| |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /lɪˈnɛzəlɪd, -ˈneɪz-/ |
| Trade names | Zyvox, Zyvoxam, others |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a602004 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | Intravenous infusion, by mouth |
| Drug class | Oxazolidinone antibiotic |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~100% (oral) |
| Protein binding | Low (31%) |
| Metabolism | Liver (50–70%, CYP not involved) |
| Elimination half-life | 3–7 hours; [9] longer half-life in CSF than plasma [9] |
| Excretion | non-kidney, kidney, and fecal [10] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| PDB ligand | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.121.520 |
| Chemical and physical data | |
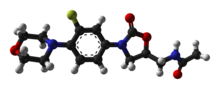
| Formula | C16H20FN3O4 |
| Molar mass | 337.351 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| (verify) | |
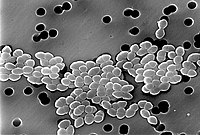
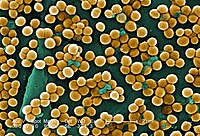
Linezolid is an antibiotic used for the treatment of infections caused by Gram-positive bacteria that are resistant to other antibiotics. [9] [10] Linezolid is active against most Gram-positive bacteria that cause disease, including streptococci, vancomycin-resistant enterococci (VRE), and methicillin-resistant Staphylococcus aureus (MRSA). [9] [8] The main uses are infections of the skin and pneumonia although it may be used for a variety of other infections including drug-resistant tuberculosis. [10] [11] It is used either by injection into a vein or by mouth. [10]
When given for short periods, linezolid is a relatively safe antibiotic. [12] It can be used in people of all ages and in people with liver disease or poor kidney function. [10] Common side effects with short-term use include headache, diarrhea, rash, and nausea. [10] Serious side effects may include serotonin syndrome, bone marrow suppression, and high blood lactate levels, particularly when used for more than two weeks. [10] [13] If used for longer periods it may cause nerve damage, including optic nerve damage, which may be irreversible. [13]
As a protein synthesis inhibitor, linezolid works by suppressing bacterial protein production. [14] This either stops growth or results in bacterial death. [10] Although many antibiotics work this way, the exact mechanism of action of linezolid appears to be unique in that it blocks the initiation of protein production, rather than one of the later steps. [14] As of 2014, bacterial resistance to linezolid has remained low. [15] Linezolid is a member of the oxazolidinone class of medications. [10]
Linezolid was discovered in the mid-1990s, and was approved for commercial use in 2000. [16] [17] It is on the World Health Organization's List of Essential Medicines. [18] The World Health Organization classifies linezolid as critically important for human medicine. [19] Linezolid is available as a generic medication. [10]
Medical uses
The main use of linezolid is the treatment of severe infections caused by aerobic Gram-positive bacteria that are resistant to other antibiotics; it should not be used against bacteria that are sensitive to drugs with a narrower spectrum of activity, such as penicillins and cephalosporins. In both the popular press and the scientific literature, linezolid has been called a "reserve antibiotic"—one that should be used sparingly so that it will remain effective as a drug of last resort against potentially intractable infections. [20] [21] [22]
In the United States, the indications for linezolid use approved by the U.S. Food and Drug Administration (FDA) are the treatment of vancomycin-resistant Enterococcus faecium infections, with or without bacterial invasion of the bloodstream; nosocomial pneumonia (hospital-acquired) and community-acquired pneumonia caused by S. aureus or S. pneumoniae; complicated skin and skin structure infections (cSSSI) caused by susceptible bacteria, including diabetic foot infection, unless complicated by osteomyelitis (infection of the bone and bone marrow); and uncomplicated skin and soft tissue infections caused by S. pyogenes or S. aureus. [8] The manufacturer advises against the use of linezolid for community-acquired pneumonia or uncomplicated skin and soft tissue infections caused by MRSA. [8] In the United Kingdom, pneumonia and cSSSIs are the only indications noted in the product labeling. [7]
Linezolid appears to be as safe and effective for use in children and newborns as it is in adults. [23]
Skin and soft tissue infections
A large meta-analysis of randomized controlled trials found linezolid to be more effective than glycopeptide antibiotics (such as vancomycin and teicoplanin) and beta-lactam antibiotics in the treatment of skin and soft tissue infections (SSTIs) caused by Gram-positive bacteria, [24] and smaller studies appear to confirm its superiority over teicoplanin in the treatment of all serious Gram-positive infections. [25]
In the treatment of diabetic foot infections, linezolid appears to be cheaper and more effective than vancomycin. [26] In a 2004 open-label study, it was as effective as ampicillin/sulbactam and amoxicillin/clavulanic acid, and far superior in patients with foot ulcers and no osteomyelitis, but with significantly higher rates of adverse effects. [27] [28] A 2008 meta-analysis of 18 randomized controlled trials, however, found that linezolid treatment failed as often as other antibiotics, regardless of whether patients had osteomyelitis. [29]
Some authors have recommended that combinations of cheaper or more cost-effective drugs (such as co-trimoxazole with rifampicin or clindamycin) be tried before linezolid in the treatment of SSTIs when susceptibility of the causative organism allows it. [28] [30]
Pneumonia
No significant difference appears in treatment success rates between linezolid, glycopeptides, or appropriate beta-lactam antibiotics in the treatment of pneumonia. [24] Clinical guidelines for the treatment of community-acquired pneumonia developed by the American Thoracic Society and the Infectious Diseases Society of America recommend that linezolid be reserved for cases in which MRSA has been confirmed as the causative organism, or when MRSA infection is suspected based on the clinical presentation. [31] The guidelines of the British Thoracic Society do not recommend it as first-line treatment, but rather as an alternative to vancomycin. [32] Linezolid is also an acceptable second-line treatment for community-acquired pneumococcal pneumonia when penicillin resistance is present. [31]
U.S. guidelines recommend either linezolid or vancomycin as the first-line treatment for hospital-acquired (nosocomial) MRSA pneumonia. [33] Some studies have suggested that linezolid is better than vancomycin against nosocomial pneumonia, particularly ventilator-associated pneumonia caused by MRSA, perhaps because the penetration of linezolid into bronchial fluids is much higher than that of vancomycin. Several issues in study design have been raised, however, calling into question results that suggest the superiority of linezolid. [28] Regardless, linezolid's advantages include its high oral bioavailability—which allows easy switching to oral therapy—and the fact that poor kidney function is not an obstacle to use. [33] In contrast, achieving the correct dosage of vancomycin in patients with kidney failure is very difficult. [33]
Other
It is traditionally believed that so-called "deep" infections—such as osteomyelitis or infective endocarditis—should be treated with bactericidal antibiotics, not bacteriostatic ones. Nevertheless, preclinical studies were conducted to assess the efficacy of linezolid for these infections, [35] and the drug has been used successfully to treat them in clinical practice. Linezolid appears to be a reasonable therapeutic option for infective endocarditis caused by multi-resistant Gram-positive bacteria, despite a lack of high-quality evidence to support this use. [36] [37] Results in the treatment of enterococcal endocarditis have varied, with some cases treated successfully and others not responding to therapy. [38] [39] [40] [41] [42] [43] Low- to medium-quality evidence is also mounting for its use in bone and joint infections, including chronic osteomyelitis, although adverse effects are a significant concern when long-term use is necessary. [44] [45] [46] [47] [48] [49]
In combination with other drugs, linezolid has been used to treat tuberculosis. [50] The optimal dose for this purpose has not been established. In adults, daily and twice-daily dosing have been used to good effect. Many months of treatment are often required, and the rate of adverse effects is high regardless of dosage. [51] [52] There is not enough reliable evidence of efficacy and safety to support this indication as a routine use. [23]
