 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈaɪbjuːproʊfɛn/, /aɪbjuːˈproʊfən/, EYE-bew-PROH-fən |
| Trade names | Advil, Motrin, Nurofen, others |
| Other names | isobutylphenylpropionic acid |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a682159 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | Oral (by mouth), rectal, topical, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80–100% (orally), [4] 87% (rectal) |
| Protein binding | 98% [5] |
| Metabolism | Liver ( CYP2C9) [5] |
| Metabolites | ibuprofen glucuronide, 2-hydroxyibuprofen, 3-hydroxyibuprofen, carboxy-ibuprofen, 1-hydroxyibuprofen |
| Onset of action | 30 min [6] |
| Elimination half-life | 2–4 h [7] |
| Excretion | Urine (95%) [5] [8] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.036.152 |
| Chemical and physical data | |
| Formula | C13H18O2 |
| Molar mass | 206.285 g·mol−1 |
| 3D model ( JSmol) | |
| Chirality | Racemic mixture |
| Density | 1.03 g/cm3 |
| Melting point | 75 to 78 °C (167 to 172 °F) |
| Boiling point | 157 °C (315 °F) at 4 mmHg |
| Solubility in water | 0.021 mg/mL (20 °C) |
| |
| |
| (verify) | |
Ibuprofen is a nonsteroidal anti-inflammatory drug (NSAID) that is used to relieve pain, fever, and inflammation. [9] This includes painful menstrual periods, migraines, and rheumatoid arthritis. [9] It may also be used to close a patent ductus arteriosus in a premature baby. [3] [9] It can be used orally (by mouth) or intravenously. [9] It typically begins working within an hour. [9]
Common side effects include heartburn and a rash. [9] Compared to other NSAIDs, it may have other side effects such as gastrointestinal bleeding. [10] It increases the risk of heart failure, kidney failure, and liver failure. [9] At low doses, it does not appear to increase the risk of heart attack; however, at higher doses it may. [10] Ibuprofen can also worsen asthma. [10] While its safety in early pregnancy is unclear, [9] it appears to be harmful in later pregnancy, so it is not recommended during that period. [11] Like other NSAIDs, it works by inhibiting the production of prostaglandins by decreasing the activity of the enzyme cyclooxygenase (COX). [9] Ibuprofen is a weaker anti-inflammatory agent than other NSAIDs. [10]
Ibuprofen was discovered in 1961 by Stewart Adams and John Nicholson [12] while working at Boots UK Limited and initially marketed as Brufen. [13] It is available under a number of brand names, including Nurofen, Advil, and Motrin. [9] [14] Ibuprofen was first marketed in 1969 in the United Kingdom and in 1974 in the United States. [9] [13] It is on the World Health Organization's List of Essential Medicines. [15] It is available as a generic medication. [9] In 2021, it was the 36th-most commonly prescribed medication in the United States, with more than 17 million prescriptions. [16] [17]
Medical uses


Ibuprofen is used primarily to treat fever (including postvaccination fever), mild to moderate pain (including pain relief after surgery), painful menstruation, osteoarthritis, dental pain, headaches, and pain from kidney stones. About 60% of people respond to any NSAID; those who do not respond well to a particular one may respond to another. [18] A Cochrane medical review of 51 trials of NSAIDS for the treatment of lower back pain found that "NSAIDs are effective for short-term symptomatic relief in patients with acute low back pain". [19]
It is used for inflammatory diseases such as juvenile idiopathic arthritis and rheumatoid arthritis. [20] [21] It is also used for pericarditis and patent ductus arteriosus. [3] [22] [23]
Ibuprofen lysine
In some countries, ibuprofen lysine (the lysine salt of ibuprofen, sometimes called "ibuprofen lysinate") is licensed for treatment of the same conditions as ibuprofen; the lysine salt is used because it is more water-soluble. [24]
Ibuprofen lysine is sold for rapid pain relief; [25] given in the form of its lysine salt, absorption is much quicker (35 minutes for the salt compared to 90–120 minutes for ibuprofen). However, a clinical trial with 351 participants in 2020, funded by Sanofi, found no significant difference between ibuprofen and ibuprofen lysine concerning the eventual onset of action or analgesic efficacy. [26][ unreliable medical source]
In 2006, ibuprofen lysine was approved in the US by the Food and Drug Administration (FDA) for closure of patent ductus arteriosus in premature infants weighing between 500 and 1,500 g (1 and 3 lb), who are no more than 32 weeks gestational age when usual medical management (such as fluid restriction, diuretics, and respiratory support) is not effective. [27]
Adverse effects
Adverse effects include nausea, indigestion, diarrhea, constipation, gastrointestinal ulceration, headache, dizziness, rash, salt and fluid retention, and high blood pressure. [21] [28]
Infrequent adverse effects include esophageal ulceration, heart failure, high blood levels of potassium, kidney impairment, confusion, and bronchospasm. [21] Ibuprofen can exacerbate asthma, sometimes fatally. [29]
Allergic reactions, including anaphylaxis and anaphylactic shock, may occur. [30] Ibuprofen may be quantified in blood, plasma, or serum to demonstrate the presence of the drug in a person having experienced an anaphylactic reaction, confirm a diagnosis of poisoning in people who are hospitalized, or assist in a medicolegal death investigation. A monograph relating ibuprofen plasma concentration, time since ingestion, and risk of developing renal toxicity in people who have overdosed has been published. [31]
