 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Tasigna, others |
| Other names | AMN107 |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a608002 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 30% [3] |
| Protein binding | 98% [3] |
| Metabolism | Liver (mostly CYP3A4-mediated) [3] |
| Elimination half-life | 15-17 hours [3] |
| Excretion | Faeces (93%) [3] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.166.395 |
| Chemical and physical data | |
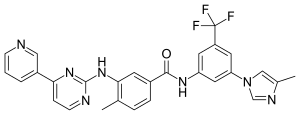
| Formula | C28H22F3N7O |
| Molar mass | 529.527 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Nilotinib, sold under the brand name Tasigna marketed worldwide by Novartis, is a medication used to treat chronic myelogenous leukemia (CML) which has the Philadelphia chromosome. [4] It may be used both in initial cases of chronic phase CML as well as in accelerated and chronic phase CML that has not responded to imatinib. [4] [5] It is taken by mouth. [5]
Common side effects may include low platelets, low white blood cells, anemia, rashes, vomiting, diarrhea, and joint pains. [5] Other serious side effects may include QT prolongation, sudden death, pancreatitis, and liver problems. [5] It is not safe for use during pregnancy. [5] Nilotinib is a Bcr-Abl tyrosine kinase inhibitor and works by interfering with signalling within the cancer cell. [5]
Nilotinib was approved for medical use in the United States in 2007. [5] It is on the World Health Organization's List of Essential Medicines. [6] It is approved as a generic medication. [7]
Medical uses
Nilotinib is used to treat Philadelphia chromosome (Ph+)-positive chronic myelogenous leukaemia. [3]
Adverse effects
Nilotinib has a number of adverse effects including headache, fatigue, gastrointestinal problems such as nausea, vomiting, diarrhea and constipation, muscle and joint pain, rash and other skin conditions, flu-like symptoms, and reduced blood cell count. Less typical side effects are those of the cardiovascular system, such as high blood pressure, various types of arrhythmia, and prolonged QT interval. Nilotinib can also affect the body's electrolyte and glucose balance. [8] Though lung-related adverse effects are rare when compared with imatinib and dasatinib, there is a case report of acute respiratory failure from diffuse alveolar hemorrhage in a people taking nilotinib. [9]
Nilotinib carries a black box warning in the United States for possible heart complications. [10] [2] Contraindications include long QT syndrome, hypokalaemia, hypomagnesaemia, pregnancy, planned pregnancy, lactation and galactose/ lactose intolerance. [3] [11]
Cautions include: [3]
- Myelosuppression
- Tumour lysis syndrome
- Liver impairment
- History of pancreatitis
- Check serum lipase periodically in order to detect pancreatitis
- Total gastrectomy
- Avoid pregnancy or impregnating women
Dose reduction has been recommended in people with liver problems which involves recommendation of lower starting dose and monitoring of any hepatic function abnormalities. [12]
Hepatitis B virus reactivation may also occur. [13]
Interactions
Nilotinib has been reported as a substrate for OATP1B1 and OATP1B3. Interaction of nilotinib with OATP1B1 and OATP1B3 may alter its hepatic disposition and can lead to transporter mediated drug-drug interactions. [12] Nilotinib is an inhibitor of OATP-1B1 transporter but not for OATP-1B3. [14]
It is a substrate for CYP3A4 and hence grapefruit juice and other CYP3A4 inhibitors [15] will increase its action and inducers like St. John's wort [16] will decrease it. Patients report that pomegranates and starfruit may also interfere.