Linezolid has been studied as an alternative to vancomycin in the treatment of febrile neutropenia in cancer patients when Gram-positive infection is suspected. [53] It is also one of few antibiotics that diffuse into the vitreous humor, and may therefore be effective in treating endophthalmitis (inflammation of the inner linings and cavities of the eye) caused by susceptible bacteria. Again, there is little evidence for its use in this setting, as infectious endophthalmitis is treated widely and effectively with vancomycin injected directly into the eye. [28]
Infections of the central nervous system
In animal studies of meningitis caused by Streptococcus pneumoniae, linezolid was found to penetrate well into cerebrospinal fluid, but its effectiveness was inferior to that of other antibiotics. [54] [55] There does not appear to be enough high-quality evidence to support the routine use of linezolid to treat bacterial meningitis. Nonetheless, it has been used successfully in many cases of central nervous system infection—including meningitis—caused by susceptible bacteria, and has also been suggested as a reasonable choice for this indication when treatment options are limited or when other antibiotics have failed. [56] [57] The guidelines of the Infectious Diseases Society of America recommend linezolid as the first-line drug of choice for VRE meningitis, and as an alternative to vancomycin for MRSA meningitis. [58] Linezolid appears superior to vancomycin in treating community-acquired MRSA infections of the central nervous system, although very few cases of such infections have been published (as of 2009 [update]). [59]
In March 2007, the FDA reported the results of a randomized, open-label, phase III clinical trial comparing linezolid to vancomycin in the treatment of catheter-related bloodstream infections. Patients treated with vancomycin could be switched to oxacillin or dicloxacillin if the bacteria that caused their infection was found to be susceptible, and patients in both groups (linezolid and vancomycin) could receive specific treatment against Gram-negative bacteria if necessary. [60] The study itself was published in January 2009. [61]
Linezolid was associated with significantly greater mortality than the comparator antibiotics. When data from all participants were pooled, the study found that 21.5% of those given linezolid died, compared to 16% of those not receiving it. The difference was found to be due to the inferiority of linezolid in the treatment of Gram-negative infections alone or mixed Gram-negative/Gram-positive infections. In participants whose infection was due to Gram-positive bacteria alone, linezolid was as safe and effective as vancomycin. [60] [61] In light of these results, the FDA issued an alert reminding healthcare professionals that linezolid is not approved for the treatment of catheter-related infections or infections caused by Gram-negative organisms, and that more appropriate therapy should be instituted whenever a Gram-negative infection is confirmed or suspected. [60]
Specific populations
In adults and children over the age of 12, linezolid is usually given every 12 hours, whether orally or intravenously. [54] [62] In younger children and infants, it is given every eight hours. [63] No dosage adjustments are required in the elderly, in people with mild-to-moderate liver failure, or in those with impaired kidney function. [64] In people requiring hemodialysis, care should be taken to give linezolid after a session, because dialysis removes 30–40% of a dose from the body; no dosage adjustments are needed in people undergoing continuous hemofiltration, [64] although more frequent administration may be warranted in some cases. [23] According to one study, linezolid may need to be given more frequently than normal in people with burns affecting more than 20% of body area, due to increased nonrenal clearance of the drug. [65]
Linezolid is in U.S. pregnancy category C, meaning there have been no adequate studies of its safety when used by pregnant women, and although animal studies have shown mild toxicity to the fetus, the benefits of using the drug may outweigh its risks. [8] It also passes into breast milk, although the clinical significance of this (if any) is unknown. [66]
Spectrum of activity
Linezolid is effective against all clinically important Gram-positive bacteria—those whose cell wall contains a thick layer of peptidoglycan and no outer membrane—notably Enterococcus faecium and Enterococcus faecalis (including vancomycin-resistant enterococci), Staphylococcus aureus (including methicillin-resistant Staphylococcus aureus, MRSA), Streptococcus agalactiae, Streptococcus pneumoniae, Streptococcus pyogenes, the viridans group streptococci, Listeria monocytogenes, and Corynebacterium species (the latter being among the most susceptible to linezolid, with minimum inhibitory concentrations routinely below 0.5 mg/L). [8] [54] [67] Linezolid is also highly active in vitro against several mycobacteria. [54] It appears to be very effective against Nocardia, but because of high cost and potentially serious adverse effects, authors have recommended that it be combined with other antibiotics or reserved for cases that have failed traditional treatment. [68]
Linezolid is considered bacteriostatic against most organisms—that is, it stops their growth and reproduction without actually killing them—but has some bactericidal (killing) activity against streptococci. [8] [69] Some authors have noted that, despite its bacteriostatic effect in vitro, linezolid "behaves" as a bactericidal antibiotic in vivo because it inhibits the production of toxins by staphylococci and streptococci. [35] It also has a post-antibiotic effect lasting one to four hours for most bacteria, meaning that bacterial growth is temporarily suppressed even after the drug is discontinued. [23]
Gram-negative bacteria
Linezolid has no clinically significant effect on most Gram-negative bacteria. Pseudomonas and the Enterobacteriaceae, for instance, are not susceptible. [69] In vitro, it is active against Pasteurella multocida, [8] [70] Fusobacterium, Moraxella catarrhalis, Legionella, Bordetella, and Elizabethkingia meningoseptica, and moderately active (having a minimum inhibitory concentration for 90% of strains of 8 mg/L) against Haemophilus influenzae. [66] [69] It has also been used to great effect as a second-line treatment for Capnocytophaga infections. [56] [71]
Comparable antibiotics
Linezolid's spectrum of activity against Gram-positive bacteria is similar to that of the glycopeptide antibiotic vancomycin, which has long been the standard for treatment of MRSA infections, and the two drugs are often compared. [12] [23] Other comparable antibiotics include glycopeptide antibiotics such as teicoplanin (trade name Targocid), dalbavancin (Dalvance), oritavancin (Orbactiv), and telavancin (Vibativ); quinupristin/dalfopristin (Synercid, a combination of two streptogramins, not active against E. faecalis); [72] daptomycin (Cubicin, a lipopeptide); and ceftobiprole (Zevtera, a 5th-generation cephalosporin). Linezolid is the only one that can be taken by mouth for the treatment of systemic infections. [23]
Adverse effects
When used for short periods, linezolid is a relatively safe drug. [12] Common side effects of linezolid use (those occurring in more than 1% of people taking linezolid) include diarrhea (reported by 3–11% of clinical trial participants), headache (1–11%), nausea (3–10%), vomiting (1–4%), rash (2%), constipation (2%), altered taste perception (1–2%), and discoloration of the tongue (0.2–1%). [64] It has also been known to cause thrombocytopenia. Fungal infections such as thrush and vaginal candidiasis may also occur as linezolid suppresses normal bacterial flora and opens a niche for fungi (so-called antibiotic candidiasis). [64] Less common (and potentially more serious) adverse effects include allergic reactions, pancreatitis, and elevated transaminases, which may be a sign of liver damage. [64] [73] Unlike some antibiotics, such as erythromycin and the quinolones, linezolid has no effect on the QT interval, a measure of cardiac electrical conduction. [73] [74] Adverse effects in children are similar to those that occur in adults. [74]
Like nearly all antibiotics, linezolid has been associated with Clostridium difficile-associated diarrhea (CDAD) and pseudomembranous colitis, although the latter is uncommon, occurring in about one in two thousand patients in clinical trials. [64] [73] [74] [75] C. difficile appears to be susceptible to linezolid in vitro, and linezolid was even considered as a possible treatment for CDAD. [76]
Long-term use
