 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Surmontil, others |
| Other names | Trimeproprimine; IF-6120; IL-6001; RP-7162; 2'-Methylimipramine; β-Methylimipramine |
| AHFS/ Drugs.com | |
| MedlinePlus | a602010 |
| License data |
|
|
Pregnancy category |
|
|
Routes of administration | Oral, intramuscular injection, intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 41% [2] [3] [4] [5] |
| Protein binding | 94.9% [2] [3] [4] [5] |
| Metabolism | Hepatic [2] [3] [4] [5] |
| Elimination half-life | 23–24 hours [2] [3] [4] [5] |
| Excretion | Renal [2] [3] [4] [5] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.010.917 |
| Chemical and physical data | |
| Formula | C20H26N2 |
| Molar mass | 294.442 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
Trimipramine, sold under the brand name Surmontil among others, is a tricyclic antidepressant (TCA) which is used to treat depression. [6] [7] [8] [9] It has also been used for its sedative, anxiolytic, and weak antipsychotic effects in the treatment of insomnia, anxiety disorders, and psychosis, respectively. [6] [7] [8] [9] The drug is described as an atypical or "second-generation" TCA because, unlike other TCAs, it seems to be a fairly weak monoamine reuptake inhibitor. [10] Similarly to other TCAs, however, trimipramine does have antihistamine, antiserotonergic, antiadrenergic, antidopaminergic, and anticholinergic activities. [6] [7] [8] [9]
Medical uses
Trimipramine's primary use in medicine is in the treatment of major depressive disorder, [11] [12] especially where sedation is helpful due to its prominent sedative effects. [12] The drug is also an effective anxiolytic, and can be used in the treatment of anxiety. [8] [9] In addition to depression and anxiety, trimipramine is effective in the treatment of insomnia, and unlike most other hypnotics, does not alter the normal sleep architecture. [8] In particular, it does not suppress REM sleep, and dreams are said to "brighten" during treatment. [8] [13]
Trimipramine also has some weak antipsychotic effects with a profile of activity described as similar to that of clozapine, and may be useful in the treatment of psychotic symptoms, such as in delusional depression, schizoaffective disorder or schizophrenia. [6] [7]
A major systematic review and network meta-analysis of medications for the treatment of insomnia published in 2022 found that trimipramine had an effect size ( standardized mean difference (SMD)) against placebo for treatment of insomnia at 4 weeks of 0.55 (95% CI –0.11 to 1.21). [14] The certainty of evidence was rated as very low, and no data were available for longer-term treatment (3 months). [14] For comparison, the other sedating antihistamines assessed, doxepin and doxylamine, had effect sizes (SMD) at 4 weeks of 0.30 (95% CI –0.05 to 0.64) (very low certainty evidence) and 0.47 (95% CI 0.06 to 0.89) (moderate certainty evidence), respectively. [14]
The effective dosage of trimipramine in depression is 150 to 300 mg/day. [9] Doses of trimipramine used for insomnia range from 25 to 200 mg/day. [15] [16] [17] However, it has been advised that doses be kept as low as possible, and a low dose of 25 mg/day has been recommended. [15]
Contraindications
Contraindications include: [2] [3] [4] [5] [11] [12]
- Recent myocardial infarction
- Any degree of heart block or other cardiac arrhythmias
- Mania
- Severe liver disease
- During breastfeeding
- Hypersensitivity to trimipramine or to any of the excipients
Side effects
The side effects of trimipramine have been said to be similar to those of other tertiary amine TCAs, with a preponderance of anticholinergic and sedative effects. [9] However, trimipramine has also been said to be associated with a different side effect profile compared to other TCAs and in general with fewer side effects, chiefly due to its lack of norepinephrine reuptake inhibition and relatively lower anticholinergic effects (although it is still a potent anticholinergic). [7] [9] Somnolence is the most common side effect of the drug. [9] Dry mouth is the most common anticholinergic side effect, but others like constipation, urinary retention, and blurred vision are also present. [9]
It is described as being associated with minimal or no orthostatic hypotension, at least in comparison to clomipramine, [6] [7] in spite of its potent and comparable activity as an alpha-1 blocker. [18] [9] However, it has also been said to have a rate of orthostatic hypotension similar to that of other TCAs. [9] Trimipramine is said to be less epileptogenic than other TCAs, although seizures have still been reported in association with it. [7] It is also less cardiotoxic than other TCAs [7] and cardiotoxicity is said to be minimal, with a "very favorable profile". [9]
Heavy exposure to any tricyclic antidepressants was associated with an elevated rate ratio for breast cancer 11–15 years later. [19] However, on tests done on Drosophila melanogaster, nongenotoxic TCAs (amitriptyline, maprotiline, nortriptyline, and protriptyline), and genotoxic TCAs (amoxapine, clomipramine, desipramine, doxepin, imipramine, and trimipramine) were identified. [19]
List of side effects
Common adverse effects include: [2] [3] [4] [5] [11] [12]
- Sedation — especially common with trimipramine compared to the other TCAs
- Anticholinergic effects including:
- - dry mouth
- - blurred vision
- - mydriasis
- - decreased lacrimation
- - constipation
- - urinary hesitancy or retention
- - reduced GI motility
- - tachycardia (high heart rate)
- - anticholinergic delirium (particularly in the elderly and in Parkinson's disease)
- Weight gain
- Orthostatic hypotension
- Sexual dysfunction including impotence, loss of libido and other sexual adverse effects
- Tremor
- Dizziness
- Sweating
- Anxiety
- Insomnia
- Agitation
- Rash
Adverse effects with an unknown incidence includes: [2] [3] [4] [5] [11] [12]
- Confusion
- Nausea
- Vomiting
- Extrapyramidal side effects (e.g. parkinsonism, dystonia, etc.)
