Nav1.8 is a sodium ion channel subtype that in humans is encoded by the SCN10A gene. [5] [6] [7] [8]
Nav1.8-containing channels are tetrodotoxin (TTX)-resistant voltage-gated channels. Nav1.8 is expressed specifically in the dorsal root ganglion (DRG), in unmyelinated, small-diameter sensory neurons called C-fibres, and is involved in nociception. [9] [10] C-fibres can be activated by noxious thermal or mechanical stimuli and thus can carry pain messages.
The specific location of Nav1.8 in sensory neurons of the DRG may make it a key therapeutic target for the development of new analgesics [11] and the treatment of chronic pain. [12]
Function
Voltage-gated sodium ion channels (VGSC) are essential in producing and propagating action potentials. Tetrodotoxin, a toxin found in pufferfish, is able to block some VGSCs and therefore is used to distinguish the different subtypes. There are three TTX-resistant VGSC: Nav1.5, Nav1.8 and Nav1.9. Nav1.8 and Nav1.9 are both expressed in nociceptors (damage-sensing neurons). Nav1.7, Nav1.8 and Nav1.9 are found in the DRG and help mediate chronic inflammatory pain. [13] Nav1.8 is an α-type channel subunit consisting of four homologous domains, each with six transmembrane regions, of which one is a voltage sensor.
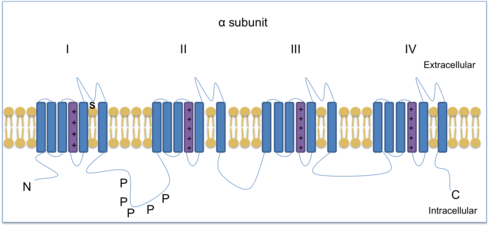
Voltage clamp methods have demonstrated that NaV1.8 is unique, among sodium channels, in exhibiting relatively depolarized steady-state inactivation. Thus, NaV1.8 remains available to operate, when neurons are depolarized to levels that inactivate other sodium channels. Voltage clamp has been used to show how action potentials in DRG cells are shaped by TTX-resistant sodium channels. Nav1.8 contributes the most to sustaining the depolarizing stage of action repetitive high-frequency potentials in nociceptive sensory neurons because it activates quickly and remaining activated after detecting a noxious stimulus. [14] [15] Therefore, Nav1.8 contributes to hyperalgesia (increased sensitivity to pain) and allodynia (pain from stimuli that do not usually cause it), which are elements of chronic pain. [16] Nav1.8 knockout mice studies have shown that the channel is associated with inflammatory and neuropathic pain. [9] [17] [18] Moreover, Nav1.8 plays a crucial role in cold pain. [19] Reducing the temperature from 30 °C to 10 °C slows the activation of VGSCs and hence decreases the current. However, Nav1.8 is cold-resistant and is able to generate action potentials in the cold to carry information from nociceptors to the central nervous system (CNS). Furthermore, Nav1.8-null mice failed to produce action potentials, indicating that Nav1.8 is essential to the perception of pain in cold temperatures. [19]
Although the early studies on the biophysics of NaV1.8 channels were carried out in rodent channels, more recent studies have examined the properties of human NaV1.8 channels. Notably, human NaV1.8 channels exhibit an inactivation voltage-dependence that is even more depolarized than that in rodents, and it also exhibits a larger persistent current. [20] Thus, the influence of human NaV1.8 channels on firing of sensory neurons may be even larger than that of rodent NaV1.8 channels.