Bone marrow suppression, characterized particularly by thrombocytopenia (low platelet count), may occur during linezolid treatment; it appears to be the only adverse effect that occurs significantly more frequently with linezolid than with glycopeptides or beta-lactams. [24] It is uncommon in patients who receive the drug for 14 days or fewer, but occurs much more frequently in patients who receive longer courses or who have renal failure. [73] [77] A 2004 case report suggested that pyridoxine (a form of vitamin B6) could reverse the anemia and thrombocytopenia caused by linezolid, [78] but a later, larger study found no protective effect. [79]
Long-term use of linezolid has also been associated with chemotherapy-induced peripheral neuropathy, a progressive and enduring often irreversible tingling numbness, intense pain, and hypersensitivity to cold, beginning in the hands and feet and sometimes involving the arms and legs. [80] Chemotherapy drugs associated with CIPN include thalidomide, the epothilones such as ixabepilone, the vinca alkaloids vincristine and vinblastine, [81] [82] [83] the taxanes paclitaxel and docetaxel, the proteasome inhibitors such as bortezomib, and the platinum-based drugs cisplatin, oxaliplatin and carboplatin. [80] [84] [85] and optic neuropathy, which is most common after several months of treatment and may also be irreversible. [86] [87] [88] [89] [90] Although the mechanism of injury is still poorly understood, mitochondrial toxicity has been proposed as a cause; [91] [92] linezolid is toxic to mitochondria, probably because of the similarity between mitochondrial and bacterial ribosomes. [93] Lactic acidosis, a potentially life-threatening buildup of lactic acid in the body, may also occur due to mitochondrial toxicity. [91] Because of these long-term effects, the manufacturer recommends weekly complete blood counts during linezolid therapy to monitor for possible bone marrow suppression, and recommends that treatment last no more than 28 days. [8] [73] A more extensive monitoring protocol for early detection of toxicity in seriously ill patients receiving linezolid has been developed and proposed by a team of researchers in Melbourne, Australia. The protocol includes twice-weekly blood tests and liver function tests; measurement of serum lactate levels, for early detection of lactic acidosis; a review of all medications taken by the patient, interrupting the use of those that may interact with linezolid; and periodic eye and neurological exams in patients set to receive linezolid for longer than four weeks. [94]
The adverse effects of long-term linezolid therapy were first identified during postmarketing surveillance. Bone marrow suppression was not identified during Phase III trials, in which treatment did not exceed 21 days. Although some participants of early trials did experience thrombocytopenia, it was found to be reversible and did not occur significantly more frequently than in controls (participants not taking linezolid). [54] There have also been postmarketing reports of seizures, and, as of 2009 [update], a single case each of Bell's palsy (paralysis of the facial nerve) and kidney toxicity. [74] Evidence of protein synthesis inhibition in mammalian cells by linezolid has been published. [95]
Interactions
Linezolid is a weak, non-selective, reversible monoamine oxidase inhibitor (MAOI), and should not be used concomitantly with other MAOIs, large amounts of tyramine-rich foods (such as pork, aged cheeses, alcoholic beverages, or smoked and pickled foods), or serotonergic drugs. There have been postmarketing reports of serotonin syndrome when linezolid was given with or soon after the discontinuation of serotonergic drugs, particularly selective serotonin reuptake inhibitors (SSRIs) such as paroxetine and sertraline. [73] [96] [97] [98] It may also enhance the blood pressure-increasing effects of sympathomimetic drugs such as pseudoephedrine or phenylpropanolamine. [54] [99] It should also not be given in combination with pethidine ( meperidine) under any circumstance due to the risk of serotonin syndrome.
Linezolid does not inhibit or induce the cytochrome P450 (CYP) system, which is responsible for the metabolism of many commonly used drugs, and therefore does not have any CYP-related interactions. [8]
Pharmacology
Pharmacodynamics

Linezolid, like other oxazolidinones, is a bacterial protein synthesis inhibitor and a weak, non-selective, reversible monoamine oxidase inhibitor. [9] [100] As a protein synthesis inhibitor, linezolid stops the growth and reproduction of bacteria by disrupting translation of messenger RNA (mRNA) into proteins in bacterial ribosomes. [9] Linezolid inhibits translation at the first step of protein synthesis, initiation, [9] [101] unlike most other protein synthesis inhibitors, which inhibit elongation. [14] [62] It does so by preventing the formation of the initiation complex, composed of the 30S and 50S subunits of the ribosome, tRNA, and mRNA. Linezolid binds to the 23S portion of the 50S subunit (the center of peptidyl transferase activity), [9] [101] close to the binding sites of chloramphenicol, lincomycin, and other antibiotics. Due to this unique mechanism of action, cross-resistance between linezolid and other protein synthesis inhibitors is highly infrequent or nonexistent. [23] [54]
In 2008, the crystal structure of linezolid bound to the 50S subunit of a ribosome from the archaean Haloarcula marismortui was elucidated by a team of scientists from Yale University and deposited in the Protein Data Bank. [102] Another team in 2008 determined the structure of linezolid bound to a 50S subunit of Deinococcus radiodurans. The authors proposed a refined model for the mechanism of action of oxazolidinones, finding that linezolid occupies the A site of the 50S ribosomal subunit, inducing a conformational change that prevents tRNA from entering the site and ultimately forcing tRNA to separate from the ribosome. [103]
Pharmacokinetics

One of the advantages of linezolid is that it has an absolute oral bioavailability of 100% due to its rapid and complete absorption after oral administration; [9] in other words, the entire dose reaches the bloodstream, as if it had been given intravenously. [9] This means that people receiving intravenous linezolid may be switched to oral linezolid as soon as their condition allows it, whereas comparable antibiotics (such as vancomycin and quinupristin/dalfopristin) can only be given intravenously. [9] [62] Taking linezolid with food somewhat slows its absorption, but the area under the curve is not affected. [23]
Linezolid's plasma protein binding is approximately 31% (range 4–32%) and its volume of distribution at steady state averages 36.1–47.3 liters in healthy adult volunteers. [9] Peak plasma concentrations (Cmax) are reached one to two hours after administration of the drug. Linezolid is readily distributed to all tissues in the body apart from bone matrix and white adipose tissue. [35] Notably, the concentration of linezolid in the epithelial lining fluid (ELF) of the lower respiratory tract is at least equal to, and often higher than, that achieved in serum (some authors have reported bronchial fluid concentrations up to four times higher than serum concentrations), which may account for its efficacy in treating pneumonia. However, a meta-analysis of clinical trials found that linezolid was not superior to vancomycin, which achieves lower concentrations in the ELF. [104] Cerebrospinal fluid (CSF) concentrations vary; peak CSF concentrations are lower than serum ones, due to slow diffusion across the blood–brain barrier, and trough concentrations in the CSF are higher for the same reason. [23] The average half-life is three hours in children, four hours in teenagers, and five hours in adults. [8]
Linezolid is metabolized in the liver, by oxidation of the morpholine ring, without involvement of the cytochrome P450 system. This metabolic pathway leads to two major inactive metabolites (which each account for around 45% and 10% of an excreted dose at steady state), one minor metabolite, and several trace metabolites, none of which accounts for more than 1% of an excreted dose. [105] Clearance of linezolid varies with age and gender; it is fastest in children (which accounts for the shorter half-life), and appears to be 20% lower in women than in men. [8] [105] [106] There is a strong correlation between linezolid clearance and creatinine clearance. [107]
Chemistry
At physiological pH (7.4), linezolid exists in an uncharged state. It is moderately water-soluble (approximately 3 mg/mL), with a logP of 0.55. [23]