- Tinnitus
- Paraesthesia
- ECG changes
- Increased liver function tests
Rare adverse effects include: [2] [3] [4] [5] [11] [12]
- Seizures
- Syndrome of inappropriate secretion of antidiuretic hormone
- Blood dyscrasias including:
- Myocardial infarction
- Heart block
- QTc interval prolongation
- Sudden cardiac death
- Depression worsening
- Suicidal ideation
Overdose
Compared to other TCAs, trimipramine is relatively safe in overdose, although it is more dangerous than the selective serotonin reuptake inhibitors (SSRIs) and serotonin–norepinephrine reuptake inhibitors (SNRIs) but less dangerous than bupropion in cases of overdose. [20]
Interactions
Trimipramine should not be given with sympathomimetic agents such as epinephrine (adrenaline), ephedrine, isoprenaline, norepinephrine (noradrenaline), phenylephrine and phenylpropanolamine.
Barbiturates may increase the rate of metabolism. Trimipramine should be administered with care in patients receiving therapy for hyperthyrodism. [2] [3] [4] [5] [11] [12]
Pharmacology
Pharmacodynamics
| Site | Ki/ IC50 (nM) | Species | Ref |
|---|---|---|---|
| SERT | 149–2,110 | Human | [22] [23] |
| NET | 2,450–4,990 | Human | [22] [23] |
| DAT | ≥3,780 | Human | [22] [23] |
| 5-HT1A | 8,000 | Human | [24] |
| 5-HT1D | >10,000 | Pig | [25] |
| 5-HT2A | 32 | Human | [24] |
| 5-HT2C | 537 | Pig | [25] |
| 5-HT3 | 9,120 | Rat | [25] |
| α1 | 24 | Human | [18] |
| α2 | 680 | Human | [18] |
| D1 | 347 | Pig | [25] |
| D2 | 143–210 | Human/undefined | [18] [26] [25] |
| D3 | ND | ND | ND |
| D4 | 275 | Undefined | [25] |
| H1 | 0.27–1.48 | Human | [27] [18] [28] |
| H2 | 41 | Human | [27] |
| H3 | >100,000 | Human | [27] |
| H4 | 43,700 | Human | [27] |
| mACh | 58 | Human | [18] |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. | |||
The mechanism of action of trimipramine in terms of its antidepressant effects differs from that of other TCAs and is not fully clear. [23] [8] The mechanism of action of its anxiolytic effects is similarly unclear. [8] Trimipramine is a very weak reuptake inhibitor of serotonin, norepinephrine, and dopamine (see below), [23] and unlike most other TCAs, has been claimed to be devoid of clinically significant monoamine reuptake inhibition. [10] [7] [6] The effects of the drug are thought to be mainly due to receptor antagonism as follows: [18] [26] [24]
- Very strong: H1
- Strong: 5-HT2A, α1-adrenergic
- Moderate: D2, mACh
- Weak: 5-HT2C, D1, α2-adrenergic
In spite of its atypical nature and different profile of activity, trimipramine has been shown in head-to-head clinical studies to possess equivalent effectiveness to other antidepressants, including but not limited to other TCAs (e.g., amitriptyline, imipramine, doxepin, amineptine), tetracyclic antidepressants (TeCAs) (e.g., maprotiline), monoamine oxidase inhibitors (MAOIs) (e.g., phenelzine, isocarboxazid), and selective serotonin reuptake inhibitors (e.g., fluoxetine). [6] [7] In addition, trimipramine has been found to possess greater anxiolytic effects than other TCAs such as amitriptyline and doxepin in head-to-head comparisons. [8] Indeed, its prominent anxiolytic effects have been said to distinguish it from most other TCAs. [9] The atypicality of trimipramine in relation to its lack of monoamine reuptake inhibition is described as challenging the monoamine hypothesis of depression. [7]
The major metabolite of trimipramine, desmethyltrimipramine, is considered to possess pharmacological activity similar to that of other demethylated tertiary amine TCA variants. [23]
Monoamine reuptake inhibition
| SERT | NET | DAT | Type | Species | Tissue | Year/Ref |
|---|---|---|---|---|---|---|
| 8,200 | 1,000 | 6,800 | IC50 (nM) | Rat | Brain | 1977 [29] |
| 2,500 | 510 | 3,400 | Ki (nM) | Rat | Brain | 1984 [30] |
| 149 | 2,450 | 3,780 | KD (nM) | Human | HEK293 | 1997 [22] |
| 2,110 | 4,990 | 55,600 | IC50 (nM) | Human | HEK293 | 2011 [23] |
