
| |
| Names | |
|---|---|
|
IUPAC name
Neptunium(IV) fluoride
| |
| Other names
Neptunium tetrafluoride
| |
| Identifiers | |
3D model (
JSmol)
|
|
PubChem
CID
|
|
| |
| |
| Properties | |
| NpF4 | |
| Molar mass | 313 g/mol |
| Appearance | Green solid [1] |
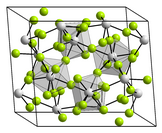
| Structure | |
| Monoclinic, mS60 [1] | |
| C2/c, No. 15 [2] | |
a = 1.27 nm, b = 1.0082 nm, c = 0.833 nm α = 90°, β = 126.03°, γ = 90°
| |
Lattice volume (V)
|
0.86256 nm3 |
Formula units (Z)
|
12 |
| Thermochemistry | |
Heat capacity (C)
|
116 ± 4 J/mol·K [1] |
Std molar
entropy (S⦵298) |
148 ± 3 J/mol·K [1] |
Std enthalpy of
formation (ΔfH⦵298) |
−1874 ± 16 kJ/mol [1] |
Gibbs free energy (ΔfG⦵)
|
-1783 ± 16 kJ/mol [1] |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Neptunium(IV) fluoride or neptunium tetrafluoride is a inorganic compound with the formula NpF4. It is a green salt and is isostructural with UF4. [3]
Synthesis
Neptunium(IV) fluoride can be prepared by reacting neptunium(III) fluoride or neptunium dioxide with a gas mixture of oxygen and hydrogen fluoride at 500 °C: [1]
It can also be prepared by treating neptunium dioxide with HF gas: [1]
References
- ^ a b c d e f g h Haire, Richard G. (2006). "Neptunium". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. pp. 730–736. doi: 10.1007/1-4020-3598-5_9. ISBN 1-4020-3555-1.
- ^ Zachariasen, W. H. (1949). "Crystal chemical studies of the 5f-series of elements. XII. New compounds representing known structure types". Acta Crystallographica. 2 (6): 388–390. doi: 10.1107/S0365110X49001016.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.