![Skeletal formula of N-{[(5S)-3-[3-fluoro-4-(morpholin-4-yl)phenyl]-2-oxo-1,3-oxazolidin-5-yl]methyl}acetamide, highlighting the morpholino and fluoro groups in orange, with the rest in blue. The carbon atoms of the parent chain are numbered.](https://upload.wikimedia.org/wikipedia/commons/thumb/e/ea/Linezolid_showing_oxazolidinone_pharmacophore.svg/220px-Linezolid_showing_oxazolidinone_pharmacophore.svg.png)
The oxazolidinone pharmacophore—the chemical "template" essential for antimicrobial activity—consists of a 1,3-oxazolidin-2-one moiety with an aryl group at position 3 and an S- methyl group, with another substituent attached to it, at position 5 (the R- enantiomers of all oxazolidinones are devoid of antibiotic properties). [108] In addition to this essential core, linezolid also contains several structural characteristics that improve its effectiveness and safety. An acetamide substituent on the 5-methyl group is the best choice in terms of antibacterial efficacy, and is used in all of the more active oxazolidinones developed thus far; in fact, straying too far from an acetamide group at this position makes the drug lose its antimicrobial power, although weak to moderate activity is maintained when some isosteric groups are used. A fluorine atom at the 3′ position practically doubles in vitro and in vivo activity, and the electron-donating nitrogen atom in the morpholine ring helps maintain high antibiotic potency and an acceptable safety profile. [35] [108]
The anticoagulant rivaroxaban (Xarelto) bears a striking structural similarity to linezolid; both drugs share the oxazolidinone pharmacophore, differing in only three areas (an extra ketone and chloro thiophene, and missing the fluorine atom). However this similarity appears to carry no clinical significance. [109]
Synthesis
Linezolid is a completely synthetic drug: it does not occur in nature (unlike erythromycin and many other antibiotics) and was not developed by building upon a naturally occurring skeleton (unlike most beta-lactams, which are semisynthetic). Many approaches are available for oxazolidinone synthesis, and several routes for the synthesis of linezolid have been reported in the chemistry literature. [108] [110] Despite good yields, the original method (developed by Upjohn for pilot plant-scale production of linezolid and eperezolid) is lengthy, requires the use of expensive chemicals—such as palladium on carbon and the highly sensitive reagents methanesulfonyl chloride and n-butyllithium—and needs low-temperature conditions. [108] [110] [111] Much of the high cost of linezolid has been attributed to the expense of its synthesis. [111] A somewhat more concise and cost-effective route better suited to large-scale production was patented by Upjohn in 1998. [35] [112]
Later syntheses have included an " atom-economical" method starting from D-mannitol, developed by Indian pharmaceutical company Dr. Reddy's and reported in 1999, [113] and a route starting from (S)-glyceraldehyde acetonide (prepared from ascorbic acid), developed by a team of researchers from Hunan Normal University in Changsha, Hunan, China. [110] On 25 June 2008, during the 12th Annual Green Chemistry and Engineering Conference in New York, Pfizer reported the development of their "second-generation" synthesis of linezolid: a convergent, green synthesis starting from (S)- epichlorohydrin, with higher yield and a 56% reduction in total waste. [114]
Resistance
Acquired resistance to linezolid was reported as early as 1999, in two patients with severe, multidrug-resistant Enterococcus faecium infection who received the drug through a compassionate use program. [69] Linezolid-resistant Staphylococcus aureus was first isolated in 2001. [115]
In the United States, resistance to linezolid has been monitored and tracked since 2004 through a program named LEADER, which (as of 2007 [update]) was conducted in 60 medical institutions throughout the country. Resistance has remained stable and extremely low—less than one-half of one percent of isolates overall, and less than one-tenth of one percent of S. aureus samples. [116] A similar, worldwide program—the "Zyvox Annual Appraisal of Potency and Spectrum Study", or ZAAPS—has been conducted since 2002. As of 2007 [update], overall resistance to linezolid in 23 countries was less than 0.2%, and nonexistent among streptococci. Resistance was only found in Brazil, China, Ireland, and Italy, among coagulase-negative staphylococci (0.28% of samples resistant), enterococci (0.11%), and S. aureus (0.03%). [117] In the United Kingdom and Ireland, no resistance was found in staphylococci collected from bacteremia cases between 2001 and 2006, [118] although resistance in enterococci has been reported. [119] Some authors have predicted that resistance in E. faecium will increase if linezolid use continues at current levels or increases. [120] Nevertheless, linezolid continues to be an important antimicrobial agent with near-complete activity (0.05% resistance). [107]
Mechanism
The intrinsic resistance of most Gram-negative bacteria to linezolid is due to the activity of efflux pumps, which actively "pump" linezolid out of the cell faster than it can accumulate. [35] [121]
Gram-positive bacteria usually develop resistance to linezolid as the result of a point mutation known as G2576T, in which a guanine base is replaced with thymine in base pair 2576 of the genes coding for 23S ribosomal RNA. [122] [123] This is the most common mechanism of resistance in staphylococci, and the only one known to date in isolates of E. faecium. [120] Other mechanisms have been identified in Streptococcus pneumoniae (including mutations in an RNA methyltransferase that methylates G2445 of the 23S rRNA and mutations causing increased expression of ABC transporter genes) [124] and in Staphylococcus epidermidis. [125] [126]
History
The oxazolidinones have been known as monoamine oxidase inhibitors since the late 1950s. Their antimicrobial properties were discovered by researchers at E.I. duPont de Nemours in the 1970s. [108] In 1978, DuPont patented a series of oxazolidinone derivatives as being effective in the treatment of bacterial and fungal plant diseases, and in 1984, another patent described their usefulness in treating bacterial infections in mammals. [54] [108] In 1987, DuPont scientists presented a detailed description of the oxazolidinones as a new class of antibiotics with a novel mechanism of action. [108] [127] Early compounds were found to produce liver toxicity, however, and development was discontinued. [72]
Pharmacia & Upjohn (now part of Pfizer) started its own oxazolidinone research program in the 1990s. Studies of the compounds' structure–activity relationships led to the development of several subclasses of oxazolidinone derivatives, with varying safety profiles and antimicrobial activity. Two compounds were considered drug candidates: eperezolid (codenamed PNU-100592) and linezolid (PNU-100766). [35] [73] In the preclinical stages of development, they were similar in safety and antibacterial activity, so they were taken to Phase I clinical trials to identify any difference in pharmacokinetics. [72] [128] Linezolid was found to have a pharmacokinetic advantage—requiring only twice-daily dosage, while eperezolid needed to be given three times a day to achieve similar exposure—and therefore proceeded to further trials. [35] The U.S. Food and Drug Administration (FDA) approved linezolid on 18 April 2000. [129] Approval followed in Brazil (June 2000), [130] the United Kingdom (January 2001), [7] [73] Japan and Canada (April 2001), [131] [132] [133] Europe (throughout 2001), [134] and other countries in Latin America and Asia. [132]
As of 2009 [update], linezolid was the only oxazolidinone antibiotic available. [135] Other members of this class have entered development, such as posizolid (AZD2563), [136] ranbezolid (RBx 7644), [137] and radezolid (RX-1741). [138] In 2014, the FDA approved tedizolid phosphate, a second-generation oxazolidinone derivative, for acute bacterial skin and skin structure infection. [139] [140]
Society and culture
Economics
Linezolid was quite expensive in 2009; a course of treatment may cost one or two thousand U.S. dollars for the drug alone, [64] not to mention other costs (such as those associated with hospital stay). With the medication becoming generic the price has decreased. In India as of 2015 a month of linezolid, as would be used to treat tuberculosis cost about US$60. [11]
However, because intravenous linezolid may be switched to an oral formulation (tablets or oral solution) without jeopardizing efficacy, people may be discharged from hospital relatively early and continue treatment at home, whereas home treatment with injectable antibiotics may be impractical. [141] Reducing the length of hospital stay reduces the overall cost of treatment, even though linezolid may have a higher acquisition cost—that is, it may be more expensive—than comparable antibiotics.