Studies have generally found only very weak inhibition of serotonin and norepinephrine reuptake with trimipramine, [23] and the drug has been described by various authors as devoid of monoamine reuptake inhibition. [10] Richelson & Pfenning (1984) found a relatively high Ki for the NET of 510 nM in rat brain synaptosomes [30] and Tatsumi et al. (1997) found a relatively high KD of 149 nM for the SERT in human HEK293 cells, [22] but other authors and a more recent study with an improved design have not had the same findings. [23] In the most recent study, by Haenisch et al. (2011), the researchers suggested that the discrepant findings from the Tatsumi et al. study were due to methodological differences, in particular the use of radioligand binding in isolated membranes (KD) to study interactions as opposed to actual functional reuptake inhibition (IC50). [23]
| Compound | SERT | NET | DAT |
|---|---|---|---|
| Trimipramine | 5.675 | 5.302 | >30 μM |
| Desmethyltrimipramine | 5.206 | 5.535 | 4.530 |
| 2-Hydroxytrimipramine | >30 μM | 4.960 | 4.585 |
| Trimipramine-N-oxide | 5.445 | 4.930 | 5.027 |
| Values are
pIC50. The higher the value, the more strongly the drug binds to the site. | |||
Trimipramine is extensively metabolized, so its metabolites may contribute to its pharmacology, including potentially to monoamine reuptake inhibition. [23] [31] In what was the only study to date to have assessed the activity profiles of the metabolites of trimipramine, Haenisch et al. (2011) assayed desmethyltrimipramine, 2-hydroxytrimipramine, and trimipramine-N-oxide in addition to trimipramine and found that these metabolites showed IC50 values for the SERT, NET, and DAT similar to those of trimipramine (see table to the right). [23] [31] Like other secondary amine TCAs, desmethyltrimipramine was slightly more potent than trimipramine in its norepinephrine reuptake inhibition but less potent in its inhibition of serotonin reuptake. [23] However, desmethyltrimipramine still showed only very weak inhibition of the NET. [23]
Therapeutic concentrations of trimipramine are between 0.5 and 1.2 μM (150–350 ng/mL) and hence significant monoamine reuptake inhibition would not be expected with it or its metabolites. [23] However, these concentrations are nearly 2-fold higher if the active metabolites of trimipramine are also considered, and studies of other TCAs have found that they cross the blood–brain barrier and accumulate in the brain to levels of up to 10-fold those in the periphery. [23] As such, trimipramine and its metabolites might at least partially inhibit reuptake of serotonin and/or norepinephrine, though not of dopamine, at therapeutic concentrations, and this could be hypothesized to contribute at least in part to its antidepressant effects. [23] [31] This is relevant as Haenisch et al. has stated that these are the only actions known at present which could explain or at least contribute to the antidepressant effects of trimipramine. [23] That said, blockade of the 5-HT2A, 5-HT2C, and α2-adrenergic receptors, as with mirtazapine, [32] has also been implicated in antidepressant effects. [33] [10] [34]
In any case, there is also clinical and animal evidence that trimipramine does not inhibit the reuptake of monoamines. [7] Unlike other TCAs, it does not downregulate β3-adrenergic receptors, which is likely the reason that it does not cause orthostatic hypotension. [7] [6] It can be safely combined with MAOIs apparently without risk of serotonin syndrome or hypertensive crisis. [7] Indeed, in rabbits, whereas hyperpyrexia (a symptom of serotonin syndrome) occurs with imipramine and an MAOI and to a lesser extent with amitriptyline and an MAOI, it does not occur at all with trimipramine and an MAOI, likely due to trimipramine's lack of serotonin reuptake inhibition. [7]
Antihistamine activity
Trimipramine is a very potent antihistamine; it has the third highest affinity for the H1 receptor (Ki = 0.27 nM) after mirtazapine (Ki = 0.14 nM) and doxepin (Ki = 0.24 nM) among the TCAs and tetracyclic antidepressants (TeCAs). [18] [35] The TeCA mianserin (Ki = 0.40) and the TCA amitriptyline (Ki = 1.0) are also very potent H1 receptor antagonists, [18] [35] [36] whereas other TCAs and TeCAs are less potent. [10] These TCAs and TeCAs, including trimipramine, are far more potent than the standard antihistamine diphenhydramine (approximately 800 times for doxepin and 250 times for trimipramine), [37] and are among the most potent antihistamines available. [36] [38]
Trimipramine is also an antagonist of the H2 receptor with lower potency and has been found to be effective in the treatment of duodenal ulcers. [9]
As a hypnotic
