 | |
| Clinical data | |
|---|---|
| Other names | Raxatrigine; CNV1014802; GSK-1014802; BIIB 074 |
|
Routes of administration | By mouth |
| ATC code |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
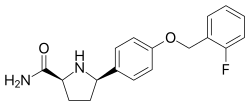
| Formula | C18H19FN2O2 |
| Molar mass | 314.360 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Vixotrigine ( INN, USAN), formerly known as raxatrigine ( INN, USAN), is an analgesic which is under development by Convergence Pharmaceuticals for the treatment of lumbosacral radiculopathy (sciatica) and trigeminal neuralgia (TGN). [1] [2] [3] Vixotrigine was originally claimed to be a selective central Nav1.3 blocker, but was subsequently redefined as a selective peripheral Nav1.7 blocker.[ citation needed] Following this, vixotrigine was redefined once again, as a non-selective voltage-gated sodium channel blocker.[ citation needed] As of January 2018, it is in phase III clinical trials for trigeminal neuralgia and is in phase II clinical studies for erythromelalgia and neuropathic pain. [4] It was previously under investigation for the treatment of bipolar disorder, but development for this indication was discontinued. [4]
See also
References
- ^ "CNV1014802". Convergence Pharmaceuticals.
- ^ McMahon S, Koltzenburg M, Tracey I, Turk DC (1 March 2013). Wall & Melzack's Textbook of Pain: Expert Consult - Online. Elsevier Health Sciences. p. 508. ISBN 978-0-7020-5374-0.
- ^ Bagal SK, Chapman ML, Marron BE, Prime R, Storer RI, Swain NA (August 2014). "Recent progress in sodium channel modulators for pain". Bioorganic & Medicinal Chemistry Letters. 24 (16): 3690–3699. doi: 10.1016/j.bmcl.2014.06.038. PMID 25060923.
- ^ a b "Vixotrigine - Biogen". Adis Insight. Springer Nature Switzerland AG.
External links