 | |
| Clinical data | |
|---|---|
|
Routes of administration | Oral |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | 80–90% |
| Protein binding | 5–7% |
| Metabolism |
Hepatic
deacetylation Minor involvement of CYP2D6 and CYP2A6 |
| Elimination half-life | 5–7 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| Chemical and physical data | |
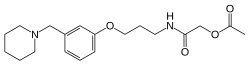
| Formula | C19H28N2O4 |
| Molar mass | 348.443 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Roxatidine acetate is a specific and competitive histamine H2 receptor antagonist drug that is used to treat gastric ulcers, Zollinger–Ellison syndrome, erosive esophagitis, gastro-oesophageal reflux disease, and gastritis. [1] [2]
Pharmacodynamic studies showed that 150 mg of roxatidine acetate were optimal in suppressing gastric acid secretion, and that a single bedtime dose of 150 mg was more effective than a dose of 75 mg twice daily in terms of inhibiting nocturnal acid secretion. [1]
It was patented in 1979 and approved for medical use in 1986. [3] It is available in countries including China, Japan, Korea, Germany, Italy, the Netherlands, Greece and South Africa. [2]
References
- ^ a b Murdoch D, McTavish D (August 1991). "Roxatidine acetate. A review of its pharmacodynamic and pharmacokinetic properties, and its therapeutic potential in peptic ulcer disease and related disorders". Drugs. 42 (2): 240–260. doi: 10.2165/00003495-199142020-00006. PMID 1717223. S2CID 46973503.
- ^ a b BioSpectrum Bureau 1 November 2012 Sinhuan's generic heart drug gets production approval
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 444. ISBN 9783527607495.