Blockade of the H1 receptor is responsible for the sedative effects of trimipramine and other TCAs and their effectiveness in the treatment of insomnia. [39]
Most antidepressants suppress REM sleep, in parallel with their alleviation of depressive symptoms (although suppression of REM sleep is not required for antidepressant effects). [6] This includes TCAs (e.g., amitriptyline, nortriptyline), TeCAs (e.g., mianserin, maprotiline), MAOIs (e.g., clorgiline, pargyline), and SSRIs (e.g., fluoxetine, zimelidine, indalpine). [6] Trimipramine is unique in that it is an exception and produces antidepressant effects without compromising or otherwise affecting REM sleep. [6] [9] Even long-term treatment with trimipramine for up to 2 years has not been found to suppress REM sleep. [40] In addition, trimipramine has been found to decrease nocturnal cortisol levels to normal and to normalize cortisol response in depressed patients; hence, it normalizes the hypothalamic–pituitary–adrenal axis, whereas imipramine and other antidepressants tend to increase nocturnal cortisol secretion. [25]
In clinical studies, trimipramine has been found in doses of 50 to 200 mg/day to significantly increase sleep efficiency and total sleep time and to decrease waking time for up to 3 weeks in patients with insomnia. [6] It also improved subjectively perceived sleep quality and well-being during daytime. [6] Monitoring of patients upon discontinuation of trimipramine found that it did not cause rebound insomnia or worsening of sleep quality in subjective evaluations of sleep, although objective measurements found total sleep time below baseline in a subset of patients during trimipramine withdrawal. [6]
Antidopaminergic activity
| Site | Trimipramine | Clozapine | |
|---|---|---|---|
| 5-HT2A | 7.71 | 7.84 | |
| α1A/B | 7.62 | 8.54 | |
| D2 | 7.24 | 7.01 | |
| D1 | 6.46 | 6.58 | |
| α2B | 6.42 | 7.08 | |
| 5-HT2C | 6.27 | 8.40 | |
| α2A | 5.86 | 7.10 | |
| 5-HT3 | 5.04 | 7.00 | |
| 5-HT1A | <5.00 | 6.17 | |
| 5-HT1D | <5.00 | 5.68 | |
| Values are pKi (nM). The higher the value, the more strongly the drug binds to the site. | |||
Trimipramine is a weak but significant antagonist of the dopamine D1 and D2 receptors, and also binds to the D4 receptor (Ki = 275 nM). [6] [23] Its affinities for various monoamine receptors including the D2 and 5-HT2A receptors closely resemble those of the atypical antipsychotic clozapine. [6] In accordance, high doses of trimipramine have been found to have antipsychotic effects in schizophrenic patients, notably without causing extrapyramidal symptoms, and trimipramine has recently been found to be effective in reducing psychotic symptoms in patients with delusional depression. [6] [23] The lack of extrapyramidal symptoms with trimipramine may be related to its affinity for the D4 receptor, these both being properties it shares with clozapine. [25] Unlike other TCAs, but reminiscent of antipsychotics, trimipramine has been found to markedly increase plasma prolactin levels (a marker of D2 receptor antagonism) at a dose of 75 mg/day and to increase nocturnal prolactin secretion at doses of 75 and 200 mg/day. [6] These findings are suggestive of important antidopaminergic actions of trimipramine. [6] [23]
Unlike various other TCAs, trimipramine shows marked antagonism of presynaptic dopamine autoreceptors, potentially resulting in increased dopaminergic neurotransmission. [7] This effect has also been observed with low-potency tricyclic antipsychotics like thioridazine and chlorprothixene. [7] Notably, these two antipsychotics have been claimed many times to also possess antidepressant effects. [7] [40] As such, blockade of inhibitory dopamine autoreceptors and hence facilitation of dopaminergic signaling could be involved in the antidepressant effects of trimipramine. [7] [40] However, other authors have attributed the claimed antidepressant effects of antipsychotics like the two previously mentioned to α2-adrenergic receptor antagonism, although trimipramine specifically has only weak affinity for this receptor. [7] Aside from antidepressant effects, low doses of antipsychotics have been found to increase REM sleep, and so dopamine autoreceptor antagonism could be involved in the unique effects of trimipramine in terms of REM sleep and sleep architecture. [40]
Pharmacokinetics