Gain-of-function mutations of NaV1.8, identified in patients with painful peripheral neuropathies, have been found to make DRG neurons hyper excitable, and thus are causes of pain. [21] [22] Although NaV1.8 is not normally expressed within the cerebellum, its expression is up-regulated in cerebellar Purkinje cells in animal models of MS (Multiple Sclerosis), and in human MS. [23] The presence of NaV1.8 channels within these cerebellar neurons, where it is not normally present, increases their excitability and alters their firing pattern in vitro, [24] and in rodents with experimental autoimmune encephalomyelitis, a model of MS. [25] At a behavioral level, the ectopic expression of NaV1.8 within cerebellar Purkinje neurons has been shown to impair motor performance in a transgenic model. [26]
Clinical significance
Pain signalling pathways
Nociceptors are different from other sensory neurons in that they have a low activating threshold and consequently increase their response to constant stimuli. Therefore, nociceptors are easily sensitised by agents such as bradykinin and nerve growth factor, which are released at the site of tissue injury, ultimately causing changes to ion channel conductance. VGSCs have been shown to increase in density after nerve injury. [27] Therefore, VGSCs can be modulated by many different hyperalgesic agents that are released after nerve injury. Further examples include prostaglandin E2 (PGE2), serotonin and adenosine, which all act to increase the current through Nav1.8. [28]
Prostaglandins such as PGE2 can sensitise nociceptors to thermal, chemical and mechanical stimuli and increase the excitability of DRG sensory neurons. This occurs because PGE2 modulates the trafficking of Nav1.8 by binding to G-protein-coupled EP2 receptor, which in turn activates protein kinase A. [29] [30] Protein kinase A phosphorylates Nav1.8 at intracellular sites, resulting in increased sodium ion currents. Evidence for a link between PGE2 and hyperalgesia comes from an antisense deoxynucleotide knockdown of Nav1.8 in the DRG of rats. [31] Another modulator of Nav1.8 is the ε isoform of PKC. This isoform is activated by the inflammatory mediator bradykinin and phosphorylates Nav1.8, causing an increase in sodium current in the sensory neurons, which promotes mechanical hyperalgesia. [32]
Brugada syndrome
Mutations in SCN10A are associated with Brugada syndrome. [33] [34] [35]
Membrane trafficking
Nerve growth factor levels in inflamed or injured tissues are increased creating an increased sensitivity to pain (hyperalgesia). [36] The increased levels of nerve growth factor and tumour necrosis factor-α (TNF-α) causes the upregulation of Nav1.8 in sensory neurons via the accessory protein p11 (annexin II light chain). It has been shown using the yeast-two hybrid screening method that p11 binds to a 28-amino-acid fragment at the N terminus of Nav1.8 and promotes its translocation to the plasma membrane. This contributes to the hyperexcitability of sensory neurons during pain. [37] p11-null nociceptive sensory neurons in mice, created using the Cre-loxP recombinase system, show a decrease in Nav1.8 expression at the plasma membrane. [38] Therefore, disrupting the interactions between p11 and Nav1.8 may be a good therapeutic target for lowering pain.
In myelinated fibres, VGSCs are located at the nodes of Ranvier; however, in unmyelinated fibres, the exact location of VGSCs has not been determined. Nav1.8 in unmyelinated fibres has been found in clusters associated with lipid rafts along DRG fibers both in vitro and in vivo. [39] Lipid rafts organise the cell membrane, which includes trafficking and localising ion channels. Removal of lipid rafts in the membrane using MβCD, which depletes cholesterol from the plasma membrane, leads to a shift of Nav1.8 to a non-raft portion of the membrane, causing reduced action potential firing and propagation. [39]
Painful peripheral neuropathies
Painful peripheral neuropathies or small-fibre neuropathies are disorders of unmyelinated nociceptive C-fibres causing neuropathic pain; in some cases there is no known cause. [40] Genetic screening of patients with these idiopathic neuropathies has uncovered mutations in the SCN9A gene, encoding the related channel Nav1.7. A gain-of-function mutation in Nav1.7 located in the DRG sensory neurons was found in nearly 30% of patients with idiopathic small fiber neuropathy in one study. [41] This gain-of-function mutation causes an increase in excitability (hyperexcitability) of DRG sensory neurons and thus an increase in pain. Nav1.7 thus been shown to be linked to human pain; Nav1.8, by contrast, had only been associated to pain in animal studies until recently. A gain-of-function mutation was found in the Nav1.8-encoding SCN10A gene in patients with painful peripheral neuropathy. [21] A study of 104 patients with idiopathic peripheral neuropathies who did not have the mutation in SCN9A used voltage clamp and current clamp methods, along with predictive algorithms, and yielded two gain-of-function mutations in SCN10A in three patients. Both mutations cause increased excitability in DRG sensory neurons and hence contribute to pain, but the mechanism by which they do so is not understood.
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000185313 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000034533 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Entrez Gene: sodium channel".
- ^ Rabert DK, Koch BD, Ilnicka M, Obernolte RA, Naylor SL, Herman RC, Eglen RM, Hunter JC, Sangameswaran L (November 1998). "A tetrodotoxin-resistant voltage-gated sodium channel from human dorsal root ganglia, hPN3/SCN10A". Pain. 78 (2): 107–14. doi: 10.1016/S0304-3959(98)00120-1. PMID 9839820. S2CID 45480324.
