| Carcinoembryonic antigen | |
|---|---|
 | |
| Identifiers | |
| Symbol | CEACAM |
| Membranome | 211 |
Carcinoembryonic antigen (CEA) describes a set of highly-related glycoproteins involved in cell adhesion. CEA is normally produced in gastrointestinal tissue during fetal development, but the production stops before birth. Consequently, CEA is usually present at very low levels in the blood of healthy adults (about 2–4 ng/mL). [2] However, the serum levels are raised in some types of cancer, which means that it can be used as a tumor marker in clinical tests. Serum levels can also be elevated in heavy smokers. [3]
CEA are glycosyl phosphatidyl inositol (GPI) cell-surface-anchored glycoproteins whose specialized sialo fucosylated glycoforms serve as functional colon carcinoma L-selectin and E-selectin ligands, which may be critical to the metastatic dissemination of colon carcinoma cells. [4] [5] [6] Immunologically they are characterized as members of the CD66 cluster of differentiation. The proteins include CD66a, CD66b, CD66c, CD66d, CD66e, CD66f.
History
CEA was first identified in 1965 by Phil Gold,a Canadian physician, scientist and professor and Samuel O. Freedman who is also a Canadian professor of immunology in human colon cancer tissue extracts. [7]
Diagnostic significance
The CEA blood test is not reliable for diagnosing cancer or as a screening test for early detection of cancer. [8] Most types of cancer do not result in a high CEA level. [9]
Serum from individuals with colorectal carcinoma often has higher levels of CEA than healthy individuals (above approximately 2.5ng/mL). [10] CEA measurement is mainly used as a tumor marker to monitor colorectal carcinoma treatment, to identify recurrences after surgical resection, for staging or to localize cancer spread through measurement of biological fluids. [11] CEA levels may also be raised in gastric carcinoma, pancreatic carcinoma, lung carcinoma, breast carcinoma, and medullary thyroid carcinoma, as well as some non-neoplastic conditions like ulcerative colitis, pancreatitis, cirrhosis, [12] COPD, Crohn's disease, hypothyroidism [13] as well as in smokers. [14] Elevated CEA levels should return to normal after successful surgical removal of the tumor and can be used in follow up, especially of colorectal cancers. [15]
CEA elevation is known to be affected by multiple factors. It varies inversely with tumor grade; well-differentiated tumors secrete more CEA. CEA is elevated more in tumors with lymph node and distant metastasis than in organ-confined tumors and, thus, varies directly with tumor stage. Left-sided tumors generally tend to have higher CEA levels than right-sided tumors. [16] Tumors causing bowel obstruction produce higher CEA levels. [16] Aneuploid tumors produce more CEA than diploid tumors. [17] Liver dysfunction increases CEA levels as the liver is the primary site of CEA metabolism. [3]
Antibodies
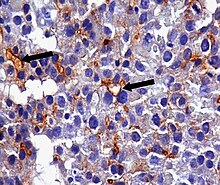
An anti-CEA antibody is an antibody against CEA. Such antibodies to CEA are commonly used in immunohistochemistry to identify cells expressing the glycoprotein in tissue samples. In adults, CEA is primarily expressed in cells of tumors (some malignant, some benign) [19] but they are particularly associated with the adenocarcinomas, such as those arising in the colon, lung, breast, stomach, or pancreas. It can therefore be used to distinguish between these and other similar cancers. For example, it can help to distinguish between adenocarcinoma of the lung and mesothelioma, a different type of lung cancer which is not normally CEA positive. Because even monoclonal antibodies to CEA tend to have some degree of cross-reactivity, occasionally giving false positive results, it is commonly employed in combination with other immunohistochemistry tests, such as those for BerEp4, WT1, and calretinin. [20] For cancers that highly express CEA, targeting CEA through radioimmunotherapy is one of the therapy approaches. [21] Engineered antibodies such as single-chain Fv antibodies (sFvs) or bispecific antibodies have been used for targeting and therapy of CEA expressing tumors both in vitro and in vivo with promising results [22] [23] Regions of high CEA levels in the body can be detected with the monoclonal antibody arcitumomab. [24]
Genetics
CEA and related genes make up the CEA family belonging to the immunoglobulin superfamily.
