primary radiation therapy". Annals of Oncology. 27 (suppl_6): vi263. doi: 10.1093/annonc/mdw372.52. ISSN 0923-7534.
- ^ Chi K, Chowdhury S, Radziszewski P, Lebret T, Ozguroglu M, Sternberg C, et al. (2016). "TITAN: A randomized, double-blind, placebo-controlled, phase 3 trial of apalutamide (ARN-509) plus androgen deprivation therapy (ADT) in metastatic hormone-sensitive prostate cancer (mHSPC)". Annals of Oncology. 27 (suppl_6): vi265. doi: 10.1093/annonc/mdw372.54. ISSN 0923-7534.
- ^ "Janssen Submits New Drug Application to U.S. FDA for Apalutamide (ARN-509) to Treat Men with Non-Metastatic Castration-Resistant Prostate Cancer". PR Newswire (Press release).
- ^ a b c d "Erleada (Apalutamide): Side Effects, Dosage & Uses". Drugs.com.
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN)" (PDF). WHO Drug Information. 29 (2). 2015. Archived from the original (PDF) on 18 May 2016.
Further reading
- Al-Salama ZT (April 2018). "Apalutamide: First Global Approval". Drugs. 78 (6): 699–705. doi: 10.1007/s40265-018-0900-z. PMID 29626324. S2CID 4653827.
- Chong JT, Oh WK, Liaw BC (2018). "Profile of apalutamide in the treatment of metastatic castration-resistant prostate cancer: evidence to date". OncoTargets Ther. 11: 2141–2147. doi: 10.2147/OTT.S147168. PMC 5905496. PMID 29695920.
- Dellis AE, Papatsoris AG (June 2018). "Apalutamide: The established and emerging roles in the treatment of advanced prostate cancer". Expert Opin Investig Drugs. 27 (6): 553–559. doi: 10.1080/13543784.2018.1484107. PMID 29856649. S2CID 46925616.