In October 2020, the US FDA required the drug label to be updated for all NSAID medications to describe the risk of kidney problems in unborn babies that result in low amniotic fluid. [32] [33]
Cardiovascular risk
Along with several other NSAIDs, chronic ibuprofen use has been found correlated with risk of progression to hypertension in women, though less than for paracetamol (acetaminophen), [34] and myocardial infarction (heart attack), [35] particularly among those chronically using higher doses. On 9 July 2015, the US FDA toughened warnings of increased heart attack and stroke risk associated with ibuprofen and related NSAIDs; the NSAID aspirin is not included in this warning. [36] The European Medicines Agency (EMA) issued similar warnings in 2015. [37] [38]
Skin
Along with other NSAIDs, ibuprofen has been associated with the onset of bullous pemphigoid or pemphigoid-like blistering. [39] As with other NSAIDs, ibuprofen has been reported to be a photosensitising agent, [40] but it is considered a weak photosensitising agent compared to other members of the 2-arylpropionic acid class. Like other NSAIDs, ibuprofen is an extremely rare cause of the autoimmune disease Stevens–Johnson syndrome (SJS). [41] [42] Ibuprofen is also an extremely rare cause of toxic epidermal necrolysis. [43]
Interactions
Alcohol
Drinking alcohol when taking ibuprofen may increase the risk of stomach bleeding. [44]
Aspirin
According to the FDA, "ibuprofen can interfere with the antiplatelet effect of low-dose aspirin, potentially rendering aspirin less effective when used for cardioprotection and stroke prevention". Allowing sufficient time between doses of ibuprofen and immediate-release (IR) aspirin can avoid this problem. The recommended elapsed time between a dose of ibuprofen and a dose of aspirin depends on which is taken first. It would be 30 minutes or more for ibuprofen taken after IR aspirin, and 8 hours or more for ibuprofen taken before IR aspirin. However, this timing cannot be recommended for enteric-coated aspirin. If ibuprofen is taken only occasionally without the recommended timing, though, the reduction of the cardioprotection and stroke prevention of a daily aspirin regimen is minimal. [45]
Paracetamol
Ibuprofen combined with paracetamol is considered generally safe in children for short-term usage. [46]
Overdose
Ibuprofen overdose has become common since it was licensed for OTC use. Many overdose experiences are reported in the medical literature, although the frequency of life-threatening complications from ibuprofen overdose is low. [47] Human responses in cases of overdose range from an absence of symptoms to a fatal outcome despite intensive-care treatment. Most symptoms are an excess of the pharmacological action of ibuprofen, and include abdominal pain, nausea, vomiting, drowsiness, dizziness, headache, ear ringing, and nystagmus. Rarely, more severe symptoms, such as gastrointestinal bleeding, seizures, metabolic acidosis, hyperkalemia, low blood pressure, slow heart rate, fast heart rate, atrial fibrillation, coma, liver dysfunction, acute kidney failure, cyanosis, respiratory depression, and cardiac arrest have been reported. [48] The severity of symptoms varies with the ingested dose and the time elapsed; however, individual sensitivity also plays an important role. Generally, the symptoms observed with an overdose of ibuprofen are similar to the symptoms caused by overdoses of other NSAIDs.
Correlation between severity of symptoms and measured ibuprofen plasma levels is weak. Toxic effects are unlikely at doses below 100 mg/kg, but can be severe above 400 mg/kg (around 150 tablets of 200 mg units for an average man); [49] however, large doses do not indicate the clinical course is likely to be lethal. [50] A precise lethal dose is difficult to determine, as it may vary with age, weight, and concomitant conditions of the individual person.
Treatment to address an ibuprofen overdose is based on how the symptoms present. In cases presenting early, decontamination of the stomach is recommended. This is achieved using activated charcoal; charcoal absorbs the drug before it can enter the bloodstream. Gastric lavage is now rarely used, but can be considered if the amount ingested is potentially life-threatening, and it can be performed within 60 minutes of ingestion. Purposeful vomiting is not recommended. [51] Most ibuprofen ingestions produce only mild effects, and the management of overdose is straightforward. Standard measures to maintain normal urine output should be instituted and kidney function monitored. [49] Since ibuprofen has acidic properties and is also excreted in the urine, forced alkaline diuresis is theoretically beneficial. However, because ibuprofen is highly protein-bound in the blood, the kidneys' excretion of unchanged drug is minimal. Forced alkaline diuresis is, therefore, of limited benefit. [52]
Miscarriage
A Canadian study of pregnant women suggests that those taking any type or amount of NSAIDs (including ibuprofen, diclofenac, and naproxen) were 2.4 times more likely to miscarry than those not taking the medications. [53] However, an Israeli study found no increased risk of miscarriage in the group of mothers using NSAIDs. [54]
Pharmacology
NSAIDs such as ibuprofen work by inhibiting the cyclooxygenase (COX) enzymes, which convert arachidonic acid to prostaglandin H2 (PGH2). PGH2, in turn, is converted by other enzymes to several other prostaglandins (which are mediators of pain, inflammation, and fever) and to thromboxane A2 (which stimulates platelet aggregation, leading to the formation of blood clots).