In humans, the carcinoembryonic antigen family consists of 29 genes, 18 of which are normally expressed. [25] The following is a list of human genes which encode carcinoembryonic antigen-related cell adhesion proteins: CEACAM1, CEACAM3, CEACAM4, CEACAM5, CEACAM6, CEACAM7, CEACAM8, CEACAM16, CEACAM18, CEACAM19, CEACAM20, CEACAM21
See also
References
- ^ Boehm MK, Perkins SJ (June 2000). "Structural models for carcinoembryonic antigen and its complex with the single-chain Fv antibody molecule MFE23". FEBS Letters. 475 (1): 11–6. doi: 10.1016/S0014-5793(00)01612-4. PMID 10854848. S2CID 5765612.
- ^ Gan N, Jia L, Zheng L (2011-10-28). "A sandwich electrochemical immunosensor using magnetic DNA nanoprobes for carcinoembryonic antigen". International Journal of Molecular Sciences. 12 (11): 7410–23. doi: 10.3390/ijms12117410. PMC 3233412. PMID 22174606.
- ^ a b Duffy MJ (April 2001). "Carcinoembryonic antigen as a marker for colorectal cancer: is it clinically useful?". Clinical Chemistry. 47 (4): 624–30. doi: 10.1093/clinchem/47.4.624. PMID 11274010.
- ^ Thomas SN, Zhu F, Schnaar RL, Alves CS, Konstantopoulos K (June 2008). "Carcinoembryonic antigen and CD44 variant isoforms cooperate to mediate colon carcinoma cell adhesion to E- and L-selectin in shear flow". The Journal of Biological Chemistry. 283 (23): 15647–55. doi: 10.1074/jbc.M800543200. PMC 2414264. PMID 18375392.
- ^ Konstantopoulos K, Thomas SN (2009). "Cancer cells in transit: the vascular interactions of tumor cells". Annual Review of Biomedical Engineering. 11: 177–202. doi: 10.1146/annurev-bioeng-061008-124949. PMID 19413512.
- ^ Thomas SN, Tong Z, Stebe KJ, Konstantopoulos K (2009). "Identification, characterization and utilization of tumor cell selectin ligands in the design of colon cancer diagnostics". Biorheology. 46 (3): 207–25. doi: 10.3233/BIR-2009-0534. PMID 19581728.
- ^ Gold P, Freedman SO (March 1965). "Demonstration of tumor-specific antigens in human colonic carcinomata by immunological tolerance and absorption techniques". The Journal of Experimental Medicine. 121 (3): 439–62. doi: 10.1084/jem.121.3.439. PMC 2137957. PMID 14270243.
- ^ Duffy MJ, van Dalen A, Haglund C, Hansson L, Klapdor R, Lamerz R, Nilsson O, Sturgeon C, Topolcan O (April 2003). "Clinical utility of biochemical markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines". European Journal of Cancer. 39 (6): 718–27. doi: 10.1016/S0959-8049(02)00811-0. PMID 12651195.
- ^ Asad-Ur-Rahman F, Saif MW (June 2016). "Elevated Level of Serum Carcinoembryonic Antigen (CEA) and Search for a Malignancy: A Case Report". Cureus. 8 (6): e648. doi: 10.7759/cureus.648. PMC 4954749. PMID 27446768.
- ^ Ballesta AM, Molina R, Filella X, Jo J, Giménez N (1995). "Carcinoembryonic antigen in staging and follow-up of patients with solid tumors". Tumour Biology. 16 (1): 32–41. doi: 10.1159/000217926. PMID 7863220.
- ^ Duffy MJ (April 2001). "Carcinoembryonic antigen as a marker for colorectal cancer: is it clinically useful?". Clinical Chemistry. 47 (4): 624–30. doi: 10.1093/clinchem/47.4.624. PMID 11274010.
- ^ Maestranzi S, Przemioslo R, Mitchell H, Sherwood RA (January 1998). "The effect of benign and malignant liver disease on the tumour markers CA19-9 and CEA". Annals of Clinical Biochemistry. 35 ( Pt 1) (1): 99–103. doi: 10.1177/000456329803500113. PMID 9463746. S2CID 39789615.
- ^ De Mais D (2009). ASCP Quick Compendium of Clinical Pathology (2nd ed.). ASCP Press. ISBN 978-0-89189-567-1.
- ^ Sajid KM, Parveen R, Durr-e-Sabih, Chaouachi K, Naeem A, Mahmood R, Shamim R (December 2007). "Carcinoembryonic antigen (CEA) levels in hookah smokers, cigarette smokers and non-smokers". The Journal of the Pakistan Medical Association. 57 (12): 595–9. PMID 18173042.