The time to peak concentrations following a dose is 2 to 4 hours. [9] The typical antidepressant therapeutic range of trimipramine concentrations is 150 to 300 ng/mL. [41] The terminal half-life of trimipramine has been variously reported to be as little as 8 hours (in plasma) [7] and as long as 24 hours. [9] In any case, the terminal half-life of trimipramine is described as shorter than that of other TCAs, which makes it ideal for use in the treatment of insomnia. [9] [7]
Trimipramine is a racemic compound with two enantiomers. [1] CYP2C19 is responsible for the demethylation of (D)- and (L)-trimipramine to (D)- (L)-desmethyltrimipramine, respectively, and CYP2D6 is responsible for the 2- hydroxylation of ( D)- and ( L)-desmethyltrimipramine to (D)- and (L)-2-hydroxydesmethyltrimipramine, respectively. [42] CYP2D6 also metabolizes (L)-trimipramine into (L)-2-hydroxytrimipramine. [42]
Chemistry
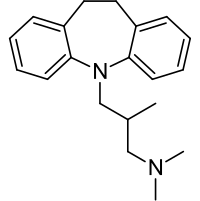
Trimipramine is a tricyclic compound, specifically a dibenzazepine, and possesses three rings fused together with a side chain attached in its chemical structure. [43] Other dibenzazepine TCAs include imipramine, desipramine, and clomipramine. [43] Trimipramine is a derivative of imipramine with a methyl group added to its side chain [44] and is also known as 2'-methylimipramine or β-methylimipramine. [43] [45] The tri- prefix in its name may allude to the fact that its side chain features three methyl groups. Trimipramine is a tertiary amine TCA, with its side chain- demethylated metabolite desmethyltrimipramine being a secondary amine. [46] [47] Other tertiary amine TCAs include amitriptyline, imipramine, clomipramine, dosulepin (dothiepin), and doxepin. [48] [49] The chemical name of trimipramine is 3-(10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)-N,N,2-trimethylpropan-1-amine and its free base form has a chemical formula of C20H26N2 with a molecular weight of 294.434 g/mol. [50] The drug is used commercially as the maleate salt. [50] [51] The CAS Registry Number of the free base is 739-71-9 and of the maleate is 521-78-8. [50] [51]
History
Trimipramine was developed by Rhône-Poulenc. [52] It was patented in 1959 and first appeared in the literature in 1961. [52] The drug was first introduced for medical use in 1966, in Europe. [52] [53] It was not introduced in the United States until later in 1979 or 1980. [54] [55]
Society and culture
Generic names
Trimipramine is the generic name of the drug and its INN, USAN, BAN, and DCF, while trimipramine maleate is its USAN, USP, BANM, and JAN. [50] [51] [56] [57] Its generic name in Latin is trimipraminum, in German is trimipramin, and in Spanish is trimipramina. [51] [57]
Brand names
Trimipramine is marketed throughout the world mainly under the brand name Surmontil. [51] [57] Other notable brand names of trimipramine have included Herphonal, Rhotrimine, Sapilent, Stangyl, and Tydamine. [51] [57]
Availability
Trimipramine is no longer marketed in Australia, though it was previously. [58] [59]
In film
The sedative effects of Trimipramine in off-prescription, recreational use are described in the 1987 film Withnail and I where the eponymous character declares "This is the plan. We get in there and get wrecked, then we'll eat a pork pie, then we'll drop a couple of Surmontil-50's each. That means we'll miss out Monday but come up smiling Tuesday morning." [60]
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- ^ a b c d e f g h i j "PRODUCT INFORMATION SURMONTIL Tablets and Capsules" (PDF). TGA eBusiness Services. Aspen Pharmacare Australia Pty Ltd. 28 November 2012. Retrieved 30 November 2013.
- ^ a b c d e f g h i j "SURMONTIL (trimipramine maleate) capsule [Duramed Pharmaceuticals Inc]". DailyMed. Duramed Pharmaceuticals Inc. December 2012. Retrieved 30 November 2013.
- ^ a b c d e f g h i j "Surmontil, Trimip (trimipramine) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 30 November 2013.
- ^ a b c d e f g h i j "Trimipramine 50mg Capsules - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Zentiva. 19 November 2012. Retrieved 30 November 2013.
- ^ a b c d e f g h i j k l m n o p q r s t Berger M, Gastpar M (1996). "Trimipramine: a challenge to current concepts on antidepressives". Eur Arch Psychiatry Clin Neurosci. 246 (5): 235–9. doi: 10.1007/bf02190274. PMID 8863001. S2CID 29596291.