Like aspirin and indomethacin, ibuprofen is a nonselective COX inhibitor, in that it inhibits two isoforms of cyclooxygenase, COX-1 and COX-2. The analgesic, antipyretic, and anti-inflammatory activity of NSAIDs appears to operate mainly through inhibition of COX-2, which decreases the synthesis of prostaglandins involved in mediating inflammation, pain, fever, and swelling. Antipyretic effects may be due to action on the hypothalamus, resulting in an increased peripheral blood flow, vasodilation, and subsequent heat dissipation. Inhibition of COX-1 instead would be responsible for unwanted effects on the gastrointestinal tract. [55] However, the role of the individual COX isoforms in the analgesic, anti-inflammatory, and gastric damage effects of NSAIDs is uncertain, and different compounds cause different degrees of analgesia and gastric damage. [56]
Ibuprofen is administered as a racemic mixture. The R-enantiomer undergoes extensive interconversion to the S-enantiomer in vivo. The S-enantiomer is believed to be the more pharmacologically active enantiomer. [57] The R-enantiomer is converted through a series of three main enzymes. These enzymes include acyl-CoA-synthetase, which converts the R-enantiomer to (−)-R-ibuprofen I-CoA; 2-arylpropionyl-CoA epimerase, which converts (−)-R-ibuprofen I-CoA to (+)-S-ibuprofen I-CoA; and hydrolase, which converts (+)-S-ibuprofen I-CoA to the S-enantiomer. [43] In addition to the conversion of ibuprofen to the S-enantiomer, the body can metabolize ibuprofen to several other compounds, including numerous hydroxyl, carboxyl and glucuronyl metabolites. Virtually all of these have no pharmacological effects. [43]
Unlike most other NSAIDs, ibuprofen also acts as an inhibitor of Rho kinase and may be useful in recovery from spinal-cord injury. [58] [59] Another unusual activity is inhibition of the sweet taste receptor. [60]
Pharmacokinetics
After oral administration, peak serum concentration is reached after 1–2 hours, and up to 99% of the drug is bound to plasma proteins. [61] The majority of ibuprofen is metabolized and eliminated within 24 hours in the urine; however, 1% of the unchanged drug is removed through biliary excretion. [57]
Chemistry

Ibuprofen is practically insoluble in water, but very soluble in most organic solvents like ethanol (66.18 g/100 mL at 40 °C for 90% EtOH), methanol, acetone and dichloromethane. [62]
The original synthesis of ibuprofen by the Boots Group started with the compound isobutylbenzene. The synthesis took six steps. A modern, greener technique with fewer waste byproducts for the synthesis involves only three steps was developed in the 1980s by the Celanese Chemical Company. [63] [64] The synthesis is initiated with the acylation of isobutylbenzene using the recyclable Lewis acid catalyst hydrogen fluoride. [65] [66] The following catalytic hydrogenation of isobutylacetophenone is performed with either Raney nickel or palladium on carbon to lead into the key-step, the carbonylation of 1-(4-isobutylphenyl)ethanol. This is achieved by a PdCl2(PPh3)2 catalyst, at around 50 bar of CO pressure, in the presence of HCl (10%). [67] The reaction presumably proceeds through the intermediacy of the styrene derivative (acidic elimination of the alcohol) and (1-chloroethyl)benzene derivative (Markovnikow addition of HCl to the double bond). [68]
Stereochemistry
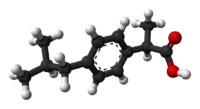
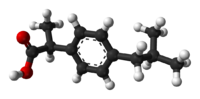
Ibuprofen, like other 2-arylpropionate derivatives such as ketoprofen, flurbiprofen and naproxen, contains a stereocenter in the α-position of the propionate moiety.
 |

|
 |
|
| (R)-ibuprofen | (S)-ibuprofen |
The product sold in pharmacies is a racemic mixture of the S and R-isomers. The S (dextrorotatory) isomer is the more biologically active; this isomer has been isolated and used medically (see dexibuprofen for details). [62]
The isomerase enzyme, alpha-methylacyl-CoA racemase, converts (R)-ibuprofen into the (S)- enantiomer. [69] [70] [71]
(S)-ibuprofen, the eutomer, harbors the desired therapeutic activity. Interestingly, the inactive (R)-enantiomer, the distomer, undergoes a unidirectional chiral inversion to offer the active (S)-enantiomer. That is, when the ibuprofen is administered as a racemate the distomer is converted in vivo into the eutomer while the latter is unaffected. [72] [73] [74]
History

Ibuprofen was derived from propionic acid by the research arm of Boots Group during the 1960s. [75] The name is derived from the 3 functional groups: isobutyl (ibu) propionic acid (pro) phenyl (fen). [76] Its discovery was the result of research during the 1950s and 1960s to find a safer alternative to aspirin. [13] [77] The molecule was discovered and synthesized by a team led by Stewart Adams, with a patent application filed in 1961. [13] Adams initially tested the drug as treatment for his hangover. [78] In 1985, Boots' worldwide patent for ibuprofen expired and generic products were launched. [79]
The medication was launched as a treatment for rheumatoid arthritis in the United Kingdom in 1969, and in the United States in 1974. Later, in 1983 and 1984, it became the first NSAID (other than aspirin) to be available over the counter (OTC) in these two countries. [13] [77] Boots was awarded the Queen's Award for Technical Achievement in 1985 for the development of the drug. [80]
In November 2013, work on ibuprofen was recognized by the erection of a Royal Society of Chemistry blue plaque at Boots' Beeston Factory site in Nottingham, [81] which reads:
In recognition of the work during the 1980s by The Boots Company PLC on the development of ibuprofen which resulted in its move from prescription only status to over the counter sale, therefore expanding its use to millions of people worldwide
and another at BioCity Nottingham, the site of the original laboratory, [81] which reads:
In recognition of the pioneering research work, here on Pennyfoot Street, by Dr Stewart Adams and Dr John Nicholson in the Research Department of Boots which led to the discovery of ibuprofen used by millions worldwide for the relief of pain.