Pharmacology

Nilotinib inhibits the kinases BCR-ABL, [17] KIT, LCK, EPHA3, EPHA8, DDR1, DDR2, PDGFRB, MAPK11 and ZAK. [18]
Structurally related to imatinib, [19] It is 10–30 fold more potent than imatinib in inhibiting Bcr-Abl tyrosine kinase activity and proliferation of Bcr-Abl expressing cells. [19] [20] [21] [22]
History
Nilotinib was developed by Novartis. [5] It was developed based on the structure of the Abl-imatinib complex to address imatinib intolerance and resistance. [23] [20] [21]
Society and culture
Legal status
It was approved for medical use by the FDA in October 2007, [8] EMA in September 2009, [24] MHRA in November 2007, [25] and TGA in January 2008. [11]
Research
Parkinson's disease
There is weak evidence that nilotinib may be beneficial with Parkinson's disease (PD), with a small clinical trial suggesting it might halt progression and improve symptoms. [26] However, there were significant side effects including infection, liver function tests abnormalities, hallucinations and heart attack, and the benefit in PD disappeared at follow up after drug discontinuation, raising question as to whether it was truly a disease modifying therapy. Nilotinib is currently undergoing phase II studies for treatment of Parkinson's. [27] Scientists and medical professionals have advised caution with over-optimistic interpretation of its effects in Parkinson's due to the significant media hype surrounding the small and early clinical trial. [28] [29] Dystonia and cognitive impairment have also been reported as side effects. [30]
Other
Novartis announced in April 2011, that it was discontinuing a phase III trial of nilotinib as the first-line treatment of gastrointestinal stromal tumor (GIST) based on the recommendation of an independent data monitoring committee. Interim results showed Tasigna is unlikely to demonstrate superiority compared to Novartis's Gleevec (imatinib)*, the current standard of care in this setting. [31]
Low dose nilotinib is also being investigated for use in Alzheimer's disease, as well as for ALS, dementia and Huntington's disease. [32]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved October 22, 2023.
- ^ a b "Tasigna- nilotinib capsule". DailyMed. February 8, 2024. Retrieved March 9, 2024.
- ^ a b c d e f g h "Tasigna (nilotinib) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved January 25, 2014.
- ^ a b "Nilotinib". National Cancer Institute. February 1, 2008. Retrieved November 14, 2019.
- ^ a b c d e f g h "Nilotinib Monograph for Professionals". Drugs.com. Retrieved November 14, 2019.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl: 10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "First-Time Generic Drug Approvals 2024". U.S. Food and Drug Administration (FDA). March 8, 2024. Retrieved March 9, 2024.
- ^ a b "Complete Nilotinib information from Drugs.com". Drugs.com. Retrieved January 25, 2014.
- ^ Donatelli C, Chongnarungsin D, Ashton R (October 2014). "Acute respiratory failure from nilotinib-associated diffuse alveolar hemorrhage". Leukemia & Lymphoma. 55 (10): 2408–2409. doi: 10.3109/10428194.2014.887714. PMID 24467220. S2CID 43118790.
- ^ "FDA Approves Tasigna for Treatment of Philadelphia Chromosome Positive Chronic Myeloid Leukemia". U.S. Food and Drug Administration. October 30, 2007. Retrieved August 4, 2009.
- ^ a b "Tasigna nilotinib" (PDF). TGA eBusiness Services. October 21, 2013. Retrieved January 25, 2014.
- ^ a b Khurana V, Minocha M, Pal D, Mitra AK (March 2014). "Role of OATP-1B1 and/or OATP-1B3 in hepatic disposition of tyrosine kinase inhibitors". Drug Metabolism and Drug Interactions. 29 (3): 179–190. doi: 10.1515/dmdi-2013-0062. PMC 4407685. PMID 24643910.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. p. 960. ISBN 978-0-85711-338-2.
- ^ Khurana V, Minocha M, Pal D, Mitra AK (May 2014). "Inhibition of OATP-1B1 and OATP-1B3 by tyrosine kinase inhibitors". Drug Metabolism and Drug Interactions. 29 (4): 249–259. doi: 10.1515/dmdi-2014-0014. PMC 4407688. PMID 24807167.
- ^ Bailey DG, Malcolm J, Arnold O, Spence JD (August 1998). "Grapefruit juice-drug interactions". British Journal of Clinical Pharmacology. 46 (2): 101–110. doi: 10.1046/j.1365-2125.1998.00764.x. PMC 1873672. PMID 9723817.
- ^ Komoroski BJ, Zhang S, Cai H, Hutzler JM, Frye R, Tracy TS, et al. (May 2004). "Induction and inhibition of cytochromes P450 by the St. John's wort constituent hyperforin in human hepatocyte cultures". Drug Metabolism and Disposition. 32 (5): 512–518. doi: 10.1124/dmd.32.5.512. PMID 15100173.
