| |
| Names | |
|---|---|
| Other names
neptunium(IV) bromide
| |
| Identifiers | |
| Properties | |
| Br4Np | |
| Molar mass | 557 g·mol−1 |
| Appearance | red-brown crystals |
| Density | 5.5 g/cm3 |
| Melting point | 464 °C (867 °F; 737 K) |
| Related compounds | |
Related compounds
|
Uranium tetrabromide, Thorium tetrabromide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Neptunium tetrabromide is a binary inorganic compound of neptunium metal and bromine with the chemical formula NpBr4. [1] [2] [3] [4]
Synthesis
The compound can be prepared from the reaction of bromine with metallic neptunium:
- Np + 2Br2 → NpBr4
Also, a reaction of aluminum bromide with neptunium(IV) oxide: [5] [6]
- 3NpO2 + 4AlBr3 → 3NpBr4 + 2Al2O3
Physical properties
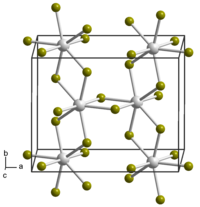
Neptunium tetrabromide forms red-brown hygroscopic crystals of monoclinic crystal system, spatial group P 2/c, [7] [8] cell parameters a = 1.089 nm, b = 0.874 nm, c = 0.705 nm, β = 95.19°, Z = 4. [9]
NpBr4 is easily purified by sublimation in a vacuum. [10]
Chemical properties
Decomposes under strong heating to form neptunium tribromide:
- 2NpBr4 → 2NpBr3 + Br2
With careful oxidation with antimony(III) oxide, the compound forms neptunium oxidibromide:
- NpBr4 + Sb2O3 → NpOBr2 + 2SbOBr
References
- ^ "WebElements Periodic Table » Neptunium » neptunium tetrabromide". webelements.com. Retrieved 2 April 2024.
- ^ Stevens, J. L.; Jones, E. R.; Karraker, D. G. (15 February 1976). "Mössbauer spectra and magnetic susceptibility of neptunium tetrabromide". The Journal of Chemical Physics. 64 (4): 1492–1494. doi: 10.1063/1.432366. Retrieved 2 April 2024.
- ^ Brown, D.; Hill, J.; Rickard, C. E. F. (1 January 1970). "Preparation of actinide bromides and bromo-complexes by use of liquid boron tribromide or liquid bromine". Journal of the Chemical Society A: Inorganic, Physical, Theoretical (0): 476–480. doi: 10.1039/J19700000476. ISSN 0022-4944. Retrieved 2 April 2024.
- ^ Yaws, Carl L. (6 January 2015). The Yaws Handbook of Physical Properties for Hydrocarbons and Chemicals: Physical Properties for More Than 54,000 Organic and Inorganic Chemical Compounds, Coverage for C1 to C100 Organics and Ac to Zr Inorganics. Gulf Professional Publishing. p. 740. ISBN 978-0-12-801146-1. Retrieved 2 April 2024.
- ^ Fried, S. (1947). The Basic Dry Chemistry of Neptunium. Atomic Energy Commission. p. 10. Retrieved 2 April 2024.
- ^ Abstracts of Declassified Documents. Technical Information Division, Oak Ridge Directed Operations. 1947. p. 740. Retrieved 2 April 2024.
- ^ Morss, L. R.; Edelstein, Norman M.; Fuger, Jean (21 October 2010). The Chemistry of the Actinide and Transactinide Elements (Set Vol.1-6): Volumes 1-6. Springer Science & Business Media. p. 2416. ISBN 978-94-007-0211-0. Retrieved 2 April 2024.
- ^ Kirk-Othmer Concise Encyclopedia of Chemical Technology, 2 Volume Set. John Wiley & Sons. 16 July 2007. p. 47. ISBN 978-0-470-04748-4. Retrieved 2 April 2024.
- ^ The Chemical Thermodynamics of Actinide Elements and Compounds: The actinide halides. International Atomic Energy Agency. 1983. p. 162. Retrieved 2 April 2024.
- ^ Satya, Prakash (2013). Advanced Chemistry of Rare Elements. S. Chand Publishing. p. 684. ISBN 978-81-219-4254-6. Retrieved 2 April 2024.