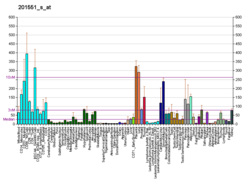
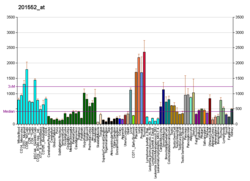
Lysosomal-associated membrane protein 1 (LAMP-1) also known as lysosome-associated membrane glycoprotein 1 and CD107a (Cluster of Differentiation 107a), is a protein that in humans is encoded by the LAMP1 gene. The human LAMP1 gene is located on the long arm (q) of chromosome 13 at region 3, band 4 (13q34).

Lysosomal-associated membrane protein 1 is a glycoprotein from a family of Lysosome-associated membrane glycoproteins. [5] The LAMP-1 glycoprotein is a type I transmembrane protein [6] which is expressed at high or medium levels in at least 76 different normal tissue cell types. [7] It resides primarily across l ysosomal membranes, [8] and functions to provide selectins with carbohydrate ligands. [5] CD107a has also been shown to be a marker of degranulation on lymphocytes such as CD8+ and NK cells, [9] and may also play a role in tumor cell differentiation and metastasis.
Structure
Residing primarily across lysosomal membranes, these glycoproteins consist of a large, highly glycosylated end with N-linked carbon chains on the luminal side of the membrane, and a short C-terminal tail [6] exposed to the cytoplasm. [8] The extracytoplasmic region contains a hinge-like structure which can form disulphide bridges homologous to those observed in human immunoglobulin A. [8] Other characteristics of the structure of the LAMP-1 glycoproteins include:
- A polypeptide core of ~40kDa [8]
- 18 { N-glycosylation} sites to help with the addition of sugar chains [10]
- Polylactosamine attachments which protect the glyocoprotein from degradation by lysosomal proteases [10]
- Significant quantities of polylactosaminoglycan and sialic acid to traverse the trans- Golgi cisternae. [10]
- poly-N-acetyllactosamine groups which are involved in interactions with selectin and other glycan-binding proteins [11]
Function
LAMP1 and LAMP2 glycoproteins comprise 50% of all lysosomal membrane proteins, [6] and are thought to be responsible in part for maintaining lysosomal integrity, pH and catabolism. [6] [11] The expression of LAMP1 and LAMP2 glycoproteins are linked, as deficiencies in LAMP1 gene will lead to increased expression of LAMP2 glycoproteins. [11] The two are therefore thought to share similar functions in vivo. [6] However, this makes the determining the precise function of LAMP1 difficult, because while the LAMP1 deficient phenotype is little different than the wild type due to LAMP2 up regulation, [6] [11] the LAMP1/LAMP2 double deficient phenotype leads to embryonic lethality. [11]
Although the LAMP1 glycoproteins primarily reside across lysosomal membranes, in certain cases they can be expressed across the plasma membrane of the cell. [11] Expression of LAMP1 at the cell surface can occur due to lysosomal fusion with the cell membrane. [12] Cell surface expression of LAMP1 can serve as a ligand for selectins [13] [14] and help mediate cell- cell adhesion. [15] Accordingly, cell surface expression of LAMP1 is seen in cells with migratory or invasive functions, such as cytotoxic T cells, platelets and macrophages. [16] Cell surface expression of LAMP1 and LAMP2 is also often seen in cancer cells, [16] [17] particularly cancers with high metastatic potential, such as colon carcinoma and melanoma, [16] and has been shown to correlate with their metastatic potential. [11]
Role in cancer
LAMP1 expression on the surface of tumor cells has been observed for a number of different cancer types, particularly in highly metastatic cancers such as pancreatic cancer, [18] [19] colon cancer [16] [17] and melanoma. [16] [17] The structure of LAMP1 correlates with differentiation [8] [20] and metastatic potential [11] of tumor cells as it is thought to help mediate cell-cell adhesion [17] and migration. [15] [18] Indeed, the adhesion of some cancer cells to the extracellular matrix is mediated by interactions between LAMP1 and LAMP2 and E-selectin and galectins, with the LAMPs serving as ligands for the cell-adhesion molecules. [17]
Cell membrane expression of LAMP-1 observed in the following cancer types:
- Human fibrosarcoma, [17]
- Colon adenocarcinoma, [17]
- Melanoma, [17]
- Pancreatic adenocarcinoma, [19] and
- Astrocytoma. [18]
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000185896 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000031447 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ a b "LAMP1 lysosomal-associated membrane protein 1". Entrez Gene.