- ^ Goldstein MJ, Mitchell EP (2005). "Carcinoembryonic antigen in the staging and follow-up of patients with colorectal cancer". Cancer Investigation. 23 (4): 338–51. doi: 10.1081/CNV-58878. PMID 16100946. S2CID 27100764.
- ^ a b "Carcinoembryonic Antigen (CEA) Exeter Clinical Laboratory International". www.exeterlaboratory.com. Retrieved 2023-10-13.
- ^ Rodriguez-Bigas, Miguel A.; Lin, Edward H.; Crane, Christopher H. (2003), "Tumor Markers", Holland-Frei Cancer Medicine. 6th edition, BC Decker, retrieved 2023-10-13
-
^ Machado-Netto MC, Lacerda EC, Heinke T, Maia DC, Lowen MS, Saad ED (2006).
"Massive orbital metastasis of hepatocellular carcinoma". Clinics (Sao Paulo). 61 (4): 359–62.
doi:
10.1590/s1807-59322006000400015.
PMID
16924330.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link)
" This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0)" - ^ Kankanala, Vijaya L.; Mukkamalla, Shiva Kumar R. (2023), "Carcinoembryonic Antigen", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 35201700, retrieved 2023-10-13
- ^ Leong AS, Cooper K, Leong FJ (2003). Manual of Diagnostic Cytology (2ND ed.). Greenwich Medical Media, Ltd. pp. 51–52. ISBN 978-1-84110-100-2.
- ^ Wong, JY; Chu, DZ; Williams, LE; Liu, A; Zhan, J; Yamauchi, DM; Wilczynski, S; Wu, AM; Yazaki, PJ; Shively, JE; Leong, L; Raubitschek, AA (April 2006). "A phase I trial of (90)Y-DOTA-anti-CEA chimeric T84.66 (cT84.66) radioimmunotherapy in patients with metastatic CEA-producing malignancies". Cancer Biotherapy & Radiopharmaceuticals. 21 (2): 88–100. doi: 10.1089/cbr.2006.21.88. PMID 16706629.
- ^ Chester, Kerry A.; Mayer, Astrid; Bhatia, Jeetendra; Robson, Lynda; Spencer, Daniel I. R.; Cooke, Stephen P.; Flynn, Aiden A.; Sharma, Surinder K.; Boxer, Geoffery; Pedley, R. Barbara; Begent, Richard H. J. (19 July 2000). "Recombinant anti-carcinoembryonic antigen antibodies for targeting cancer". Cancer Chemotherapy and Pharmacology. 46 (S1): S8–S12. doi: 10.1007/PL00014055. PMID 10950140. S2CID 2199095.
- ^ Bacac, Marina; Fauti, Tanja; Sam, Johannes; Colombetti, Sara; Weinzierl, Tina; Ouaret, Djamila; Bodmer, Walter; Lehmann, Steffi; Hofer, Thomas; Hosse, Ralf J.; Moessner, Ekkehard; Ast, Oliver; Bruenker, Peter; Grau-Richards, Sandra; Schaller, Teilo; Seidl, Annette; Gerdes, Christian; Perro, Mario; Nicolini, Valeria; Steinhoff, Nathalie; Dudal, Sherri; Neumann, Sebastian; von Hirschheydt, Thomas; Jaeger, Christiane; Saro, Jose; Karanikas, Vaios; Klein, Christian; Umaña, Pablo (1 July 2016). "A Novel Carcinoembryonic Antigen T-Cell Bispecific Antibody (CEA TCB) for the Treatment of Solid Tumors". Clinical Cancer Research. 22 (13): 3286–3297. doi: 10.1158/1078-0432.CCR-15-1696. PMID 26861458. S2CID 2605514.
- ^ Cheng KT (2013). "99mTc-Arcitumomab". Molecular Imaging and Contrast Agent Database (MICAD). Bethesda (MD): National Center for Biotechnology Information (US).
- ^ Hammarström S (April 1999). "The carcinoembryonic antigen (CEA) family: structures, suggested functions and expression in normal and malignant tissues". Seminars in Cancer Biology. 9 (2): 67–81. doi: 10.1006/scbi.1998.0119. PMID 10202129.
External links
- Carcinoembryonic+Antigen at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- CEA at Lab Tests Online
- CEA: analyte monograph from The Association for Clinical Biochemistry and Laboratory Medicine
- National Cancer Institute Definition of anti-CEA antibody