Studies have been conducted in several countries with different health care system models to assess the cost-effectiveness of linezolid compared to glycopeptides such as vancomycin or teicoplanin. In most countries, linezolid was more cost-effective than comparable antibiotics for the treatment of hospital-acquired pneumonia and complicated skin and skin structure infections, either due to higher cure and survival rates or lower overall treatment costs. [141]
In 2009, Pfizer paid $2.3 billion and entered a corporate integrity agreement to settle charges that it had misbranded and illegally promoted four drugs, and caused false claims to be submitted to government healthcare programs for uses that had not been approved by the United States Food and Drug Administration. [142] $1.3 billion was paid to settle criminal charges of illegally marketing the anti-inflammatory valdecoxib, while $1 billion was paid in civil fines regarding illegal marketing of three other drugs, including Zyvox. [143]
Brand names
| A | Amizole 500 (Kenya), Anozilad (Poland), Antizolid (Greece), Arlid (India), Arlin (Bangladesh), Averozolid & Debacozoline (Egypt) |
| B | |
| C | |
| D | Dilizolen (Poland, Slovakia, Netherlands, Bulgaria) |
| E | Entavar (India) |
| F | |
| G | Grampolid (Netherlands), Grampolyve (Netherlands), Gramposimide (Poland, Netherlands), Grampoxid (Netherlands) |
| H | |
| I | |
| J | |
| K | |
| L | Linzolid (Bangladesh), Lidobact (Netherlands), Linez (Bangladesh, Egypt), Linezolid Accord (Netherlands), Linezolid Amneal (Netherlands), Linezolid Betapharm (Netherlands), Linezolid Farmaprojects (Netherlands), Linezolid Fresenius Kabi (Netherlands), Linezolid GNP (Egypt), Linezolid Hetero (Netherlands), Linezolid Kabi (Croatia, Poland), Linezolid Mylan (Netherlands), Linezolid Pfizer (Netherlands), Linezolid Pliva (Croatia), Linezolid Polpharma (Netherlands, Poland), Linezolid Richet (Argentina), Linezolid Sandoz (Belgium, Switzerland, Netherlands, Slovakia, Estonia, Croatia, Poland), Linezolid Teva (Netherlands, Romania), Linezolid Zentiva (Poland), Linezolida Teva (Portugal), Linezone (Turkey), Linid (India), Linosept (India), Linozid (Bangladesh), Linxyd (Netherlands), Linzowin (India), Litrecan (Argentina), Livegramide (Netherlands), Lizbid (India), Lizemox (India), Lizolid (India, Vietnam), Lizoliden (Netherlands), Lizomac (India), Lizomed (India), Lizorex (India), Lizox (Netherlands), Lorezogram (Netherlands), Lynvox (Netherlands), Lynz (Croatia) |
| M | |
| N | Natlinez (Netherlands) |
| O | |
| P | Pneumolid (Croatia, Netherlands, Poland, Romania, Bulgaria) |
| Q | |
| R | Ralinz (India), Respenzo (Egypt) |
| S | Synzolid (Netherlands) |
| T | |
| U | |
| V | Voxazoldin (Egypt) |
| W | |
| X | |
| Y | |
| Z | Zenix (Bosnia and Herzegovina, Serbia), Zizolid (Turkey), Zodlin (India), Zolinid (Bulgaria), Zyvox (Georgia, Chile, Argentina, Australia, China, Ecuador, Egypt, United Kingdom, Hong Kong, Indonesia, Ireland, South Korea, Malta, Malaysia, New Zealand, Philippines, Singapore, Thailand, Taiwan, Japan, United States), Zyvoxam (Canada), Zyvoxid (Israel, Austria, Belgium, Bulgaria, Switzerland, Czech Republic, Denmark, Estonia, Spain, Finland, France, Greece, Germany, Croatia, Iceland, Lithuania, Latvia, Netherlands, Norway, Portugal, Romania, Sweden, Slovakia, Tunisia, Turkey, South Africa, Poland, Italy, Bosnia and Herzegovina) |
| Generic Combination | Brand Name |
|---|---|
| linezolid and cefixime | Zifi-Turbo (India) |
References
- ^ "Linezolid (Zyvox) Use During Pregnancy". Drugs.com. 16 April 2018. Archived from the original on 17 March 2020. Retrieved 17 March 2020.
- ^ "Linezolid, injection, 600 mg per 300 mL, tablet, 600 mg and powder for oral suspension, 20 mg per mL, Zyvox®". Australian Government Department of Health and Aged Care. 24 September 2001. Archived from the original on 18 November 2022. Retrieved 30 March 2024.
- ^ "Public Summary" (PDF). Retrieved 30 March 2024.
- ^ "Prescription medicines: registration of new generic medicines and biosimilar medicines, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Archived from the original on 6 July 2023. Retrieved 30 March 2024.
- ^ "Product information". health-products.canada.ca. 31 July 2020. Archived from the original on 29 March 2024. Retrieved 29 March 2024.
- ^ "Product information". health-products.canada.ca. 7 February 2006. Archived from the original on 29 March 2024. Retrieved 29 March 2024.
- ^ a b c "Zyvox 600 mg Film-Coated Tablets, 100 mg/5 ml Granules for Oral Suspension, 2 mg/ml Solution for Infusion – Summary of Product Characteristics (SPC)". electronic Medicines Compendium. 24 June 2009. Archived from the original on 6 August 2012. Retrieved 3 July 2009.
- ^ a b c d e f g h i j k l "Zyvox- linezolid injection, solution Zyvox- linezolid tablet, film coated Zyvox- linezolid suspension". DailyMed. 21 February 2020. Archived from the original on 26 April 2021. Retrieved 17 March 2020.
- ^ a b c d e f g h i j k l Roger C, Roberts JA, Muller L (May 2018). "Clinical Pharmacokinetics and Pharmacodynamics of Oxazolidinones". Clinical Pharmacokinetics. 57 (5): 559–575. doi: 10.1007/s40262-017-0601-x. PMID 29063519. S2CID 4961324.
- ^ a b c d e f g h i j "Linezolid". The American Society of Health-System Pharmacists. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ^ a b World Health Organization (2015). The selection and use of essential medicines. Twentieth report of the WHO Expert Committee 2015 (including 19th WHO Model List of Essential Medicines and 5th WHO Model List of Essential Medicines for Children). Geneva: World Health Organization. pp. 26–33. hdl: 10665/189763. ISBN 9789241209946. ISSN 0512-3054. WHO technical report series;994.
- ^ a b c Marino PL, Sutin KM (2007). "Antimicrobial therapy". The ICU book. Hagerstown, MD: Lippincott Williams & Wilkins. p. 817. ISBN 978-0-7817-4802-5.
- ^ a b "Linezolid Side Effects in Detail". drugs.com. Archived from the original on 20 December 2016. Retrieved 11 December 2016.
- ^ a b c Swaney SM, Aoki H, Ganoza MC, Shinabarger DL (December 1998). "The oxazolidinone linezolid inhibits initiation of protein synthesis in bacteria". Antimicrobial Agents and Chemotherapy. 42 (12): 3251–5. doi: 10.1128/AAC.42.12.3251. PMC 106030. PMID 9835522.
-
^ Mendes RE, Deshpande LM, Jones RN (April 2014). "Linezolid update: stable in vitro activity following more than a decade of clinical use and summary of associated resistance mechanisms". Drug Resistance Updates. 17 (1–2): 1–12.
doi:
10.1016/j.drup.2014.04.002.
PMID
24880801.
Emergence of resistance has been limited ... It is still uncertain whether the occurrences of such isolates are becoming more prevalent.
- ^ Li JJ, Corey EJ (2013). Drug Discovery: Practices, Processes, and Perspectives. John Wiley & Sons. p. 6. ISBN 978-1-118-35446-9. Archived from the original on 10 September 2017.