- ^ a b c d e f Eskelinen EL (2006). "Roles of LAMP-1 and LAMP-2 in lysosome biogenesis and autophagy". Molecular Aspects of Medicine. 27 (5–6): 495–502. doi: 10.1016/j.mam.2006.08.005. PMID 16973206.
- ^ "LAMP1". The Human Protein Atlas.
- ^ a b c d e Carlsson SR, Fukuda M (Dec 1989). "Structure of human lysosomal membrane glycoprotein 1. Assignment of disulfide bonds and visualization of its domain arrangement". The Journal of Biological Chemistry. 264 (34): 20526–31. doi: 10.1016/S0021-9258(19)47094-4. PMID 2584229.
- ^ "LAMP1 - lysosomal-associated membrane protein1". Wikigenes.
- ^ a b c Carlsson SR, Roth J, Piller F, Fukuda M (Dec 1988). "Isolation and characterization of human lysosomal membrane glycoproteins, h-lamp-1 and h-lamp-2. Major sialoglycoproteins carrying polylactosaminoglycan". The Journal of Biological Chemistry. 263 (35): 18911–9. doi: 10.1016/S0021-9258(18)37369-1. PMID 3143719.
- ^ a b c d e f g h Andrejewski N, Punnonen EL, Guhde G, Tanaka Y, Lüllmann-Rauch R, Hartmann D, von Figura K, Saftig P (Apr 1999). "Normal lysosomal morphology and function in LAMP-1-deficient mice". The Journal of Biological Chemistry. 274 (18): 12692–701. doi: 10.1074/jbc.274.18.12692. PMID 10212251.
- ^ Kima, P. E.; Burleigh, B.; Andrews, N. W. (Dec 2000). "Surface-targeted lysosomal membrane glycoprotein-1 (Lamp-1) enhances lysosome exocytosis and cell invasion by Trypanosoma cruzi". Cellular Microbiology. 2 (6): 477–486. doi: 10.1046/j.1462-5822.2000.00071.x. ISSN 1462-5814. PMID 11207602. S2CID 19192092.
- ^ Laferte S, Dennis JW (Apr 1989). "Purification of two glycoproteins expressing beta 1-6 branched Asn-linked oligosaccharides from metastatic tumour cells". The Biochemical Journal. 259 (2): 569–576. doi: 10.1042/bj2590569. PMC 1138546. PMID 2719668.
- ^ Sawada R, Jardine KA, Fukuda M (Apr 1993). "The genes of major lysosomal membrane glycoproteins, lamp-1 and lamp-2. 5'-flanking sequence of lamp-2 gene and comparison of exon organization in two genes". The Journal of Biological Chemistry. 268 (12): 9014–9022. doi: 10.1016/S0021-9258(18)52972-0. PMID 8517882.
- ^ a b Acevedo-Schermerhorn C, Gray-Bablin J, Gama R, McCormick PJ (Nov 1997). "t-complex-associated embryonic surface antigen homologous to mLAMP-1. II. Expression and distribution analyses". Experimental Cell Research. 236 (2): 510–518. doi: 10.1006/excr.1997.3752. PMID 9367636.
- ^ a b c d e Agarwal, Akhil Kumar; Srinivasan, Nithya; Godbole, Rashmi; More, Shyam K.; Budnar, Srikanth; Gude, Rajiv P.; Kalraiya, Rajiv D. (2015-01-23). "Role of tumor cell surface lysosome-associated membrane protein-1 (LAMP1) and its associated carbohydrates in lung metastasis". Journal of Cancer Research and Clinical Oncology. 141 (9): 1563–1574. doi: 10.1007/s00432-015-1917-2. ISSN 0171-5216. PMID 25614122. S2CID 9133450.