- ^ Plummer NW, Meisler MH (April 1999). "Evolution and diversity of mammalian sodium channel genes". Genomics. 57 (2): 323–31. doi: 10.1006/geno.1998.5735. PMID 10198179.
- ^ Catterall WA, Goldin AL, Waxman SG (December 2005). "International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels". Pharmacological Reviews. 57 (4): 397–409. doi: 10.1124/pr.57.4.4. PMID 16382098. S2CID 7332624.
- ^ a b Akopian AN, Souslova V, England S, Okuse K, Ogata N, Ure J, Smith A, Kerr BJ, McMahon SB, Boyce S, Hill R, Stanfa LC, Dickenson AH, Wood JN (June 1999). "The tetrodotoxin-resistant sodium channel SNS has a specialized function in pain pathways". Nature Neuroscience. 2 (6): 541–8. doi: 10.1038/9195. PMID 10448219. S2CID 17487906.
- ^ Akopian AN, Sivilotti L, Wood JN (January 1996). "A tetrodotoxin-resistant voltage-gated sodium channel expressed by sensory neurons". Nature. 379 (6562): 257–62. Bibcode: 1996Natur.379..257A. doi: 10.1038/379257a0. PMID 8538791. S2CID 4360775.
- ^ Cummins TR, Sheets PL, Waxman SG (October 2007). "The roles of sodium channels in nociception: Implications for mechanisms of pain". Pain. 131 (3): 243–57. doi: 10.1016/j.pain.2007.07.026. PMC 2055547. PMID 17766042.
- ^ a b Swanwick RS, Pristerá A, Okuse K (December 2010). "The trafficking of Na(V)1.8". Neuroscience Letters. 486 (2): 78–83. doi: 10.1016/j.neulet.2010.08.074. PMC 2977848. PMID 20816723.
- ^ Strickland IT, Martindale JC, Woodhams PL, Reeve AJ, Chessell IP, McQueen DS (July 2008). "Changes in the expression of NaV1.7, NaV1.8 and NaV1.9 in a distinct population of dorsal root ganglia innervating the rat knee joint in a model of chronic inflammatory joint pain". European Journal of Pain. 12 (5): 564–72. doi: 10.1016/j.ejpain.2007.09.001. PMID 17950013. S2CID 24952010.
- ^ Blair NT, Bean BP (2002). "Roles of Tetrodotoxin (TTX)-Sensitive Na+ Current, TTX-Resistant Na+ Current, and Ca2+ Current in the Action Potentials of Nociceptive Sensory Neurons". The Journal of Neuroscience. 22 (23): 10277–10290. doi: 10.1523/JNEUROSCI.22-23-10277.2002. PMC 6758735. PMID 12451128.
- ^ Renganathan M, Cummins TR & Waxman SG (2001). "Contribution of Nav1.8 Sodium Channels to Action Potential Electrogenesis in DRG Neurons". Journal of Neurophysiology. 86 (2): 629–640. doi: 10.1152/jn.2001.86.2.629. PMID 11495938. S2CID 11579149.
- ^ Millan MJ (1999). "The induction of pain: an integrative review". Progress in Neurobiology. 57 (1): 1–164. doi: 10.1016/S0301-0082(98)00048-3. PMID 9987804. S2CID 206054345.
- ^ Matthews EA, Wood JN, Dickenson AH (February 2006). "Na(v) 1.8-null mice show stimulus-dependent deficits in spinal neuronal activity". Molecular Pain. 2: 1744-8069–2-5. doi: 10.1186/1744-8069-2-5. PMC 1403745. PMID 16478543.
- ^ Jarvis MF, Honore P, Shieh CC, Chapman M, Joshi S, Zhang XF, Kort M, Carroll W, Marron B, Atkinson R, Thomas J, Liu D, Krambis M, Liu Y, McGaraughty S, Chu K, Roeloffs R, Zhong C, Mikusa JP, Hernandez G, Gauvin D, Wade C, Zhu C, Pai M, Scanio M, Shi L, Drizin I, Gregg R, Matulenko M, Hakeem A, Gross M, Johnson M, Marsh K, Wagoner PK, Sullivan JP, Faltynek CR, Krafte DS (May 2007). "A-803467, a potent and selective Nav1.8 sodium channel blocker, attenuates neuropathic and inflammatory pain in the rat". Proceedings of the National Academy of Sciences of the United States of America. 104 (20): 8520–5. doi: 10.1073/pnas.0611364104. PMC 1895982. PMID 17483457.