- ^ Torok E, Moran E, Cooke F (2009). "Chapter 2 Antimicrobials". Oxford Handbook of Infectious Diseases and Microbiology. OUP Oxford. ISBN 978-0-19-103962-1. Archived from the original on 8 September 2017.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl: 10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ World Health Organization (2019). Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. hdl: 10665/312266. ISBN 9789241515528.
- ^ Wroe D (28 February 2002). "An antibiotic to fight immune bugs". The Age. Archived from the original on 27 January 2010. Retrieved 16 May 2009.
- ^ Wilson AP, Cepeda JA, Hayman S, Whitehouse T, Singer M, Bellingan G (August 2006). "In vitro susceptibility of Gram-positive pathogens to linezolid and teicoplanin and effect on outcome in critically ill patients". The Journal of Antimicrobial Chemotherapy. 58 (2): 470–3. doi: 10.1093/jac/dkl233. PMID 16735420.
- ^ Bozdogan B, Appelbaum PC (February 2004). "Oxazolidinones: activity, mode of action, and mechanism of resistance". International Journal of Antimicrobial Agents. 23 (2): 113–9. doi: 10.1016/j.ijantimicag.2003.11.003. PMID 15013035.
- ^ a b c d e f g h i j Herrmann DJ, Peppard WJ, Ledeboer NA, Theesfeld ML, Weigelt JA, Buechel BJ (December 2008). "Linezolid for the treatment of drug-resistant infections". Expert Review of Anti-infective Therapy. 6 (6): 825–48. doi: 10.1586/14787210.6.6.825. ISSN 1478-7210. PMID 19053895. S2CID 8791647.
- ^ a b c Falagas ME, Siempos II, Vardakas KZ (January 2008). "Linezolid versus glycopeptide or beta-lactam for treatment of Gram-positive bacterial infections: meta-analysis of randomised controlled trials". Lancet Infectious Diseases. 8 (1): 53–66. doi: 10.1016/S1473-3099(07)70312-2. ISSN 1473-3099. PMID 18156089. Structured abstract with quality assessment available at DARE Archived 4 October 2011 at the Wayback Machine.
- ^ Tascini C, Gemignani G, Doria R, et al. (June 2009). "Linezolid treatment for gram-positive infections: a retrospective comparison with teicoplanin". Journal of Chemotherapy. 21 (3): 311–6. doi: 10.1179/joc.2009.21.3.311. ISSN 1120-009X. PMID 19567352. S2CID 19381342.
- ^ Chow I, Lemos EV, Einarson TR (2008). "Management and prevention of diabetic foot ulcers and infections: a health economic review". PharmacoEconomics. 26 (12): 1019–35. doi: 10.2165/0019053-200826120-00005. PMID 19014203. S2CID 30903985.
- ^ Lipsky BA, Itani K, Norden C (January 2004). "Treating foot infections in diabetic patients: a randomized, multicenter, open-label trial of linezolid versus ampicillin-sulbactam/amoxicillin-clavulanate". Clinical Infectious Diseases. 38 (1): 17–24. doi: 10.1086/380449. PMID 14679443. S2CID 20586344.
- ^ a b c d Pigrau C, Almirante B (April 2009). "[Oxazolidinones, glycopeptides and cyclic lipopeptides]" [Oxazolidinones, glycopeptides and cyclic lipopeptides] (PDF). Enfermedades Infecciosas y Microbiologia Clinica (in Spanish). 27 (4): 236–46. doi: 10.1016/j.eimc.2009.02.004. PMID 19406516. Archived from the original (PDF) on 23 July 2011.
- ^ Vardakas KZ, Horianopoulou M, Falagas ME (June 2008). "Factors associated with treatment failure in patients with diabetic foot infections: An analysis of data from randomized controlled trials". Diabetes Research and Clinical Practice. 80 (3): 344–51. doi: 10.1016/j.diabres.2008.01.009. PMID 18291550.
- ^ Grammatikos A, Falagas ME (2008). "Linezolid for the treatment of skin and soft tissue infection". Expert Review of Dermatology. 3 (5): 539–48. doi: 10.1586/17469872.3.5.539.
- ^ a b Mandell LA, Wunderink RG, Anzueto A, et al. (March 2007). "Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults". Clinical Infectious Diseases. 44 (Suppl 2): S27–72. doi: 10.1086/511159. ISSN 1058-4838. PMC 7107997. PMID 17278083.
- ^ BTS Pneumonia Guidelines Committee (30 April 2004). "BTS guidelines for the management of community acquired pneumonia in adults – 2004 update" (PDF). British Thoracic Society. Archived from the original (PDF) on 7 April 2009. Retrieved 30 June 2009.
- ^ a b c American Thoracic Society, Infectious Diseases Society (February 2005). "Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia". American Journal of Respiratory and Critical Care Medicine. 171 (4): 388–416. doi: 10.1164/rccm.200405-644ST. PMID 15699079. S2CID 14907563.
- ^ Koya D, Shibuya K, Kikkawa R, Haneda M (December 2004). "Successful recovery of infective endocarditis-induced rapidly progressive glomerulonephritis by steroid therapy combined with antibiotics: a case report". BMC Nephrology. 5 (1): 18. doi: 10.1186/1471-2369-5-18. PMC 544880. PMID 15610562.
- ^ a b c d e f g h Barbachyn MR, Ford CW (May 2003). "Oxazolidinone structure-activity relationships leading to linezolid". Angewandte Chemie International Edition in English. 42 (18): 2010–23. doi: 10.1002/anie.200200528. ISSN 1433-7851. PMID 12746812.
- ^ Pankey GA, Sabath LD (March 2004). "Clinical relevance of bacteriostatic versus bactericidal mechanisms of action in the treatment of Gram-positive bacterial infections". Clinical Infectious Diseases. 38 (6): 864–70. doi: 10.1086/381972. ISSN 1058-4838. PMID 14999632.
- ^ Falagas ME, Manta KG, Ntziora F, Vardakas KZ (August 2006). "Linezolid for the treatment of patients with endocarditis: a systematic review of the published evidence". Journal of Antimicrobial Chemotherapy. 58 (2): 273–80. doi: 10.1093/jac/dkl219. PMID 16735427.
- ^ Babcock HM, Ritchie DJ, Christiansen E, Starlin R, Little R, Stanley S (May 2001). "Successful treatment of vancomycin-resistant Enterococcus endocarditis with oral linezolid". Clinical Infectious Diseases. 32 (9): 1373–5. doi: 10.1086/319986. ISSN 1058-4838. PMID 11303275.
- ^ Ang JY, Lua JL, Turner DR, Asmar BI (December 2003). "Vancomycin-resistant Enterococcus faecium endocarditis in a premature infant successfully treated with linezolid". The Pediatric Infectious Disease Journal. 22 (12): 1101–3. doi: 10.1097/01.inf.0000101784.83146.0c. ISSN 0891-3668. PMID 14688576.
- ^ Archuleta S, Murphy B, Keller MJ (September 2004). "Successful treatment of vancomycin-resistant Enterococcus faecium endocarditis with linezolid in a renal transplant recipient with human immunodeficiency virus infection". Transplant Infectious Disease. 6 (3): 117–9. doi: 10.1111/j.1399-3062.2004.00059.x. ISSN 1398-2273. PMID 15569227. S2CID 40817941.
- ^ Zimmer SM, Caliendo AM, Thigpen MC, Somani J (August 2003). "Failure of linezolid treatment for enterococcal endocarditis". Clinical Infectious Diseases. 37 (3): e29–30. doi: 10.1086/375877. ISSN 1058-4838. PMID 12884185.
- ^ Tsigrelis C, Singh KV, Coutinho TD, Murray BE, Baddour LM (February 2007). "Vancomycin-Resistant Enterococcus faecalis Endocarditis: Linezolid Failure and Strain Characterization of Virulence Factors". Journal of Clinical Microbiology. 45 (2): 631–5. doi: 10.1128/JCM.02188-06. PMC 1829077. PMID 17182759.
