From Wikipedia, the free encyclopedia
Epipregnanolone
Names
IUPAC name
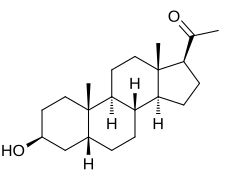
3β-Hydoxy-5β-pregnan-20-one
Systematic IUPAC name
1-[(1S ,3aS ,3bR ,5aR ,7S ,9aS ,9bS ,11aS )-7-Hydroxy-9a,11a-dimethylhexadecahydro-1H -cyclopenta[a ]phenanthren-1-yl]ethan-1-one
Other names
3β,5β-Tetrahydroprogesterone
Identifiers
ChemSpider
UNII
InChI=1S/C21H34O2/c1-13(22)17-6-7-18-16-5-4-14-12-15(23)8-10-20(14,2)19(16)9-11-21(17,18)3/h14-19,23H,4-12H2,1-3H3/t14-,15+,16+,17-,18+,19+,20+,21-/m1/s1
Key: AURFZBICLPNKBZ-GRWISUQFSA-N
CC(=O)[C@H]1CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CC[C@H]4[C@@]3(CC[C@@H](C4)O)C)C
Properties
C 21 H 34 O 2
Molar mass
−1
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Epipregnanolone , also known as 3β-hydroxy-5β-pregnan-20-one , 3β,5β-tetrahydroprogesterone , or 3β,5β-THP , is an
endogenous
neurosteroid .
[1] It acts as a
negative allosteric modulator of the
GABAA receptor and reverses the effects of
potentiators like
allopregnanolone .
[2]
[3] Epipregnanolone is
biosynthesized from
progesterone by the actions of
5β-reductase and
3β-hydroxysteroid dehydrogenase , with
5β-dihydroprogesterone as the
intermediate in this two-step transformation.
[2]
Epipregnanolone is not a
progestogen itself, but via metabolization into other steroids, behaves indirectly as one.
[4]
The
sulfate of epipreganolone,
epipregnanolone sulfate , is a negative allosteric modulator of the
NMDA
[5] and
GABAA receptors
[6] and also acts as a
TRPM3
channel activator .
[7]
[8]
Chemistry See also References
^
Neurosteroids and Brain Function ISBN
978-0-08-054423-6 ^
a b Abraham Weizman (1 February 2008).
Neuroactive Steroids in Brain Function, Behavior and Neuropsychiatric Disorders: Novel Strategies for Research and Treatment ISBN
978-1-4020-6854-6
^ Jan Egebjerg; Arne Schousboe; Povl Krogsgaard-Larsen (4 October 2001).
Glutamate and GABA Receptors and Transporters: Structure, Function and Pharmacology ISBN
978-0-7484-0881-8
^ Beyer, C.; González-Flores, O.; Ramı́rez-Orduña, J.M.; González-Mariscal, G. (1999). "Indomethacin Inhibits Lordosis Induced by Ring A-Reduced Progestins: Possible Role of 3α-Oxoreduction in Progestin-Facilitated Lordosis". Hormones and Behavior . 35 (1): 1–8.
doi :
10.1006/hbeh.1998.1457 .
ISSN
0018-506X .
PMID
10049597 .
S2CID
11520064 .
^ Norman G. Bowery (19 June 2006).
Allosteric Receptor Modulation in Drug Targeting ISBN
978-1-4200-1618-5
^ Park-Chung M, Malayev A, Purdy RH, Gibbs TT, Farb DH (1999).
"Sulfated and unsulfated steroids modulate gamma-aminobutyric acidA receptor function through distinct sites" . Brain Res . 830 (1): 72–87.
doi :
10.1016/s0006-8993(99)01381-5 .
PMID
10350561 .
S2CID
44664830 .
^
Issues in Pharmacology, Pharmacy, Drug Research, and Drug Innovation: 2011 Edition ISBN
978-1-4649-6342-1
^
Advances in Glutamic Acid Research and Application: 2013 Edition: ScholarlyBrief ISBN
978-1-4816-7049-4
Ionotropic
GABAA Tooltip γ-Aminobutyric acid A receptor
Positive modulators (abridged; see
here for a full list):
α-EMTBL
Alcohols (e.g.,
drinking alcohol ,
2M2B )
Anabolic steroids
Avermectins (e.g.,
ivermectin )
Barbiturates (e.g.,
phenobarbital )
Benzodiazepines (e.g.,
diazepam )
Bromide compounds (e.g.,
potassium bromide )
Carbamates (e.g.,
meprobamate )
Carbamazepine
Chloralose
Chlormezanone
Clomethiazole
Dihydroergolines (e.g.,
ergoloid (dihydroergotoxine) )
Etazepine
Etifoxine
Fenamates (e.g.,
mefenamic acid )
Flavonoids (e.g.,
apigenin ,
hispidulin )
Fluoxetine
Flupirtine
Imidazoles (e.g.,
etomidate )
Kava constituents (e.g.,
kavain )
Lanthanum
Loreclezole
Monastrol
Neuroactive steroids (e.g.,
allopregnanolone ,
cholesterol ,
THDOC )
Niacin
Niacinamide
Nonbenzodiazepines (e.g.,
β-carbolines (e.g.,