- ^ a b Zimmermann K, Leffler A, Babes A, Cendan CM, Carr RW, Kobayashi J, Nau C, Wood JN, Reeh PW (June 2007). "Sensory neuron sodium channel Nav1.8 is essential for pain at low temperatures". Nature. 447 (7146): 855–8. Bibcode: 2007Natur.447..856Z. doi: 10.1038/nature05880. PMID 17568746. S2CID 4391511.
- ^ Han C, Estacion M, Huang J, Vasylyev D, Zhao P, Dib-Hajj SD, Waxman SG (May 2015). "Human Na(v)1.8: enhanced persistent and ramp currents contribute to distinct firing properties of human DRG neurons". Journal of Neurophysiology. 113 (9): 3172–85. doi: 10.1152/jn.00113.2015. PMC 4432682. PMID 25787950.
- ^ Huang J, Yang Y, Zhao P, Gerrits MM, Hoeijmakers JG, Bekelaar K, Merkies IS, Faber CG, Dib-Hajj SD, Waxman SG (August 2013). "Small-fiber neuropathy Nav1.8 mutation shifts activation to hyperpolarized potentials and increases excitability of dorsal root ganglion neurons". The Journal of Neuroscience. 33 (35): 14087–97. doi: 10.1523/JNEUROSCI.2710-13.2013. PMC 6618513. PMID 23986244.
- ^ Black JA, Dib-Hajj S, Baker D, Newcombe J, Cuzner ML, Waxman SG (October 2000). "Sensory neuron-specific sodium channel SNS is abnormally expressed in the brains of mice with experimental allergic encephalomyelitis and humans with multiple sclerosis". Proceedings of the National Academy of Sciences of the United States of America. 97 (21): 11598–602. Bibcode: 2000PNAS...9711598B. doi: 10.1073/pnas.97.21.11598. PMC 17246. PMID 11027357.
- ^ Renganathan M, Gelderblom M, Black JA, Waxman SG (January 2003). "Expression of Nav1.8 sodium channels perturbs the firing patterns of cerebellar Purkinje cells". Brain Research. 959 (2): 235–42. doi: 10.1016/s0006-8993(02)03750-2. PMID 12493611. S2CID 34784900.
- ^ Saab CY, Craner MJ, Kataoka Y, Waxman SG (September 2004). "Abnormal Purkinje cell activity in vivo in experimental allergic encephalomyelitis". Experimental Brain Research. 158 (1): 1–8. doi: 10.1007/s00221-004-1867-4. PMID 15118796. S2CID 34656521.
- ^ Shields SD, Cheng X, Gasser A, Saab CY, Tyrrell L, Eastman EM, Iwata M, Zwinger PJ, Black JA, Dib-Hajj SD, Waxman SG (February 2012). "A channelopathy contributes to cerebellar dysfunction in a model of multiple sclerosis". Annals of Neurology. 71 (2): 186–94. doi: 10.1002/ana.22665. PMID 22367990. S2CID 25128887.
- ^ Devor M; Govrin-Lippmann R & Angelides (1993). "Na+ Channel lmmunolocalization in Peripheral Mammalian Axons and Changes following Nerve Injury and Neuroma Formation". The Journal of Neuroscience. 13 (5): 1976–1992. doi: 10.1523/JNEUROSCI.13-05-01976.1993. PMC 6576562. PMID 7683047.
- ^ Gold MS, Reichling DB, Shuster MJ, Levine JD (February 1996). "Hyperalgesic agents increase a tetrodotoxin-resistant Na+ current in nociceptors". Proceedings of the National Academy of Sciences of the United States of America. 93 (3): 1108–12. Bibcode: 1996PNAS...93.1108G. doi: 10.1073/pnas.93.3.1108. PMC 40039. PMID 8577723.
- ^ Hector TH (January 1975). "A simple method for making chromatographic records using transparent acetate sheet". The Journal of Physiology. 32 (1): 31–2. doi: 10.1113/jphysiol.1996.sp021604. PMC 1160802. PMID 8887754.