- ^ Berdal JE, Eskesen A (2008). "Short-term success, but long-term treatment failure with linezolid for enterococcal endocarditis". Scandinavian Journal of Infectious Diseases. 40 (9): 765–6. doi: 10.1080/00365540802087209. ISSN 0036-5548. PMID 18609208. S2CID 12651659.
- ^ Falagas ME, Siempos II, Papagelopoulos PJ, Vardakas KZ (March 2007). "Linezolid for the treatment of adults with bone and joint infections". International Journal of Antimicrobial Agents. 29 (3): 233–9. doi: 10.1016/j.ijantimicag.2006.08.030. ISSN 0924-8579. PMID 17204407. Review.
- ^ Bassetti M, Vitale F, Melica G, et al. (March 2005). "Linezolid in the treatment of Gram-positive prosthetic joint infections". Journal of Antimicrobial Chemotherapy. 55 (3): 387–90. doi: 10.1093/jac/dki016. PMID 15705640.
- ^ Aneziokoro CO, Cannon JP, Pachucki CT, Lentino JR (December 2005). "The effectiveness and safety of oral linezolid for the primary and secondary treatment of osteomyelitis". Journal of Chemotherapy. 17 (6): 643–50. doi: 10.1179/joc.2005.17.6.643. ISSN 1120-009X. PMID 16433195. S2CID 46391229.
- ^ Senneville E, Legout L, Valette M, et al. (August 2006). "Effectiveness and tolerability of prolonged linezolid treatment for chronic osteomyelitis: a retrospective study". Clinical Therapeutics. 28 (8): 1155–63. doi: 10.1016/j.clinthera.2006.08.001. ISSN 0149-2918. PMID 16982292.
- ^ Rao N, Hamilton CW (October 2007). "Efficacy and safety of linezolid for Gram-positive orthopedic infections: a prospective case series". Diagnostic Microbiology and Infectious Disease. 59 (2): 173–9. doi: 10.1016/j.diagmicrobio.2007.04.006. ISSN 0732-8893. PMID 17574788.
- ^ Papadopoulos A, Plachouras D, Giannitsioti E, Poulakou G, Giamarellou H, Kanellakopoulou K (April 2009). "Efficacy and tolerability of linezolid in chronic osteomyelitis and prosthetic joint infections: a case-control study". Journal of Chemotherapy. 21 (2): 165–9. doi: 10.1179/joc.2009.21.2.165. ISSN 1120-009X. PMID 19423469. S2CID 12400080.
- ^ von der Lippe B, Sandven P, Brubakk O (February 2006). "Efficacy and safety of linezolid in multidrug resistant tuberculosis (MDR-TB)--a report of ten cases". The Journal of Infection. 52 (2): 92–6. doi: 10.1016/j.jinf.2005.04.007. PMID 15907341.
- ^ Park IN, Hong SB, Oh YM, Kim MN, Lim CM, Lee SD, et al. (September 2006). "Efficacy and tolerability of daily-half dose linezolid in patients with intractable multidrug-resistant tuberculosis". The Journal of Antimicrobial Chemotherapy. 58 (3): 701–4. doi: 10.1093/jac/dkl298. PMID 16857689.
- ^ Fortún J, Martín-Dávila P, Navas E, Pérez-Elías MJ, Cobo J, Tato M, et al. (July 2005). "Linezolid for the treatment of multidrug-resistant tuberculosis". The Journal of Antimicrobial Chemotherapy. 56 (1): 180–5. doi: 10.1093/jac/dki148. PMID 15911549.
- ^ Jaksic B, Martinelli G, Perez-Oteyza J, Hartman CS, Leonard LB, Tack KJ (March 2006). "Efficacy and safety of linezolid compared with vancomycin in a randomized, double-blind study of febrile neutropenic patients with cancer". Clinical Infectious Diseases. 42 (5): 597–607. doi: 10.1086/500139. ISSN 1058-4838. PMID 16447103. Criticism in doi: 10.1086/504431; author reply in doi: 10.1086/504437.
- ^ a b c d e f g h Moellering RC (January 2003). "Linezolid: the first oxazolidinone antimicrobial" (PDF). Annals of Internal Medicine. 138 (2): 135–42. doi: 10.7326/0003-4819-138-2-200301210-00015. ISSN 0003-4819. PMID 12529096. S2CID 23689046. Archived (PDF) from the original on 14 March 2020. Retrieved 25 September 2019.
- ^ Cottagnoud P, Gerber CM, Acosta F, Cottagnoud M, Neftel K, Täuber MG (December 2000). "Linezolid against penicillin-sensitive and -resistant pneumococci in the rabbit meningitis model". Journal of Antimicrobial Chemotherapy. 46 (6): 981–5. doi: 10.1093/jac/46.6.981. PMID 11102418.
- ^ a b Sabbatani S, Manfredi R, Frank G, Chiodo F (June 2005). "Linezolid in the treatment of severe central nervous system infections resistant to recommended antimicrobial compounds". Le Infezioni in Medicina. 13 (2): 112–9. ISSN 1124-9390. PMID 16220032. Archived from the original on 22 July 2011.
- ^ Ntziora F, Falagas ME (February 2007). "Linezolid for the treatment of patients with central nervous system infection". Annals of Pharmacotherapy. 41 (2): 296–308. doi: 10.1345/aph.1H307. ISSN 1060-0280. PMID 17284501. S2CID 33514115. Structured abstract with quality assessment available at DARE Archived 2 September 2011 at the Wayback Machine.
- ^ Tunkel AR, Hartman BJ, Kaplan SL, et al. (November 2004). "Practice guidelines for the management of bacterial meningitis". Clinical Infectious Diseases. 39 (9): 1267–84. doi: 10.1086/425368. ISSN 1058-4838. PMID 15494903.
- ^ Naesens R, Ronsyn M, Druwé P, Denis O, Ieven M, Jeurissen A (June 2009). "Central nervous system invasion by community-acquired methicillin-resistant Staphylococcus aureus: case report and review of the literature". Journal of Medical Microbiology. 58 (Pt 9): 1247–51. doi: 10.1099/jmm.0.011130-0. ISSN 0022-2615. PMID 19528145.
- ^ a b c [No authors listed] (16 March 2007). "Information for Healthcare Professionals: Linezolid (marketed as Zyvox)". U.S. Food and Drug Administration (FDA). Archived from the original on 19 October 2010. Retrieved 15 September 2010.
- ^ a b Wilcox MH, Tack KJ, Bouza E, et al. (January 2009). "Complicated skin and skin-structure infections and catheter-related bloodstream infections: noninferiority of linezolid in a phase 3 study". Clinical Infectious Diseases. 48 (2): 203–12. doi: 10.1086/595686. ISSN 1058-4838. PMID 19072714.
- ^ a b c Ament PW, Jamshed N, Horne JP (February 2002). "Linezolid: its role in the treatment of gram-positive, drug-resistant bacterial infections". American Family Physician. 65 (4): 663–70. ISSN 0002-838X. PMID 11871684. Archived from the original on 24 July 2008.
- ^ Buck ML (June 2003). "Linezolid use for resistant Gram-positive infections in children" (PDF). Pediatric Pharmacotherapy. 9 (6). Archived from the original (PDF) on 5 June 2011. Retrieved 8 June 2009.
- ^ a b c d e f g Lexi-Comp (August 2008). "Linezolid". The Merck Manual Professional. Archived from the original on 26 April 2009. Retrieved on 14 May 2009.
- ^ Lovering AM, Le Floch R, Hovsepian L, et al. (March 2009). "Pharmacokinetic evaluation of linezolid in patients with major thermal injuries". Journal of Antimicrobial Chemotherapy. 63 (3): 553–9. doi: 10.1093/jac/dkn541. ISSN 0305-7453. PMID 19153078.