- ^ a b c d e f g h i j k l m n o p q r s t u v Gastpar M (1989). "Clinical originality and new biology of trimipramine". Drugs. 38 (Suppl 1): 43–8, discussion 49–50. doi: 10.2165/00003495-198900381-00010. PMID 2693055. S2CID 23302529.
- ^ a b c d e f g h i Pecknold JC, Luthe L (1989). "Trimipramine, anxiety, depression and sleep". Drugs. 38 (Suppl 1): 25–31, discussion 49–50. doi: 10.2165/00003495-198900381-00007. PMID 2693052. S2CID 20347877.
- ^ a b c d e f g h i j k l m n o p q r Lapierre YD (1989). "A review of trimipramine. 30 years of clinical use". Drugs. 38 (Suppl 1): 17–24, discussion 49–50. doi: 10.2165/00003495-198900381-00006. PMID 2693051. S2CID 22227558.
- ^ a b c d e Frazer A (1997). "Pharmacology of antidepressants". J Clin Psychopharmacol. 17 (Suppl 1): 2S–18S. doi: 10.1097/00004714-199704001-00002. PMID 9090573.
- ^ a b c d e f Rossi, S, ed. (2013). Australian Medicines Handbook (2013 ed.). Adelaide: The Australian Medicines Handbook Unit Trust. ISBN 978-0-9805790-9-3.
- ^ a b c d e f g Joint Formulary Committee (2013). British National Formulary (BNF) (65 ed.). London, UK: Pharmaceutical Press. ISBN 978-0-85711-084-8.
- ^ Schredl M, Berger M, Riemann D. The effect of trimipramine on dream recall and dream emotions in depressive outpatients. Psychiatry Res. 2009 May 30;167(3):279-86. doi: 10.1016/j.psychres.2008.03.002. Epub 2009 Apr 28. PMID 19403177.
- ^ a b c De Crescenzo F, D'Alò GL, Ostinelli EG, Ciabattini M, Di Franco V, Watanabe N, Kurtulmus A, Tomlinson A, Mitrova Z, Foti F, Del Giovane C, Quested DJ, Cowen PJ, Barbui C, Amato L, Efthimiou O, Cipriani A (July 2022). "Comparative effects of pharmacological interventions for the acute and long-term management of insomnia disorder in adults: a systematic review and network meta-analysis". Lancet. 400 (10347): 170–184. doi: 10.1016/S0140-6736(22)00878-9. hdl: 11380/1288245. PMID 35843245. S2CID 250536370.
- ^ a b Wiegand MH (2008). "Antidepressants for the treatment of insomnia : a suitable approach?". Drugs. 68 (17): 2411–7. doi: 10.2165/0003495-200868170-00001. PMID 19016570. S2CID 28009108.
- ^ Everitt H, Baldwin DS, Stuart B, Lipinska G, Mayers A, Malizia AL, Manson CC, Wilson S (May 2018). "Antidepressants for insomnia in adults". Cochrane Database Syst Rev. 2018 (5): CD010753. doi: 10.1002/14651858.CD010753.pub2. PMC 6494576. PMID 29761479.
- ^ Ioachimescu OC, El-Solh AA (June 2012). "Pharmacotherapy of insomnia". Expert Opin Pharmacother. 13 (9): 1243–60. doi: 10.1517/14656566.2012.683860. PMID 22578014. S2CID 9177139.
- ^ a b c d e f g h i Richelson E, Nelson A (1984). "Antagonism by antidepressants of neurotransmitter receptors of normal human brain in vitro". J. Pharmacol. Exp. Ther. 230 (1): 94–102. PMID 6086881.
- ^ a b Sharpe CR, Collet JP, Belzile E, Hanley JA, Boivin JF (January 2002). "The effects of tricyclic antidepressants on breast cancer risk". British Journal of Cancer. 86 (1): 92–97. doi: 10.1038/sj.bjc.6600013. PMC 2746543. PMID 11857018.
- ^ White N, Litovitz T, Clancy C (December 2008). "Suicidal antidepressant overdoses: a comparative analysis by antidepressant type". Journal of Medical Toxicology. 4 (4): 238–250. doi: 10.1007/BF03161207. PMC 3550116. PMID 19031375.
- ^ Roth BL, Driscol J. "PDSP Ki Database". Psychoactive Drug Screening Program (PDSP). University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Retrieved 7 May 2022.
- ^ a b c d e Tatsumi M, Groshan K, Blakely RD, Richelson E (1997). "Pharmacological profile of antidepressants and related compounds at human monoamine transporters". Eur. J. Pharmacol. 340 (2–3): 249–58. doi: 10.1016/s0014-2999(97)01393-9. PMID 9537821.