Availability and administration

Ibuprofen was made available by prescription in the United Kingdom in 1969 and in the United States in 1974. [82]
Ibuprofen is the International nonproprietary name (INN), British Approved Name (BAN), Australian Approved Name (AAN) and United States Adopted Name (USAN). In the United States, it has been sold under the brand-names Motrin and Advil since 1974 [83] and 1984, [84] respectively. Ibuprofen is commonly available in the United States up to the FDA's 1984 dose limit OTC, rarely used higher by prescription. [85][ failed verification]
In 2009, the first injectable formulation of ibuprofen was approved in the United States, under the brand name Caldolor. [86] [87]
Ibuprofen can be taken orally (by mouth) (as a tablet, a capsule, or a suspension) and intravenously. [9]
Research
Ibuprofen is sometimes used for the treatment of acne because of its anti-inflammatory properties, and has been sold in Japan in topical form for adult acne. [88] [89] As with other NSAIDs, ibuprofen may be useful in the treatment of severe orthostatic hypotension (low blood pressure when standing up). [90] NSAIDs are of unclear utility in the prevention and treatment of Alzheimer's disease. [91] [92]
Ibuprofen has been associated with a lower risk of Parkinson's disease and may delay or prevent it. Aspirin, other NSAIDs, and paracetamol (acetaminophen) had no effect on the risk for Parkinson's. [93] In March 2011, researchers at Harvard Medical School announced in Neurology that ibuprofen had a neuroprotective effect against the risk of developing Parkinson's disease. [94] [95] [96] People regularly consuming ibuprofen were reported to have a 38% lower risk of developing Parkinson's disease, but no such effect was found for other pain relievers, such as aspirin and paracetamol. Use of ibuprofen to lower the risk of Parkinson's disease in the general population would not be problem-free, given the possibility of adverse effects on the urinary and digestive systems. [97]
Some dietary supplements might be dangerous to take along with ibuprofen and other NSAIDs, but as of 2016 [update] more research needs to be conducted to be certain. These supplements include those that can prevent platelet aggregation, including ginkgo, garlic, ginger, bilberry, dong quai, feverfew, ginseng, turmeric, meadowsweet (Filipendula ulmaria), and willow (Salix spp.); those that contain coumarin, including chamomile, horse chestnut, fenugreek and red clover; and those that increase the risk of bleeding, like tamarind. [98]
References
- ^ Use During Pregnancy and Breastfeeding
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 October 2023.
- ^ a b c "Pedea EPAR". European Medicines Agency (EMA). 29 July 2004. Retrieved 24 February 2024.
- ^ Davanzo R, Bua J, Paloni G, Facchina G (November 2014). "Breastfeeding and migraine drugs". European Journal of Clinical Pharmacology (Review). 70 (11): 1313–1324. doi: 10.1007/s00228-014-1748-0. PMID 25217187. S2CID 17144030.
- ^ a b c Davies NM (February 1998). "Clinical pharmacokinetics of ibuprofen. The first 30 years". Clinical Pharmacokinetics. 34 (2): 101–154. doi: 10.2165/00003088-199834020-00002. PMID 9515184. S2CID 1186212.
- ^ "ibuprofen". Archived from the original on 13 January 2015. Retrieved 31 January 2015.
- ^ Grosser T, Ricciotti E, FitzGerald GA (August 2017). "The Cardiovascular Pharmacology of Nonsteroidal Anti-Inflammatory Drugs". Trends in Pharmacological Sciences (Review). 38 (8): 733–748. doi: 10.1016/j.tips.2017.05.008. PMC 5676556. PMID 28651847.
- ^ "Brufen Tablets And Syrup" (PDF). Therapeutic Goods Administration. 31 July 2012. Archived from the original on 20 August 2016. Retrieved 8 May 2014.
- ^ a b c d e f g h i j k l m "Ibuprofen". The American Society of Health-System Pharmacists. Archived from the original on 9 September 2017. Retrieved 12 October 2016.
- ^ a b c d British National Formulary, March 2014–September 2014 (2014 ed.). London: British Medical Association. 2014. pp. 686–688. ISBN 978-0857110862.
- ^ "Ibuprofen Pregnancy and Breastfeeding Warnings". Drugs.com. Archived from the original on 9 September 2017. Retrieved 22 May 2016.
-
^ Kindy D.
"The Inventor of Ibuprofen Tested the Drug on His Own Hangover". Smithsonian Magazine. Retrieved 3 July 2021.
Stewart Adams and his associate John Nicholson invented a pharmaceutical drug known as 2-(4-isobutylphenyl) propionic acid.
- ^ a b c d e Halford GM, Lordkipanidzé M, Watson SP (2012). "50th anniversary of the discovery of ibuprofen: an interview with Dr Stewart Adams". Platelets. 23 (6): 415–422. doi: 10.3109/09537104.2011.632032. PMID 22098129. S2CID 26344532.
- ^ "Chemistry in your cupboard: Nurofen". RSC Education. Archived from the original on 5 June 2014.
