| |||
| Names | |||
|---|---|---|---|
|
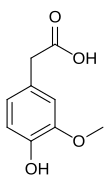
Preferred IUPAC name
(4-Hydroxy-3-methoxyphenyl)acetic acid | |||
| Other names
2-(4-Hydroxy-3-methoxyphenyl)acetic acid; 3-Methoxy-4-hydroxyphenylacetic acid; 4-Hydroxy-3-methoxy-benzeneacetic acid
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.616 | ||
| KEGG | |||
| MeSH | Homovanillic+acid | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C9H10O4 | |||
| Molar mass | 182.175 g·mol−1 | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Homovanillic acid (HVA) is a major catecholamine metabolite that is produced by a consecutive action of monoamine oxidase and catechol-O-methyltransferase on dopamine. [1] Homovanillic acid is used as a reagent to detect oxidative enzymes, and is associated with dopamine levels in the brain.
In psychiatry and neuroscience, brain and cerebrospinal fluid levels of HVA are measured as a marker of metabolic stress caused by 2-deoxy-D-glucose. [2] HVA presence supports a diagnosis of neuroblastoma and malignant pheochromocytoma.
Fasting plasma levels of HVA are known to be higher in females than in males.[ citation needed] This does not seem to be influenced by adult hormonal changes, as the pattern is retained in the elderly and post- menopausal as well as transgender people according to their genetic sex, both before and during cross- sex hormone administration. [3] Differences in HVA have also been correlated to tobacco usage, with smokers showing significantly lower amounts of plasma HVA.
See also
References
- ^ Lambert, G.W.; Eisenhofer, G.; Jennings, G.L.; Esler, M.D. (1993). "Regional homovanillic acid production in humans". Life Sciences. 53 (1): 63–75. doi: 10.1016/0024-3205(93)90612-7. PMID 8515683.
- ^ Marcelis M, Suckling J, Hofman P, Woodruff P, Bullmore E, van Os J (September 2006). "Evidence that brain tissue volumes are associated with HVA reactivity to metabolic stress in schizophrenia". Schizophr. Res. 86 (1–3): 45–53. doi: 10.1016/j.schres.2006.05.001. PMID 16806836.
- ^ Giltay E, Kho K, Blandjaar B, Verbeek M, Geurtz P, Geleijnse J, Gooren L (July 2005). "The sex difference of plasma homovanillic acid is unaffected by cross-sex hormone administration in transgender people". J Endocrinol. 187 (1): 109–16. doi: 10.1677/joe.1.06307. hdl: 2066/47682. PMID 16214946. Archived from the original on 2011-02-27. Retrieved 2011-01-30.