From Wikipedia, the free encyclopedia
Cancelled developmental cancer drug
Rociletinib
 |
|
|
Trade names | Xegafri |
|---|
| Other names | CO-1686, AVL-301 |
|---|
Routes of
administration |
By mouth |
|---|
|
ATC code | |
|---|
|
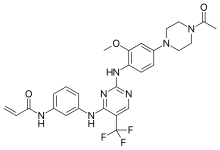
N-(3-{[2-{[4-(4-Acetyl-1-piperazinyl)-2-methoxyphenyl]amino}-5-(trifluoromethyl)-4-pyrimidinyl]amino}phenyl)acrylamide
|
|
CAS Number | |
|---|
|
PubChem
CID | |
|---|
|
DrugBank | |
|---|
|
ChemSpider | |
|---|
|
UNII | |
|---|
|
KEGG | |
|---|
|
|
Formula | C27H28F3N7O3 |
|---|
|
Molar mass | 555.562 g·mol−1 |
|---|
| 3D model (
JSmol) | |
|---|
CC(=O)N1CCN(CC1)c2ccc(c(c2)OC)Nc3ncc(c(n3)Nc4cccc(c4)NC(=O)C=C)C(F)(F)F
|
InChI=1S/C27H28F3N7O3/c1-4-24(39)32-18-6-5-7-19(14-18)33-25-21(27(28,29)30)16-31-26(35-25)34-22-9-8-20(15-23(22)40-3)37-12-10-36(11-13-37)17(2)38/h4-9,14-16H,1,10-13H2,2-3H3,(H,32,39)(H2,31,33,34,35) Key:HUFOZJXAKZVRNJ-UHFFFAOYSA-N
|
Rociletinib is a medication developed to treat
non-small cell lung carcinomas with a specific mutation. It is a third-generation
epidermal growth factor receptor
tyrosine kinase inhibitor.
[1] It was being
developed by
Clovis Oncology as a potential treatment for
non-small-cell lung cancer.
[1] In May 2016, development of rociletinib was halted, along with its associated clinical trials, and Clovis Oncology withdrew its
marketing authorisation application from the
European Medicines Agency.
[1]
References
|
|---|
|
CI
monoclonal antibodies ("-mab") | |
|---|
|
Tyrosine kinase inhibitors ("-nib") | |
Receptor tyrosine kinase |
-
RET inhibitors:
Entrectinib (ALK, ROS1, NTRK),
Futibatinib (FGFR2),
Infigratinib,
Larotrectinib (NTRK),
Pemigatinib (FGFR),
Pralsetinib,
Repotrectinib (ROS1, TRK, ALK),
Selpercatinib (VEGFR, FGFR),
Vandetanib (VEGFR, EGFR).
|
|---|
|
Non-receptor | |
|---|
|
|---|
| Other | |
|---|
|
|---|
|
Angiopoietin | |
|---|
|
CNTF | |
|---|
|
EGF (ErbB) | |
|---|
|
FGF | |
FGFR1 | |
|---|
|
FGFR2 |
- Agonists:
Ersofermin
-
FGF (
1,
2 (bFGF),
3,
4,
5,
6,
7 (
KGF),
8,
9,
10 (KGF2),
17,
18,
22)
-
Palifermin
-
Repifermin
-
Selpercatinib
-
Sprifermin
-
Trafermin
|
|---|
|
FGFR3 | |
|---|
|
FGFR4 | |
|---|
| Unsorted | |
|---|
|
|---|
|
HGF (c-Met) | |
|---|
|
IGF | |
|---|
|
LNGF (p75NTR) | |
|---|
|
PDGF | |
|---|
|
RET (GFL) | |
|---|
|
SCF (c-Kit) | |
|---|
|
TGFβ | |
|---|
|
Trk | |
TrkA |
- Negative allosteric modulators:
VM-902A
|
|---|
|
TrkB |
- Agonists:
3,7-DHF
-
3,7,8,2'-THF
-
4'-DMA-7,8-DHF
-
7,3'-DHF
-
7,8-DHF
-
7,8,2'-THF
-
7,8,3'-THF
-
Amitriptyline
-
BDNF
-
BNN-20
-
Deoxygedunin
-
Deprenyl
-
Diosmetin
-
DMAQ-B1
-
HIOC
-
LM22A-4
-
N-Acetylserotonin
-
NT-3
-
NT-4
-
Norwogonin (5,7,8-THF)
-
R7
-
R13
-
TDP6
|
|---|
|
TrkC | |
|---|
|
|---|
|
VEGF | |
|---|
| Others |
- Additional growth factors:
Adrenomedullin
-
Colony-stimulating factors (see
here instead)
-
Connective tissue growth factor (CTGF)
-
Ephrins (
A1,
A2,
A3,
A4,
A5,
B1,
B2,
B3)
-
Erythropoietin (see
here instead)
-
Glucose-6-phosphate isomerase (GPI; PGI, PHI, AMF)
-
Glia maturation factor (GMF)
-
Hepatoma-derived growth factor (HDGF)
-
Interleukins/
T-cell growth factors (see
here instead)
-
Leukemia inhibitory factor (LIF)
-
Macrophage-stimulating protein (MSP; HLP, HGFLP)
-
Midkine (NEGF2)
-
Migration-stimulating factor (MSF; PRG4)
-
Oncomodulin
-
Pituitary adenylate cyclase-activating peptide (PACAP)
-
Pleiotrophin
-
Renalase
-
Thrombopoietin (see
here instead)
-
Wnt signaling proteins
- Additional growth factor receptor modulators:
Cerebrolysin (neurotrophin mixture)
|
|---|