- ^ World Health Organization (2021). World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. hdl: 10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Ibuprofen. Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ "10.1.1 Non-steroidal anti-inflammatory drugs". British National Formulary. Archived from the original on 17 November 2016. Retrieved 13 April 2016.
- ^ Griffin G, Tudiver F, Grant WD (April 2002). "Do NSAIDs help in acute or chronic low back pain?". American Family Physician. 65 (7): 1319–1321. PMID 11996413.
- ^ Joint Formulary Committee (2013). British National Formulary (BNF) (65 ed.). London, UK: Pharmaceutical Press. pp. 665, 671. ISBN 978-0-85711-084-8.
- ^ a b c Rossi S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ Alabed S, Cabello JB, Irving GJ, Qintar M, Burls A (August 2014). "Colchicine for pericarditis" (PDF). The Cochrane Database of Systematic Reviews (Review). 2014 (8): CD010652. doi: 10.1002/14651858.CD010652.pub2. PMC 10645160. PMID 25164988.
- ^ Rostas SE, McPherson CC (2016). "Pharmacotherapy for Patent Ductus Arteriosus: Current Options and Outstanding Questions". Current Pediatric Reviews (Review). 12 (2): 110–119. doi: 10.2174/157339631202160506002028. PMID 27197952.
- ^ Beaver WT (April 2003). "Review of the analgesic efficacy of ibuprofen". International Journal of Clinical Practice. Supplement (135): 13–17. PMID 12723741.
- ^ Seibel K, Schaffler K, Reeh P, Reitmeir P (2004). "Comparison of two different preparations of ibuprofen with regard to the time course of their analgesic effect. A randomised, placebo-controlled, double-blind cross-over study using laser somatosensory evoked potentials obtained from UW-irritated skin in healthy volunteers". Arzneimittel-Forschung. 54 (8): 444–451. doi: 10.1055/s-0031-1296997. PMID 15460211. S2CID 592438.
- ^ Kyselovič J, Koscova E, Lampert A, Weiser T (June 2020). "A Randomized, Double-Blind, Placebo-Controlled Trial of Ibuprofen Lysinate in Comparison to Ibuprofen Acid for Acute Postoperative Dental Pain". Pain and Therapy. 9 (1): 249–259. doi: 10.1007/s40122-019-00148-1. PMC 7203382. PMID 31912434.
- ^ Fanos V, Antonucci R, Zaffanello M (2010). "Ibuprofen and acute kidney injury in the newborn". The Turkish Journal of Pediatrics. 52 (3): 231–238. PMID 20718179.
- ^ Castellsague J, Riera-Guardia N, Calingaert B, Varas-Lorenzo C, Fourrier-Reglat A, Nicotra F, et al. (December 2012). "Individual NSAIDs and upper gastrointestinal complications: a systematic review and meta-analysis of observational studies (the SOS project)". Drug Safety. 35 (12): 1127–46. doi: 10.1007/BF03261999. PMC 3714137. PMID 23137151.
- ^ Ayres JG, Fleming DM, Whittington RM (May 1987). "Asthma death due to ibuprofen". Lancet. 1 (8541): 1082. doi: 10.1016/S0140-6736(87)90499-5. PMID 2883408. S2CID 38589434.
- ^ Shaikhain TA, Al-Husayni F, Elder K (December 2019). "Ibuprofen-induced Anaphylactic Shock in Adult Saudi Patient". Cureus. 11 (12): e6425. doi: 10.7759/cureus.6425. PMC 6970456. PMID 31993263.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, USA: Biomedical Publications. pp. 758–761.
-
^
"FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S.
Food and Drug Administration (FDA) (Press release). 15 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
-
^
"NSAIDs may cause rare kidney problems in unborn babies". U.S.
Food and Drug Administration (FDA). 21 July 2017. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the
public domain.
This article incorporates text from this source, which is in the
public domain.
- ^ Forman JP, Stampfer MJ, Curhan GC (September 2005). "Non-narcotic analgesic dose and risk of incident hypertension in US women". Hypertension. 46 (3): 500–7. doi: 10.1161/01.HYP.0000177437.07240.70. PMID 16103274.
- ^ Hippisley-Cox J, Coupland C (June 2005). "Risk of myocardial infarction in patients taking cyclo-oxygenase-2 inhibitors or conventional non-steroidal anti-inflammatory drugs: population based nested case-control analysis". BMJ. 330 (7504): 1366. doi: 10.1136/bmj.330.7504.1366. PMC 558288. PMID 15947398.
- ^ "FDA Drug Safety Communication: FDA strengthens warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) can cause heart attacks or strokes". U.S. Food and Drug Administration (FDA). 9 July 2015. Archived from the original on 28 October 2019. Retrieved 9 July 2015.
- ^ "Ibuprofen- and dexibuprofen-containing medicines". European Medicines Agency (EMA). 22 May 2015. EMA/325007/2015. Archived from the original on 28 October 2019. Retrieved 28 October 2019.
- ^ "High-dose ibuprofen (≥2400mg/day): small increase in cardiovascular risk". Medicines and Healthcare products Regulatory Agency (MHRA). 26 June 2015. Archived from the original on 28 October 2019. Retrieved 28 October 2019.
- ^ Chan LS (12 June 2014). Hall R, Vinson RP, Nunley JR, Gelfand JM, Elston DM (eds.). "Bullous Pemphigoid Clinical Presentation". Medscape Reference. United States: WebMD. Archived from the original on 10 November 2011.