- ^ a b c d e f g h Sarafian V, Jadot M, Foidart JM, Letesson JJ, Van den Brûle F, Castronovo V, Wattiaux R, Coninck SW (Jan 1998). "Expression of Lamp-1 and Lamp-2 and their interactions with galectin-3 in human tumor cells". International Journal of Cancer. 75 (1): 105–111. doi: 10.1002/(sici)1097-0215(19980105)75:1<105::aid-ijc16>3.0.co;2-f. PMID 9426697.
- ^ a b c Jensen SS, Aaberg-Jessen C, Christensen KG, Kristensen B (2013). "Expression of the lysosomal-associated membrane protein-1 (LAMP-1) in astrocytomas". International Journal of Clinical and Experimental Pathology. 6 (7): 1294–1305. PMC 3693194. PMID 23826410.
- ^ a b Künzli BM, Berberat PO, Zhu ZW, Martignoni M, Kleeff J, Tempia-Caliera AA, Fukuda M, Zimmermann A, Friess H, Büchler MW (Jan 2002). "Influences of the lysosomal associated membrane proteins (Lamp-1, Lamp-2) and Mac-2 binding protein (Mac-2-BP) on the prognosis of pancreatic carcinoma". Cancer. 94 (1): 228–239. doi: 10.1002/cncr.10162. PMID 11815981. S2CID 12702437.
- ^ Lee N, Wang WC, Fukuda M (Nov 1990). "Granulocytic differentiation of HL-60 cells is associated with increase of poly-N-acetyllactosamine in Asn-linked oligosaccharides attached to human lysosomal membrane glycoproteins". The Journal of Biological Chemistry. 265 (33): 20476–87. doi: 10.1016/S0021-9258(17)30529-X. PMID 2243101.
Further reading
- Chang MH, Karageorgos LE, Meikle PJ (2003). "CD107a (LAMP-1) and CD107b (LAMP-2)". Journal of Biological Regulators and Homeostatic Agents. 16 (2): 147–51. PMID 12144129.
- Schleutker J, Haataja L, Renlund M, Puhakka L, Viitala J, Peltonen L, Aula P (Nov 1991). "Confirmation of the chromosomal localization of human lamp genes and their exclusion as candidate genes for Salla disease". Human Genetics. 88 (1): 95–7. doi: 10.1007/BF00204936. PMID 1959930. S2CID 31520394.
- Carlsson SR, Fukuda M (Nov 1990). "The polylactosaminoglycans of human lysosomal membrane glycoproteins lamp-1 and lamp-2. Localization on the peptide backbones". The Journal of Biological Chemistry. 265 (33): 20488–95. doi: 10.1016/S0021-9258(17)30530-6. PMID 2243102.
- Mattei MG, Matterson J, Chen JW, Williams MA, Fukuda M (May 1990). "Two human lysosomal membrane glycoproteins, h-lamp-1 and h-lamp-2, are encoded by genes localized to chromosome 13q34 and chromosome Xq24-25, respectively". The Journal of Biological Chemistry. 265 (13): 7548–51. doi: 10.1016/S0021-9258(19)39148-3. PMID 2332441.
- Carlsson SR, Fukuda M (Dec 1989). "Structure of human lysosomal membrane glycoprotein 1. Assignment of disulfide bonds and visualization of its domain arrangement". The Journal of Biological Chemistry. 264 (34): 20526–31. doi: 10.1016/S0021-9258(19)47094-4. PMID 2584229.
- Mane SM, Marzella L, Bainton DF, Holt VK, Cha Y, Hildreth JE, August JT (Jan 1989). "Purification and characterization of human lysosomal membrane glycoproteins". Archives of Biochemistry and Biophysics. 268 (1): 360–78. doi: 10.1016/0003-9861(89)90597-3. PMID 2912382.
- Viitala J, Carlsson SR, Siebert PD, Fukuda M (Jun 1988). "Molecular cloning of cDNAs encoding lamp A, a human lysosomal membrane glycoprotein with apparent Mr approximately equal to 120,000". Proceedings of the National Academy of Sciences of the United States of America. 85 (11): 3743–7. Bibcode: 1988PNAS...85.3743V. doi: 10.1073/pnas.85.11.3743. PMC 280294. PMID 3131762.