abecarnil ),
cyclopyrrolones (e.g.,
zopiclone ),
imidazopyridines (e.g.,
zolpidem ),
pyrazolopyrimidines (e.g.,
zaleplon ))
Norfluoxetine
Petrichloral
Phenols (e.g.,
propofol )
Phenytoin
Piperidinediones (e.g.,
glutethimide )
Propanidid
Pyrazolopyridines (e.g.,
etazolate )
Quinazolinones (e.g.,
methaqualone )
Retigabine (ezogabine)
ROD-188
Skullcap constituents (e.g.,
baicalin )
Stiripentol
Sulfonylalkanes (e.g.,
sulfonmethane (sulfonal) )
Topiramate
Valerian constituents (e.g.,
valerenic acid )
Volatiles /
gases (e.g.,
chloral hydrate ,
chloroform ,
diethyl ether ,
paraldehyde ,
sevoflurane ) Negative modulators:
1,3M1B
3M2B
11-Ketoprogesterone
17-Phenylandrostenol
α3IA
α5IA (LS-193,268)
β-CCB
β-CCE
β-CCM
β-CCP
β-EMGBL
Anabolic steroids
Amiloride
Anisatin
β-Lactams (e.g.,
penicillins ,
cephalosporins ,
carbapenems )
Basmisanil
Bemegride
Bicyclic phosphates (
TBPS ,
TBPO ,
IPTBO )
BIDN
Bilobalide
Bupropion
CHEB
Chlorophenylsilatrane
Cicutoxin
Cloflubicyne
Cyclothiazide
DHEA
DHEA-S
Dieldrin
(+)-DMBB
DMCM
DMPC
EBOB
Etbicyphat
FG-7142 (ZK-31906)
Fiproles (e.g.,
fipronil )
Flavonoids (e.g.,
amentoflavone ,
oroxylin A )
Flumazenil
Fluoroquinolones (e.g.,
ciprofloxacin )
Flurothyl
Furosemide
Golexanolone
Iomazenil (123 I)
IPTBO
Isopregnanolone (sepranolone)
L-655,708
Laudanosine
Lindane
MaxiPost
Morphine
Morphine-3-glucuronide
MRK-016
Naloxone
Naltrexone
Nicardipine
Nonsteroidal antiandrogens (e.g.,
apalutamide ,
bicalutamide ,
enzalutamide ,
flutamide ,
nilutamide )
Oenanthotoxin
Pentylenetetrazol (pentetrazol)
Phenylsilatrane
Picrotoxin (i.e.,
picrotin ,
picrotoxinin and
dihydropicrotoxinin )
Pregnenolone sulfate
Propybicyphat
PWZ-029
Radequinil
Ro 15-4513
Ro 19-4603
RO4882224
RO4938581
Sarmazenil
SCS
Suritozole
TB-21007
TBOB
TBPS
TCS-1105
Terbequinil
TETS
Thujone
U-93631
Zinc
ZK-93426
GABAA -ρ Tooltip γ-Aminobutyric acid A-rho receptor
Metabotropic
GABAB Tooltip γ-Aminobutyric acid B receptor
PR Tooltip Progesterone receptor
Agonists
Testosterone derivatives: Progestins:
6,6-Difluoronorethisterone
6,6-Difluoronorethisterone acetate
17α-Allyl-19-nortestosterone
Allylestrenol
Altrenogest
Chloroethynylnorgestrel
Cingestol
Danazol
Desogestrel
Dienogest
Ethinylandrostenediol
Ethisterone
Ethynerone
Etonogestrel
Etynodiol
Etynodiol diacetate
Gestodene
Gestrinone
Levonorgestrel
Levonorgestrel esters (e.g.,
levonorgestrel butanoate )
Lynestrenol
Lynestrenol phenylpropionate
Metynodiol
Metynodiol diacetate
Norelgestromin
Norethisterone (norethindrone)
Norethisterone esters (e.g.,
norethisterone acetate ,
norethisterone enanthate )
Noretynodrel
Norgesterone
Norgestimate
Norgestrel
Norgestrienone
Norvinisterone
Oxendolone
Quingestanol
Quingestanol acetate
Tibolone
Tigestol
Tosagestin ; Anabolic–androgenic steroids:
11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone dodecylcarbonate
19-Nor-5-androstenediol
19-Nor-5-androstenedione
19-Nordehydroepiandrosterone
Bolandiol
Bolandiol dipropionate
Bolandione
Dimethisterone
Dienedione
Dienolone
Dimethandrolone
Dimethandrolone buciclate
Dimethandrolone dodecylcarbonate
Dimethandrolone undecanoate
Dimethyldienolone
Dimethyltrienolone
Ethyldienolone
Ethylestrenol (ethylnandrol)
Methyldienolone
Metribolone (R-1881)
Methoxydienone (methoxygonadiene)
Mibolerone
Nandrolone
Nandrolone esters (e.g.,
nandrolone decanoate ,
nandrolone phenylpropionate )
Norethandrolone
Normethandrone (methylestrenolone, normethandrolone, normethisterone)
RU-2309
Tetrahydrogestrinone
Trenbolone (trienolone)
Trenbolone esters (e.g.,
trenbolone acetate ,
trenbolone enanthate )
Trendione
Trestolone
Trestolone acetate Mixed
SPRMs Tooltip Selective progesterone receptor modulators ) Antagonists
mPR Tooltip Membrane progesterone receptor
PAQR Tooltip Progestin and adipoQ receptor )