| |
| Names | |
|---|---|
|
IUPAC name
tetraiodoplatinum
| |
| Other names
Platinum tetraiodide, platinic iodide, platinum(4+) tetraiodide
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ECHA InfoCard | 100.029.280 |
| EC Number |
|
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| I4Pt | |
| Molar mass | 702.702 g·mol−1 |
| Appearance | brown crystals |
| Density | 6.06 g/cm3 |
| Melting point | 130 °C (266 °F; 403 K) |
| decomposes in water | |
| Related compounds | |
Related compounds
|
Iridium tetraiodide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Platinum(IV) iodide is a inorganic compound with the formula PtI4. [1] A dark brown diamagnetic solid and is one of several binary iodides of platinum.
Preparation
Platinum(IV) iodide can be prepared from the effect of iodine on platinum: [2]
- Pt + 2I2 → PtI4
It can also be obtained from the decomposition of hydrogen hexaiodoplatinate(IV) at 80 °C:
- H2[PtI6] → PtI4 + 2HI
Physical properties
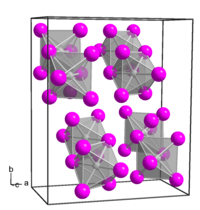
Platinum(IV) iodide forms dark brown crystals of several modifications: [3]
- α-PtI4, rhombic crystal system, spatial group P bca, [4] cell parameters a = 1.290 nm, b = 1.564 nm, c = 0.690 nm, Z = 8;
- β-PtI4, cubic crystal system, spatial group P m3m, cell parameters a = 0.56 nm, Z = 1;
- γ-PtI4, tetragonal crystal system, spatial group I 41/a, cell parameters a = 0.677 nm, c = 3.110 nm, Z = 8.
Platinum(IV) iodide decomposes in water. It is also soluble in ethanol, acetone, alkali, HI, KI, liquid NH3. [5]
Chemical properties
It decomposes when heated:
- PtI4 → Pt + 2I2
When dissolved in hydroiodic acid, platinum(IV) iodide forms hydrogen hexaiodoplatinate(IV):
- PtI4 + 2HI → H2[PtI6
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ^ Wicks, Charles E.; Block, Frank E. (1963). Thermodynamic Properties of 65 Elements: Their Oxides, Halides, Carbides and Nitrides. U.S. Government Printing Office. p. 92. Retrieved 28 March 2024.
- ^ Macintyre, Jane E. (23 July 1992). Dictionary of Inorganic Compounds. CRC Press. p. 3510. ISBN 978-0-412-30120-9. Retrieved 28 March 2024.
- ^ Donnay, Joseph Désiré Hubert (1978). Crystal Data: Inorganic compounds 1967-1969. National Bureau of Standards. p. 153. Retrieved 28 March 2024.
- ^ "Platinum(IV) iodide, 99.95% (Metals basis), Pt 27.3% min., Thermo Scientific Chemicals, Premion | Fisher Scientific". Fisher Scientific. Retrieved 28 March 2024.