- Howe CL, Granger BL, Hull M, Green SA, Gabel CA, Helenius A, Mellman I (Oct 1988). "Derived protein sequence, oligosaccharides, and membrane insertion of the 120-kDa lysosomal membrane glycoprotein (lgp120): identification of a highly conserved family of lysosomal membrane glycoproteins". Proceedings of the National Academy of Sciences of the United States of America. 85 (20): 7577–81. Bibcode: 1988PNAS...85.7577H. doi: 10.1073/pnas.85.20.7577. PMC 282235. PMID 3174652.
- Fukuda M, Viitala J, Matteson J, Carlsson SR (Dec 1988). "Cloning of cDNAs encoding human lysosomal membrane glycoproteins, h-lamp-1 and h-lamp-2. Comparison of their deduced amino acid sequences". The Journal of Biological Chemistry. 263 (35): 18920–8. doi: 10.1016/S0021-9258(18)37370-8. PMID 3198605.
- Ohno H, Stewart J, Fournier MC, Bosshart H, Rhee I, Miyatake S, Saito T, Gallusser A, Kirchhausen T, Bonifacino JS (Sep 1995). "Interaction of tyrosine-based sorting signals with clathrin-associated proteins". Science. 269 (5232): 1872–5. Bibcode: 1995Sci...269.1872O. doi: 10.1126/science.7569928. PMID 7569928.
- Carlsson SR, Lycksell PO, Fukuda M (Jul 1993). "Assignment of O-glycan attachment sites to the hinge-like regions of human lysosomal membrane glycoproteins lamp-1 and lamp-2". Archives of Biochemistry and Biophysics. 304 (1): 65–73. doi: 10.1006/abbi.1993.1322. PMID 8323299.
- Sawada R, Jardine KA, Fukuda M (Apr 1993). "The genes of major lysosomal membrane glycoproteins, lamp-1 and lamp-2. 5'-flanking sequence of lamp-2 gene and comparison of exon organization in two genes". The Journal of Biological Chemistry. 268 (12): 9014–22. doi: 10.1016/S0021-9258(18)52972-0. PMID 8517882.
- Rohrer J, Schweizer A, Russell D, Kornfeld S (Feb 1996). "The targeting of Lamp1 to lysosomes is dependent on the spacing of its cytoplasmic tail tyrosine sorting motif relative to the membrane". The Journal of Cell Biology. 132 (4): 565–76. doi: 10.1083/jcb.132.4.565. PMC 2199866. PMID 8647888.
- Höning S, Sandoval IV, von Figura K (Aug 1998). "A di-leucine-based motif in the cytoplasmic tail of LIMP-II and tyrosinase mediates selective binding of AP-3". The EMBO Journal. 17 (5): 1304–14. doi: 10.1093/emboj/17.5.1304. PMC 1170479. PMID 9482728.
- Furuta K, Yang XL, Chen JS, Hamilton SR, August JT (May 1999). "Differential expression of the lysosome-associated membrane proteins in normal human tissues". Archives of Biochemistry and Biophysics. 365 (1): 75–82. doi: 10.1006/abbi.1999.1147. PMID 10222041.
- Raposo G, Moore M, Innes D, Leijendekker R, Leigh-Brown A, Benaroch P, Geuze H (Oct 2002). "Human macrophages accumulate HIV-1 particles in MHC II compartments". Traffic. 3 (10): 718–29. doi: 10.1034/j.1600-0854.2002.31004.x. PMID 12230470. S2CID 7055266.
- Zhang H, Li XJ, Martin DB, Aebersold R (Jun 2003). "Identification and quantification of N-linked glycoproteins using hydrazide chemistry, stable isotope labeling and mass spectrometry". Nature Biotechnology. 21 (6): 660–6. doi: 10.1038/nbt827. PMID 12754519. S2CID 581283.
External links
- LAMP1+protein,+human at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- PDBe-KB provides an overview of all the structure information available in the PDB for Human Lysosome-associated membrane glycoprotein 1
This article incorporates text from the
United States National Library of Medicine, which is in the
public domain.