- ^ a b c d e f g h i j k l m n o p q r s t u v Haenisch B, Hiemke C, Bönisch H (2011). "Inhibitory potencies of trimipramine and its main metabolites at human monoamine and organic cation transporters". Psychopharmacology. 217 (2): 289–95. doi: 10.1007/s00213-011-2281-9. PMID 21484238. S2CID 28707541.
- ^ a b c Wander TJ, Nelson A, Okazaki H, Richelson E (1986). "Antagonism by antidepressants of serotonin S1 and S2 receptors of normal human brain in vitro". Eur. J. Pharmacol. 132 (2–3): 115–21. doi: 10.1016/0014-2999(86)90596-0. PMID 3816971.
- ^ a b c d e f g h i Gross G, Xin X, Gastpar M (1991). "Trimipramine: pharmacological reevaluation and comparison with clozapine". Neuropharmacology. 30 (11): 1159–66. doi: 10.1016/0028-3908(91)90160-d. PMID 1663593. S2CID 38254883.
- ^ a b Cusack B, Nelson A, Richelson E (1994). "Binding of antidepressants to human brain receptors: focus on newer generation compounds". Psychopharmacology. 114 (4): 559–65. doi: 10.1007/bf02244985. PMID 7855217. S2CID 21236268.
- ^ a b c d Appl H, Holzammer T, Dove S, Haen E, Strasser A, Seifert R (2012). "Interactions of recombinant human histamine H1R, H2R, H3R, and H4R receptors with 34 antidepressants and antipsychotics". Naunyn-Schmiedeberg's Arch. Pharmacol. 385 (2): 145–70. doi: 10.1007/s00210-011-0704-0. PMID 22033803. S2CID 14274150.
- ^ Kanba S, Richelson E (1984). "Histamine H1 receptors in human brain labelled with [3H]doxepin". Brain Res. 304 (1): 1–7. doi: 10.1016/0006-8993(84)90856-4. PMID 6146381. S2CID 45303586.
- ^ Randrup A, Braestrup C (1977). "Uptake inhibition of biogenic amines by newer antidepressant drugs: relevance to the dopamine hypothesis of depression". Psychopharmacology. 53 (3): 309–14. doi: 10.1007/bf00492370. PMID 408861. S2CID 8183053.
- ^ a b Richelson E, Pfenning M (1984). "Blockade by antidepressants and related compounds of biogenic amine uptake into rat brain synaptosomes: most antidepressants selectively block norepinephrine uptake". Eur. J. Pharmacol. 104 (3–4): 277–86. doi: 10.1016/0014-2999(84)90403-5. PMID 6499924.
- ^ a b c López-Muñoz F, Alamo C (2013). "Active metabolites as antidepressant drugs: the role of norquetiapine in the mechanism of action of quetiapine in the treatment of mood disorders". Front Psychiatry. 4: 102. doi: 10.3389/fpsyt.2013.00102. PMC 3770982. PMID 24062697.
- ^ Anttila SA, Leinonen EV (2001). "A review of the pharmacological and clinical profile of mirtazapine". CNS Drug Rev. 7 (3): 249–64. doi: 10.1111/j.1527-3458.2001.tb00198.x. PMC 6494141. PMID 11607047.
- ^ Celada P, Puig M, Amargós-Bosch M, Adell A, Artigas F (2004). "The therapeutic role of 5-HT1A and 5-HT2A receptors in depression". J Psychiatry Neurosci. 29 (4): 252–65. PMC 446220. PMID 15309042.
- ^ Millan MJ (2005). "Serotonin 5-HT2C receptors as a target for the treatment of depressive and anxious states: focus on novel therapeutic strategies". Thérapie. 60 (5): 441–60. doi: 10.2515/therapie:2005065. PMID 16433010.
- ^ a b Gillman PK (2007). "Tricyclic antidepressant pharmacology and therapeutic drug interactions updated". Br. J. Pharmacol. 151 (6): 737–48. doi: 10.1038/sj.bjp.0707253. PMC 2014120. PMID 17471183.
- ^ a b Kanba S, Richelson E (22 February 1991). "Interactions with Pyschotropic Drugs". In Watanabe T, Wada H (eds.). Histaminergic Neurons. CRC Press. pp. 272–. ISBN 978-0-8493-6425-9.
- ^ Extein I, Pottash AL, Gold MS, Goggans RC, Lydiard RB (2 July 1984). "Antidepressants: Predicting Response/Maximinizing Efficacy". In Gold MS, Carman JS, Lydiard RB (eds.). Advances in Psychopharmacology. CRC Press. pp. 98–. ISBN 978-0-8493-5680-3.
- ^ Baldessarini RJ (1985). Chemotherapy in Psychiatry: Principles and Practice. Harvard University Press. pp. 158–. ISBN 978-0-674-11383-1.