- ^ Bergner T, Przybilla B (January 1992). "Photosensitization caused by ibuprofen". Journal of the American Academy of Dermatology. 26 (1): 114–6. doi: 10.1016/0190-9622(92)70018-b. PMID 1531054.
- ^ Raksha MP, Marfatia YS (2008). "Clinical study of cutaneous drug eruptions in 200 patients". Indian Journal of Dermatology, Venereology and Leprology. 74 (1): 80. doi: 10.4103/0378-6323.38431. hdl: 1807/48058. PMID 18193504.
- ^ Ward KE, Archambault R, Mersfelder TL (February 2010). "Severe adverse skin reactions to nonsteroidal antiinflammatory drugs: A review of the literature". American Journal of Health-System Pharmacy. 67 (3): 206–13. doi: 10.2146/ajhp080603. PMID 20101062.
- ^ a b c Rainsford KD (2012). Ibuprofen: Pharmacology, Therapeutics and Side Effects. London: Springer.
- ^ "Ibuprofen". Drugs.com. Archived from the original on 6 August 2011.
-
^
"Information for Healthcare Professionals: Concomitant Use of Ibuprofen and Aspirin". U.S.
Food and Drug Administration (FDA). September 2006. Archived from
the original on 13 November 2010. Retrieved 22 November 2010.
- Lay summary in: "Information about Taking Ibuprofen and Aspirin Together". U.S. Food and Drug Administration (FDA). 9 September 2019.
- ^ Kanabar DJ (February 2017). "A clinical and safety review of paracetamol and ibuprofen in children". Inflammopharmacology. 25 (1): 1–9. doi: 10.1007/s10787-016-0302-3. PMC 5306275. PMID 28063133.
- ^ McElwee NE, Veltri JC, Bradford DC, Rollins DE (June 1990). "A prospective, population-based study of acute ibuprofen overdose: complications are rare and routine serum levels not warranted". Annals of Emergency Medicine. 19 (6): 657–662. doi: 10.1016/S0196-0644(05)82471-0. PMID 2188537.
- ^ Vale JA, Meredith TJ (January 1986). "Acute poisoning due to non-steroidal anti-inflammatory drugs. Clinical features and management". Medical Toxicology. 1 (1): 12–31. doi: 10.1007/BF03259825. PMID 3537613. S2CID 25223555.
- ^ a b Volans G, Hartley V, McCrea S, Monaghan J (March–April 2003). "Non-opioid analgesic poisoning". Clinical Medicine. 3 (2): 119–123. doi: 10.7861/clinmedicine.3-2-119. PMC 4952728. PMID 12737366.
- ^ Seifert SA, Bronstein AC, McGuire T (2000). "Massive ibuprofen ingestion with survival". Journal of Toxicology. Clinical Toxicology. 38 (1): 55–57. doi: 10.1081/clt-100100917. PMID 10696926. S2CID 38588541.
- ^ American Academy Of Clinical Toxico (2004). "Position paper: Ipecac syrup". Journal of Toxicology. Clinical Toxicology. 42 (2): 133–143. doi: 10.1081/CLT-120037421. PMID 15214617. S2CID 218865551.
- ^ Hall AH, Smolinske SC, Conrad FL, Wruk KM, Kulig KW, Dwelle TL, et al. (November 1986). "Ibuprofen overdose: 126 cases". Annals of Emergency Medicine. 15 (11): 1308–1313. doi: 10.1016/S0196-0644(86)80617-5. PMID 3777588.
- ^ Verma P, Clark CA, Spitzer KA, Laskin CA, Ray J, Koren G (July 2012). "Use of non-aspirin NSAIDs during pregnancy may increase the risk of spontaneous abortion". Evidence-Based Nursing. 15 (3): 76–77. doi: 10.1136/ebnurs-2011-100439. PMID 22411163. S2CID 28521248.
- ^ Daniel S, Koren G, Lunenfeld E, Bilenko N, Ratzon R, Levy A (March 2014). "Fetal exposure to nonsteroidal anti-inflammatory drugs and spontaneous abortions". CMAJ. 186 (5): E177–E182. doi: 10.1503/cmaj.130605. PMC 3956584. PMID 24491470.
- ^ Rao P, Knaus EE (September 2008). "Evolution of nonsteroidal anti-inflammatory drugs (NSAIDs): cyclooxygenase (COX) inhibition and beyond". Journal of Pharmacy & Pharmaceutical Sciences. 11 (2): 81s–110s. doi: 10.18433/J3T886. PMID 19203472.
- ^ Kakuta H, Zheng X, Oda H, Harada S, Sugimoto Y, Sasaki K, et al. (April 2008). "Cyclooxygenase-1-selective inhibitors are attractive candidates for analgesics that do not cause gastric damage. design and in vitro/in vivo evaluation of a benzamide-type cyclooxygenase-1 selective inhibitor". Journal of Medicinal Chemistry. 51 (8): 2400–2411. doi: 10.1021/jm701191z. PMID 18363350.
- ^ a b "Ibuprofen". DrugBank. Archived from the original on 21 July 2014. Retrieved 24 July 2014.
- ^ Kopp MA, Liebscher T, Niedeggen A, Laufer S, Brommer B, Jungehulsing GJ, et al. (July 2012). "Small-molecule-induced Rho-inhibition: NSAIDs after spinal cord injury". Cell and Tissue Research. 349 (1): 119–132. doi: 10.1007/s00441-012-1334-7. PMC 3744771. PMID 22350947.