- ^ Weisberg E, Manley P, Mestan J, Cowan-Jacob S, Ray A, Griffin JD (June 2006). "AMN107 (nilotinib): a novel and selective inhibitor of BCR-ABL". British Journal of Cancer. 94 (12): 1765–1769. doi: 10.1038/sj.bjc.6603170. PMC 2361347. PMID 16721371.
- ^ Manley PW, Drueckes P, Fendrich G, Furet P, Liebetanz J, Martiny-Baron G, et al. (March 2010). "Extended kinase profile and properties of the protein kinase inhibitor nilotinib". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1804 (3): 445–453. doi: 10.1016/j.bbapap.2009.11.008. PMID 19922818.
- ^ a b Manley PW, Cowan-Jacob SW, Mestan J (December 2005). "Advances in the structural biology, design and clinical development of Bcr-Abl kinase inhibitors for the treatment of chronic myeloid leukaemia". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1754 (1–2): 3–13. doi: 10.1016/j.bbapap.2005.07.040. PMID 16172030.
- ^ a b Jabbour E, Cortes J, Kantarjian H (June 2010). "Nilotinib for the treatment of chronic myeloid leukemia: An evidence-based review". Core Evidence. 4: 207–213. doi: 10.2147/CE.S6003. PMC 2899790. PMID 20694077.
- ^ a b Olivieri A, Manzione L (June 2007). "Dasatinib: a new step in molecular target therapy". Annals of Oncology. 18 (Suppl 6): vi42–vi46. doi: 10.1093/annonc/mdm223. PMID 17591830.
- ^ Breccia M, Alimena G (February 2010). "Nilotinib: a second-generation tyrosine kinase inhibitor for chronic myeloid leukemia". Leukemia Research. 34 (2): 129–134. doi: 10.1016/j.leukres.2009.08.031. PMID 19783301.
- ^ Manley PW, Stiefl N, Cowan-Jacob SW, Kaufman S, Mestan J, Wartmann M, et al. (October 2010). "Structural resemblances and comparisons of the relative pharmacological properties of imatinib and nilotinib". Bioorganic & Medicinal Chemistry. 18 (19): 6977–6986. doi: 10.1016/j.bmc.2010.08.026. PMID 20817538.
- ^ "Tasigna : EPAR - Product Information" (PDF). European Medicines Agency. Novartis Europharm Ltd. October 18, 2013. Retrieved January 25, 2014.
- ^ "Tasigna 150mg Hard Capsules - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Novartis Pharmaceuticals UK Ltd. September 9, 2013. Retrieved January 25, 2014.
- ^ Pagan F, Hebron M, Valadez EH, Torres-Yaghi Y, Huang X, Mills RR, et al. (July 2016). "Nilotinib Effects in Parkinson's disease and Dementia with Lewy bodies". Journal of Parkinson's Disease. 6 (3): 503–517. doi: 10.3233/JPD-160867. PMC 5008228. PMID 27434297.
- ^ Dash D, Goyal V (2019). "Anticancer Drugs for Parkinson's Disease: Is It a Ray of Hope or Only Hype?". Annals of Indian Academy of Neurology. 22 (1): 13–16. doi: 10.4103/aian.AIAN_177_18. PMC 6327695. PMID 30692753.
- ^ Robledo I, Jankovic J (September 2017). "Media hype: Patient and scientific perspectives on misleading medical news". Movement Disorders. 32 (9): 1319–1323. doi: 10.1002/mds.26993. PMID 28370445. S2CID 30022509.
- ^ Wyse RK, Brundin P, Sherer TB (July 2016). "Nilotinib - Differentiating the Hope from the Hype". Journal of Parkinson's Disease. 6 (3): 519–522. doi: 10.3233/JPD-160904. PMC 5044778. PMID 27434298.
- ^ Dash D, Goyal V (2019). "Anticancer Drugs for Parkinson's Disease: Is It a Ray of Hope or Only Hype?". Annals of Indian Academy of Neurology. 22 (1): 13–16. doi: 10.4103/aian.AIAN_177_18. PMC 6327695. PMID 30692753.
- ^ "Global Novartis News Archive". Archived from the original on February 22, 2014. Retrieved February 18, 2014.
- ^ "Cancer drug prevents build-up of toxic brain protein". MedicalXpress.com. May 10, 2013. Retrieved April 11, 2017.