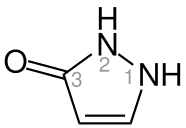Pyrazolone is 5-membered heterocycle containing two adjacent nitrogen atoms. It can be viewed as a derivative of pyrazole possessing an additional carbonyl (C=O) group. Compounds containing this functional group are useful commercially in analgesics and dyes. [1]
Structure and synthesis
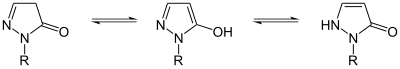
Pyrazolone can exist in two isomers: 3-pyrazolone and 4-pyrazolone.
These isomers can interconvert via lactam–lactim and imine–enamine tautomerism; these conversion often display photochromism. For pyrazolone derivatives, the 3-pyrazolone isomer can be stabilized with N-alkyl or N-aryl substituents.
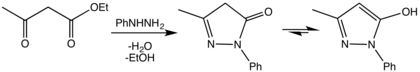
The first synthesis of pyrazolones was reported in 1883 by Ludwig Knorr, via a condensation reaction between ethyl acetoacetate and phenylhydrazine. [2]
Many pyrazolones are produced by functionalization of preformed pyrazolones.
Applications
Pharmaceuticals
This section is missing information about safety profile of analgesics (which ones are withdrawn and whether there is a common theme in adverse effects). (August 2022) |

Pyrazolones are amongst the oldest synthetic pharmaceuticals, starting with the introduction of antipyrine (phenazone) in 1880s. [3] [4] The compounds generally act as analgesics and include dipyrone (Metamizole), aminophenazone, ampyrone, famprofazone, morazone, nifenazone, piperylon and propyphenazone. Of these dipyrone is perhaps the most widely used. [3] [5]
The basic structure has been also used in a number of newer drugs of other effects. [5] Edaravone is useful for prevention and/or therapy of arterial wall injury. [6] Eltrombopag is used to address low blood platelet count.
Dyes
Pyrazolone groups are present in several important dyes. They are commonly used in combination with azo groups to give a sub-family of azo dyes; sometimes referred to as azopyrazolones ( tartrazine, orange B, mordant red 19, yellow 2G). Acid Yellow 17, Acid Yellow 23 (tartrazine), Pigment Yellow 13, and Pigment Red 38 are produced on the multi-ton scale annually. [1]
Ligands
Pyrazolones have been studied as ligands. [7]
References
- ^ a b Kornis, Gabe I. (2000). "Pyrazoles, Pyrazolines, and Pyrazolones". Kirk-Othmer Encyclopedia of Chemical Technology. doi: 10.1002/0471238961.1625180111151814.a01. ISBN 0471238961.
- ^ Knorr, Ludwig (July 1883). "Einwirkung von Acetessigester auf Phenylhydrazin". Berichte der Deutschen Chemischen Gesellschaft (in German). 16 (2): 2597–2599. doi: 10.1002/cber.188301602194.
- ^ a b Brogden, Rex N. (1986). "Pyrazolone Derivatives". Drugs. 32 (Supplement 4): 60–70. doi: 10.2165/00003495-198600324-00006. PMID 3552586. S2CID 2761563.
- ^ Brune, Kay (December 1997). "The early history of non-opioid analgesics". Acute Pain. 1 (1): 33–40. doi: 10.1016/S1366-0071(97)80033-2.
- ^ a b Zhao, Zefeng; Dai, Xufen; Li, Chenyang; Wang, Xiao; Tian, Jiale; Feng, Ying; Xie, Jing; Ma, Cong; Nie, Zhuang; Fan, Peinan; Qian, Mingcheng; He, Xirui; Wu, Shaoping; Zhang, Yongmin; Zheng, Xiaohui (2020). "Pyrazolone structural motif in medicinal chemistry: Retrospect and prospect". European Journal of Medicinal Chemistry. 186: 111893. doi: 10.1016/j.ejmech.2019.111893. PMC 7115706. PMID 31761383.
- ^ Bailly, Christian; Hecquet, Paul-Emile; Kouach, Mostafa; Thuru, Xavier; Goossens, Jean-François (2020). "Chemical reactivity and uses of 1-phenyl-3-methyl-5-pyrazolone (PMP), also known as edaravone". Bioorganic & Medicinal Chemistry. 28 (10): 115463. doi: 10.1016/j.bmc.2020.115463. PMID 32241621. S2CID 214766793.
- ^ CASAS, J; GARCIATASENDE, M; SANCHEZ, A; SORDO, J; TOUCEDA, A (June 2007). "Coordination modes of 5-pyrazolones: A solid-state overview". Coordination Chemistry Reviews. 251 (11–12): 1561–1589. doi: 10.1016/j.ccr.2007.02.010.
External links
- Pyrazolones at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Pubchem - 3-Pyrazolone
- Pubchem - 4-Pyrazolone