- ^ Luo M, Li YQ, Lu YF, Wu Y, Liu R, Zheng YR, et al. (November 2020). "Exploring the potential of RhoA inhibitors to improve exercise-recoverable spinal cord injury: A systematic review and meta-analysis". Journal of Chemical Neuroanatomy. 111: 101879. doi: 10.1016/j.jchemneu.2020.101879. PMID 33197553.
- ^ Nakagita T, Taketani C, Narukawa M, Hirokawa T, Kobayashi T, Misaka T (November 2020). "Ibuprofen, a Nonsteroidal Anti-Inflammatory Drug, is a Potent Inhibitor of the Human Sweet Taste Receptor". Chemical Senses. 45 (8): 667–673. doi: 10.1093/chemse/bjaa057. PMID 32832995.
- ^ Bushra R, Aslam N (July 2010). "An overview of clinical pharmacology of Ibuprofen". Oman Medical Journal. 25 (3): 155–161. doi: 10.5001/omj.2010.49. PMC 3191627. PMID 22043330.
- ^ a b Brayfield A, ed. (14 January 2014). "Ibuprofen". Martindale: The Complete Drug Reference. London, UK: Pharmaceutical Press. Retrieved 26 June 2014.
- ^ "The Synthesis of Ibuprofen". Royal Society of Chemistry. Retrieved 22 November 2018.
- ^ "The Ibuprofen Revolution". Science. Retrieved 17 March 2023.
- ^ Murphy MA (1 July 2018). "Early Industrial Roots of Green Chemistry and the history of the BHC Ibuprofen process invention and its Quality connection". Foundations of Chemistry. 20 (2): 121–165. doi: 10.1007/s10698-017-9300-9. ISSN 1572-8463. S2CID 254510261.
- ^ Grimaldi F, Tran NN, Sarafraz MM, Lettieri P, Morales-Gonzalez OM, Hessel V (4 October 2021). "Life Cycle Assessment of an Enzymatic Ibuprofen Production Process with Automatic Recycling and Purification" (PDF). ACS Sustainable Chemistry & Engineering. 9 (39): 13135–13150. doi: 10.1021/acssuschemeng.1c02309. ISSN 2168-0485.
- ^ US4981995A, Elango V, Murphy MA, Smith BL, Davenport KG, "Method for producing ibuprofen", issued 1991-01-01
- ^ Jayasree S, Seayad A, Chaudhari RV (1999). "Highly active supported palladium catalyst for the regioselective synthesis of 2-arylpropionic acids by carbonylation". Chemical Communications (12): 1067–1068. doi: 10.1039/a902541c. hdl: 1808/18897.
- ^ Tracy TS, Hall SD (March–April 1992). "Metabolic inversion of (R)-ibuprofen. Epimerization and hydrolysis of ibuprofenyl-coenzyme A". Drug Metabolism and Disposition. 20 (2): 322–327. PMID 1352228.
- ^ Chen CS, Shieh WR, Lu PH, Harriman S, Chen CY (July 1991). "Metabolic stereoisomeric inversion of ibuprofen in mammals". Biochimica et Biophysica Acta (BBA) - Protein Structure and Molecular Enzymology. 1078 (3): 411–417. doi: 10.1016/0167-4838(91)90164-U. PMID 1859831.
- ^ Reichel C, Brugger R, Bang H, Geisslinger G, Brune K (April 1997). "Molecular cloning and expression of a 2-arylpropionyl-coenzyme A epimerase: a key enzyme in the inversion metabolism of ibuprofen" (PDF). Molecular Pharmacology. 51 (4): 576–582. doi: 10.1124/mol.51.4.576. PMID 9106621. S2CID 835701. Archived from the original (PDF) on 2 March 2019.
- ^ Caldwell J, Hutt AJ, Fournel-Gigleux S (January 1988). "The metabolic chiral inversion and dispositional enantioselectivity of the 2-arylpropionic acids and their biological consequences". Biochemical Pharmacology. 37 (1): 105–114. doi: 10.1016/0006-2952(88)90762-9. PMID 3276314.
- ^ Simonyi M (1984). "On chiral drug action". Medicinal Research Reviews. 4 (3): 359–413. doi: 10.1002/med.2610040304. PMID 6087043. S2CID 38829275.
- ^ Hutt AJ, Caldwell J (November 1983). "The metabolic chiral inversion of 2-arylpropionic acids--a novel route with pharmacological consequences". The Journal of Pharmacy and Pharmacology. 35 (11): 693–704. doi: 10.1111/j.2042-7158.1983.tb02874.x. PMID 6139449. S2CID 40669413.
- ^ Adams SS (April 1992). "The propionic acids: a personal perspective". Journal of Clinical Pharmacology. 32 (4): 317–323. doi: 10.1002/j.1552-4604.1992.tb03842.x. PMID 1569234. S2CID 22857259.
- ^ "Molecule of the Week Archive: Ibuprofen". American Chemical Society. 14 May 2018. Retrieved 21 March 2024.
- ^ a b Rainsford KD (April 2003). "Discovery, mechanisms of action and safety of ibuprofen". International Journal of Clinical Practice. Supplement (135): 3–8. PMID 12723739.
- ^ Lambert V (8 October 2007). "Dr Stewart Adams: 'I tested ibuprofen on my hangover'". The Daily Telegraph. UK. Archived from the original on 18 November 2015. Retrieved 23 October 2015.(Subscription required.)