- ^ Liu C, Li Q, Su Y, Bao L (March 2010). "Prostaglandin E2 promotes Na1.8 trafficking via its intracellular RRR motif through the protein kinase A pathway". Traffic. 11 (3): 405–17. doi: 10.1111/j.1600-0854.2009.01027.x. PMID 20028484. S2CID 997800.
- ^ Khasar SG, Gold MS & Levine JD (1998). "A tetrodotoxin-resistant sodium current mediates inflammatory pain in the rat". Neuroscience Letters. 256 (1): 17–20. doi: 10.1016/s0304-3940(98)00738-1. PMID 9832206. S2CID 5614913.
- ^ Wu DF, Chandra D, McMahon T, Wang D, Dadgar J, Kharazia VN, Liang YJ, Waxman SG, Dib-Hajj SD, Messing RO (April 2012). "PKCε phosphorylation of the sodium channel NaV1.8 increases channel function and produces mechanical hyperalgesia in mice". The Journal of Clinical Investigation. 122 (4): 1306–15. doi: 10.1172/JCI61934. PMC 3315445. PMID 22426212.
- ^ Hu D, Barajas-Martínez H, Pfeiffer R, Dezi F, Pfeiffer J, Buch T, Betzenhauser MJ, Belardinelli L, Kahlig KM, Rajamani S, DeAntonio HJ, Myerburg RJ, Ito H, Deshmukh P, Marieb M, Nam GB, Bhatia A, Hasdemir C, Haïssaguerre M, Veltmann C, Schimpf R, Borggrefe M, Viskin S, Antzelevitch C (July 2014). "Mutations in SCN10A are responsible for a large fraction of cases of Brugada syndrome". Journal of the American College of Cardiology. 64 (1): 66–79. doi: 10.1016/j.jacc.2014.04.032. PMC 4116276. PMID 24998131.
-
^ Monasky MM, Micaglio E, Vicedomini G, Locati ET, Ciconte G, Giannelli L, Giordano F, Crisà S, Vecchi M, Borrelli V, Ghiroldi A, D'Imperio S, Di Resta C, Benedetti S, Ferrari M, Santinelli V, Anastasia L, Pappone C (2019).
"Comparable clinical characteristics in Brugada syndrome patients harboring SCN5A or novel SCN10A variants". Europace. 21 (10): 1550–1558.
doi:
10.1093/europace/euz186.
PMID
31292628. Retrieved 27 April 2021.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) -
^ Monasky MM, Micaglio E, Ciconte G, Pappone C (2020).
"Brugada Syndrome: Oligogenic or Mendelian Disease?". Int J Mol Sci. 21 (5): 1687.
doi:
10.3390/ijms21051687.
PMC
7084676.
PMID
32121523.
{{ cite journal}}: CS1 maint: multiple names: authors list ( link) - ^ McMahon SB (March 1996). "NGF as a mediator of inflammatory pain". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 351 (1338): 431–40. Bibcode: 1996RSPTB.351..431M. doi: 10.1098/rstb.1996.0039. PMID 8730782.
- ^ Okuse K, Malik-Hall M, Baker MD, Poon WY, Kong H, Chao MV, Wood JN (June 2002). "Annexin II light chain regulates sensory neuron-specific sodium channel expression". Nature. 417 (6889): 653–6. Bibcode: 2002Natur.417..653O. doi: 10.1038/nature00781. PMID 12050667. S2CID 4423351.
- ^ Foulkes T, Nassar MA, Lane T, Matthews EA, Baker MD, Gerke V, Okuse K, Dickenson AH, Wood JN (October 2006). "Deletion of annexin 2 light chain p11 in nociceptors causes deficits in somatosensory coding and pain behavior" (PDF). The Journal of Neuroscience. 26 (41): 10499–507. doi: 10.1523/JNEUROSCI.1997-06.2006. PMC 6674704. PMID 17035534.
- ^ a b Pristerà A, Baker MD, Okuse K (2012). "Association between tetrodotoxin resistant channels and lipid rafts regulates sensory neuron excitability". PLOS ONE. 7 (8): e40079. Bibcode: 2012PLoSO...740079P. doi: 10.1371/journal.pone.0040079. PMC 3411591. PMID 22870192.
- ^ Hoeijmakers JG, Faber CG, Lauria G, Merkies IS, Waxman SG (May 2012). "Small-fibre neuropathies--advances in diagnosis, pathophysiology and management". Nature Reviews. Neurology. 8 (7): 369–79. doi: 10.1038/nrneurol.2012.97. PMID 22641108. S2CID 8804151.