- ^ Krystal AD, Richelson E, Roth T (2013). "Review of the histamine system and the clinical effects of H1 antagonists: basis for a new model for understanding the effects of insomnia medications". Sleep Med Rev. 17 (4): 263–72. doi: 10.1016/j.smrv.2012.08.001. PMID 23357028.
- ^ a b c d Rüther E (1989). "Depression, circadian rhythms and trimipramine". Drugs. 38 (Suppl 1): 1–3, discussion 49–50. doi: 10.2165/00003495-198900381-00003. PMID 2693050. S2CID 22636330.
- ^ Ostad Haji E, Hiemke C, Pfuhlmann B (2012). "Therapeutic drug monitoring for antidepressant drug treatment". Curr. Pharm. Des. 18 (36): 5818–27. doi: 10.2174/138161212803523699. PMID 22681162.
- ^ a b Eap CB, Bender S, Gastpar M, et al. (2000). "Steady state plasma levels of the enantiomers of trimipramine and of its metabolites in CYP2D6-, CYP2C19- and CYP3A4/5-phenotyped patients". Ther Drug Monit. 22 (2): 209–14. doi: 10.1097/00007691-200004000-00012. PMID 10774635.
- ^ a b c Ritsner MS (15 February 2013). "Appendix 2: List of Pyschotropic Medications". Polypharmacy in Psychiatry Practice, Volume I: Multiple Medication Use Strategies. Springer Science & Business Media. pp. 270–271. ISBN 978-94-007-5805-6.
- ^ Lemke TL, Williams DA (24 January 2012). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 606–. ISBN 978-1-60913-345-0.
-
^
Acta Pharmacologica Et Toxicologica: Supplement. Munksgaard. 1985.
Triraipramine, butriptylin, and propizepine have a methyl substituent at the 2'-position of the sidechain, e.g. trimipramine is a 2'-methyl-imipramine.
- ^ Dawkins K, Manji HK, Potter WZ (20 September 1994). "Pharmacodynamics of Antidepressants". In Cutler NR, Sramek JJ, Narang PK (eds.). Pharmacodynamics and Drug Development: Perspectives in Clinical Pharmacology. John Wiley & Sons. pp. 160–. ISBN 978-0-471-95052-3.
- ^ Baumann P, Hiemke C (23 February 2012). "Central Nervous System Drugs". In Anzenbacher P, Zanger UM (eds.). Metabolism of Drugs and Other Xenobiotics. John Wiley & Sons. pp. 302–. ISBN 978-3-527-64632-6.
- ^ Anthony PK, Powers CA (2002). "Drugs that Affect the Central Neverous System". In Anthony PK (ed.). Pharmacology Secrets. Elsevier Health Sciences. pp. 39–. ISBN 978-1-56053-470-9.
- ^ Cowen P, Harrison P, Burns T (9 August 2012). "Drugs and Other Physical Treatments". Shorter Oxford Textbook of Psychiatry. OUP Oxford. pp. 532–. ISBN 978-0-19-162675-3.
- ^ a b c d Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 1251–. ISBN 978-1-4757-2085-3.
- ^ a b c d e f Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. p. 1067. ISBN 978-3-88763-075-1.
- ^ a b c Andersen J, Kristensen AS, Bang-Andersen B, Strømgaard K (2009). "Recent advances in the understanding of the interaction of antidepressant drugs with serotonin and norepinephrine transporters". Chem. Commun. (25): 3677–92. doi: 10.1039/b903035m. PMID 19557250.
- ^ Dawson AH (2004). "Cyclic Antidepressant Drugs". In Dart RC (ed.). Medical Toxicology. Lippincott Williams & Wilkins. pp. 836–. ISBN 978-0-7817-2845-4.
- ^ Gelenberg AJ, Schoonover SC (29 June 2013). "Major psychiatric disorders: depression". In Gelenberg AJ, Bassuk EL, Schoonover SC (eds.). The Practitioner's Guide to Psychoactive Drugs. Springer Science & Business Media. pp. 38–. ISBN 978-1-4757-1137-0.
- ^ Moyer TP, Boeckx RL (1 January 1982). Applied Therapeutic Drug Monitoring: Fundamentals. American Association for Clinical Chemistry. p. 249. ISBN 978-0-915274-23-9.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 281–. ISBN 978-94-011-4439-1.
- ^ a b c d "Trimipramine Uses, Side Effects & Warnings".
- ^ "TRIMIPRAMINE – Australian Medicines Handbook". www.mps-aust.com.au. MPS Australia. 2008. Archived from the original on 4 August 2017. Retrieved 4 August 2017.
- ^ "Tricyclic antidepressants - Australian Medicines Handbook". Australian Medicines Handbook. Australian Medicines Handbook Pty Ltd. Retrieved 4 August 2017.
- ^ "IMDB Quotes page". IMDB. IMDB. Retrieved 28 March 2024.