- ^ "A brief History of Ibuprofen". Pharmaceutical Journal. 27 July 2017. Retrieved 20 February 2022.
- ^ "Boots Hidden Heroes - Honoring Dr Stewart Adams" (Press release). Boots. Retrieved 20 February 2022.
- ^ a b "Chemical landmark plaque honours scientific discovery past and future" (Press release). Royal Society of Chemistry (RSC). 21 November 2013.
- ^ "Written submission to the NDAC meeting on risks of NSAIDs presented by the International Ibuprofen Foundation". U.S. Food and Drug Administration (FDA). August 2002. Archived from the original on 15 August 2013. Retrieved 20 March 2014.
- ^ "New Drug Application (NDA): 017463". U.S. Food and Drug Administration (FDA). Archived from the original on 28 October 2019. Retrieved 28 October 2019.
- ^ "New Drug Application (NDA): 018989". U.S. Food and Drug Administration (FDA). Archived from the original on 28 October 2019. Retrieved 28 October 2019.
-
^
"Ibuprofen". U.S.
Food and Drug Administration (FDA). Archived from the original on 16 September 2011.
{{ cite web}}: CS1 maint: unfit URL ( link) - ^ "Drug Approval Package: Caldolor (Ibuprofen) NDA #022348". U.S. Food and Drug Administration (FDA). 11 March 2010. Archived from the original on 19 October 2012.
- ^ "FDA Approves Injectable Form of Ibuprofen" (Press release). U.S. Food and Drug Administration (FDA). 11 June 2009. Archived from the original on 18 October 2012.
- ^ Wong RC, Kang S, Heezen JL, Voorhees JJ, Ellis CN (December 1984). "Oral ibuprofen and tetracycline for the treatment of acne vulgaris". Journal of the American Academy of Dermatology. 11 (6): 1076–1081. doi: 10.1016/S0190-9622(84)80192-9. PMID 6239884.
- ^ "In Japan, an OTC ibuprofen ointment (Fukidia) for alleviating adult acne has been launched". Inpharma Weekly. 1 (1530): 18. 25 March 2006. doi: 10.2165/00128413-200615300-00043. ISSN 1173-8324. S2CID 195105870.
- ^ Zawada ET (May 1982). "Renal consequences of nonsteroidal antiinflammatory drugs". Postgraduate Medicine. 71 (5): 223–230. doi: 10.1080/00325481.1982.11716077. PMID 7041104.
- ^ Miguel-Álvarez M, Santos-Lozano A, Sanchis-Gomar F, Fiuza-Luces C, Pareja-Galeano H, Garatachea N, et al. (February 2015). "Non-steroidal anti-inflammatory drugs as a treatment for Alzheimer's disease: a systematic review and meta-analysis of treatment effect". Drugs & Aging. 32 (2): 139–147. doi: 10.1007/s40266-015-0239-z. PMID 25644018. S2CID 35357112.
- ^ Wang J, Tan L, Wang HF, Tan CC, Meng XF, Wang C, et al. (2015). "Anti-inflammatory drugs and risk of Alzheimer's disease: an updated systematic review and meta-analysis". Journal of Alzheimer's Disease. 44 (2): 385–396. doi: 10.3233/JAD-141506. PMID 25227314.
- ^ Chen H, Jacobs E, Schwarzschild MA, McCullough ML, Calle EE, Thun MJ, et al. (December 2005). "Nonsteroidal antiinflammatory drug use and the risk for Parkinson's disease". Annals of Neurology. 58 (6): 963–967. doi: 10.1002/ana.20682. PMID 16240369. S2CID 30843070.
- ^ Bower JH, Ritz B (March 2011). "Is the answer for Parkinson disease already in the medicine cabinet?: Unfortunately not". Neurology. 76 (10): 854–855. doi: 10.1212/WNL.0b013e31820f2e7a. PMID 21368280. S2CID 46104705.
- ^ Gao X, Chen H, Schwarzschild MA, Ascherio A (March 2011). "Use of ibuprofen and risk of Parkinson disease". Neurology. 76 (10): 863–9. doi: 10.1212/WNL.0b013e31820f2d79. PMC 3059148. PMID 21368281.
- ^ McSharry C (May 2011). "Parkinson disease: Could over-the-counter treatment protect against Parkinson disease?". Nature Reviews. Neurology. 7 (5): 244. doi: 10.1038/nrneurol.2011.49. PMID 21555992. S2CID 35880887.
- ^ Gleason JM, Slezak JM, Jung H, Reynolds K, Van den Eeden SK, Haque R, et al. (April 2011). "Regular nonsteroidal anti-inflammatory drug use and erectile dysfunction". The Journal of Urology. 185 (4): 1388–1393. doi: 10.1016/j.juro.2010.11.092. PMID 21334642.
- ^ Abebe W (December 2002). "Herbal medication: potential for adverse interactions with analgesic drugs". Journal of Clinical Pharmacy and Therapeutics. 27 (6): 391–401. doi: 10.1046/j.1365-2710.2002.00444.x. PMID 12472978. S2CID 1828900.
External links
- GB patent 971700, Stewart Sanders Adams & John Stuart Nicholson, "Anti-Inflammatory Agents", published 1964-09-30, assigned to Boots Pure Drug Co Ltd
- "Evidence for the efficacy of pain medications" (PDF). National Safety Council (NSC). 26 August 2020.
- Lowe D. "The Ibuprofen Revolution". Science.