- ^ Faber CG, Hoeijmakers JG, Ahn HS, Cheng X, Han C, Choi JS, Estacion M, Lauria G, Vanhoutte EK, Gerrits MM, Dib-Hajj S, Drenth JP, Waxman SG, Merkies IS (January 2012). "Gain of function Naν1.7 mutations in idiopathic small fiber neuropathy". Annals of Neurology. 71 (1): 26–39. doi: 10.1002/ana.22485. PMID 21698661. S2CID 11711575.
Further reading
- Okuse K (2007). "Pain signalling pathways: from cytokines to ion channels". The International Journal of Biochemistry & Cell Biology. 39 (3): 490–6. doi: 10.1016/j.biocel.2006.11.016. PMID 17194618.
- Waxman SG (July 2013). "Painful Na-channelopathies: an expanding universe". Trends in Molecular Medicine. 19 (7): 406–9. doi: 10.1016/j.molmed.2013.04.003. PMID 23664154.
- Lai J, Porreca F, Hunter JC, Gold MS (2004). "Voltage-gated sodium channels and hyperalgesia". Annual Review of Pharmacology and Toxicology. 44: 371–97. doi: 10.1146/annurev.pharmtox.44.101802.121627. PMID 14744251.
- Wood JN, Boorman JP, Okuse K, Baker MD (October 2004). "Voltage-gated sodium channels and pain pathways". Journal of Neurobiology. 61 (1): 55–71. doi: 10.1002/neu.20094. PMID 15362153.
- Malik-Hall M, Poon WY, Baker MD, Wood JN, Okuse K (February 2003). "Sensory neuron proteins interact with the intracellular domains of sodium channel NaV1.8". Brain Research. Molecular Brain Research. 110 (2): 298–304. doi: 10.1016/S0169-328X(02)00661-7. PMID 12591166.
- Yamaoka K, Inoue M, Miyazaki K, Hirama M, Kondo C, Kinoshita E, Miyoshi H, Seyama I (March 2009). "Synthetic ciguatoxins selectively activate Nav1.8-derived chimeric sodium channels expressed in HEK293 cells". The Journal of Biological Chemistry. 284 (12): 7597–605. doi: 10.1074/jbc.M806481200. PMC 2658054. PMID 19164297.
- Choi JS, Hudmon A, Waxman SG, Dib-Hajj SD (July 2006). "Calmodulin regulates current density and frequency-dependent inhibition of sodium channel Nav1.8 in DRG neurons". Journal of Neurophysiology. 96 (1): 97–108. doi: 10.1152/jn.00854.2005. PMID 16598065.
- Liu CJ, Priest BT, Bugianesi RM, Dulski PM, Felix JP, Dick IE, Brochu RM, Knaus HG, Middleton RE, Kaczorowski GJ, Slaughter RS, Garcia ML, Köhler MG (February 2006). "A high-capacity membrane potential FRET-based assay for NaV1.8 channels". Assay and Drug Development Technologies. 4 (1): 37–48. doi: 10.1089/adt.2006.4.37. PMID 16506887.
- Browne LE, Blaney FE, Yusaf SP, Clare JJ, Wray D (April 2009). "Structural determinants of drugs acting on the Nav1.8 channel". The Journal of Biological Chemistry. 284 (16): 10523–36. doi: 10.1074/jbc.M807569200. PMC 2667739. PMID 19233853.
- Rabert DK, Koch BD, Ilnicka M, Obernolte RA, Naylor SL, Herman RC, Eglen RM, Hunter JC, Sangameswaran L (November 1998). "A tetrodotoxin-resistant voltage-gated sodium channel from human dorsal root ganglia, hPN3/SCN10A". Pain. 78 (2): 107–14. doi: 10.1016/S0304-3959(98)00120-1. PMID 9839820. S2CID 45480324.
- Plummer NW, Meisler MH (April 1999). "Evolution and diversity of mammalian sodium channel genes". Genomics. 57 (2): 323–31. doi: 10.1006/geno.1998.5735. PMID 10198179.
- Catterall WA, Goldin AL, Waxman SG (December 2005). "International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels". Pharmacological Reviews. 57 (4): 397–409. doi: 10.1124/pr.57.4.4. PMID 16382098. S2CID 7332624.