- ^ a b Davaro RE, Glew RH, Daly JS (2004). "Oxazolidinones, quinupristin-dalfopristin, and daptomycin". In Gorbach SL, Bartlett JG, Blacklow NR (eds.). Infectious diseases. Hagerstown, MD: Lippincott Williams & Wilkins. pp. 241–3. ISBN 978-0-7817-3371-7. Retrieved 20 June 2009.
- ^ Jones RN, Stilwell MG, Hogan PA, Sheehan DJ (April 2007). "Activity of Linezolid against 3,251 Strains of Uncommonly Isolated Gram-Positive Organisms: Report from the SENTRY Antimicrobial Surveillance Program". Antimicrobial Agents and Chemotherapy. 51 (4): 1491–3. doi: 10.1128/AAC.01496-06. PMC 1855453. PMID 17210770.
- ^ Jodlowski TZ, Melnychuk I, Conry J (October 2007). "Linezolid for the treatment of Nocardia spp. infections". Annals of Pharmacotherapy. 41 (10): 1694–9. doi: 10.1345/aph.1K196. ISSN 1060-0280. PMID 17785610. S2CID 33975237.
- ^ a b c d [No authors listed] (2001). "Linezolid: First of a New Drug Class for Gram-Positive Infections". Drugs & Therapy Perspectives. 17 (9): 1–6. doi: 10.2165/00042310-200117090-00001. S2CID 195232060. Archived from the original on 21 May 2013. Free full text with registration at Medscape.
- ^ [No authors listed] (5 August 2008). "Animal Bites and Pasteurella multocida: Information for Healthcare Staff". Health Protection Agency. Archived from the original on 26 January 2010. Retrieved on 15 May 2009.
- ^ Geisler WM, Malhotra U, Stamm WE (December 2001). "Pneumonia and sepsis due to fluoroquinolone-resistant Capnocytophaga gingivalis after autologous stem cell transplantation". Bone Marrow Transplantation. 28 (12): 1171–3. doi: 10.1038/sj.bmt.1703288. ISSN 0268-3369. PMID 11803363. S2CID 34825943.
- ^ a b c Livermore DM (September 2000). "Quinupristin/dalfopristin and linezolid: where, when, which and whether to use?". Journal of Antimicrobial Chemotherapy. 46 (3): 347–50. doi: 10.1093/jac/46.3.347. ISSN 0305-7453. PMID 10980159.
- ^ a b c d e f g h French G (May 2003). "Safety and tolerability of linezolid". The Journal of Antimicrobial Chemotherapy. 51 (Suppl 2): ii45–53. doi: 10.1093/jac/dkg253. PMID 12730142. Review. Includes extensive discussion of the hematological adverse effects of linezolid.
- ^ a b c d Metaxas EI, Falagas ME (July 2009). "Update on the safety of linezolid". Expert Opinion on Drug Safety. 8 (4): 485–91. doi: 10.1517/14740330903049706. PMID 19538105. S2CID 5691388.
- ^ Zabel LT, Worm S (June 2005). "Linezolid contributed to Clostridium difficile colitis with fatal outcome". Infection. 33 (3): 155–7. doi: 10.1007/s15010-005-4112-6. PMID 15940418. S2CID 37705529.
- ^ Peláez T, Alonso R, Pérez C, Alcalá L, Cuevas O, Bouza E (May 2002). "In vitro activity of linezolid against Clostridium difficile". Antimicrobial Agents and Chemotherapy. 46 (5): 1617–8. doi: 10.1128/AAC.46.5.1617-1618.2002. PMC 127182. PMID 11959617.
- ^ Lin YH, Wu VC, Tsai IJ, Ho YL, Hwang JJ, Tsau YK, et al. (October 2006). "High frequency of linezolid-associated thrombocytopenia among patients with renal insufficiency". International Journal of Antimicrobial Agents. 28 (4): 345–351. doi: 10.1016/j.ijantimicag.2006.04.017. PMID 16935472.
- ^ Spellberg B, Yoo T, Bayer AS (October 2004). "Reversal of linezolid-associated cytopenias, but not peripheral neuropathy, by administration of vitamin B6". Journal of Antimicrobial Chemotherapy. 54 (4): 832–5. doi: 10.1093/jac/dkh405. PMID 15317746.
- ^ Plachouras D, Giannitsioti E, Athanassia S, et al. (November 2006). "No effect of pyridoxine on the incidence of myelosuppression during prolonged linezolid treatment". Clinical Infectious Diseases. 43 (9): e89–91. doi: 10.1086/508280. ISSN 1058-4838. PMID 17029128.
- ^ a b del Pino BM (23 February 2010). "Chemotherapy-induced Peripheral Neuropathy". NCI Cancer Bulletin. 7 (4): 6. Archived from the original on 11 December 2011.
- ^ Cooper R, Deakin JJ (2016). "Africa's gift to the world". Botanical Miracles: Chemistry of Plants That Changed the World. CRC Press. pp. 46–51. ISBN 9781498704304.
- ^ Keglevich P, Hazai L, Kalaus G, Szántay C (May 2012). "Modifications on the basic skeletons of vinblastine and vincristine". Molecules. 17 (5): 5893–914. doi: 10.3390/molecules17055893. PMC 6268133. PMID 22609781.
- ^ Raviña E (2011). "Vinca alkaloids". The evolution of drug discovery: From traditional medicines to modern drugs. John Wiley & Sons. pp. 157–159. ISBN 9783527326693.
- ^ Grisold W, Oberndorfer S, Windebank AJ (2012). "Chemotherapy and polyneuropathies" (PDF). European Association of Neurooncology Magazine. 12 (1). Archived (PDF) from the original on 6 October 2014.
- ^ "Review: could Herceptin cause Peripheral sensory neuropathy". Archived from the original on 17 March 2016. Retrieved 4 May 2016.
- ^ Narita M, Tsuji BT, Yu VL (August 2007). "Linezolid-associated peripheral and optic neuropathy, lactic acidosis, and serotonin syndrome". Pharmacotherapy. 27 (8): 1189–97. doi: 10.1592/phco.27.8.1189. PMID 17655517. S2CID 40772687.
- ^ Bressler AM, Zimmer SM, Gilmore JL, Somani J (August 2004). "Peripheral neuropathy associated with prolonged use of linezolid". The Lancet. Infectious Diseases. 4 (8): 528–31. doi: 10.1016/S1473-3099(04)01109-0. PMID 15288827.
- ^ Brown J, Aitken SL, van Manen RP (June 2011). "Potential for linezolid-related blindness: a review of spontaneous adverse event reports". Pharmacotherapy. 31 (6): 585–90. doi: 10.1592/phco.31.6.585. PMID 21923442. S2CID 28881160.
- ^ Chao CC, Sun HY, Chang YC, Hsieh ST (January 2008). "Painful neuropathy with skin denervation after prolonged use of linezolid". Journal of Neurology, Neurosurgery, and Psychiatry. 79 (1): 97–9. doi: 10.1136/jnnp.2007.127910. PMC 3029234. PMID 17766431. S2CID 33138699.
- ^ Saijo T, Hayashi K, Yamada H, Wakakura M (June 2005). "Linezolid-induced optic neuropathy". American Journal of Ophthalmology. 139 (6): 1114–6. doi: 10.1016/j.ajo.2004.11.047. PMID 15953450.
- ^ a b Barnhill AE, Brewer MT, Carlson SA (August 2012). "Adverse effects of antimicrobials via predictable or idiosyncratic inhibition of host mitochondrial components". Antimicrobial Agents and Chemotherapy. 56 (8): 4046–51. doi: 10.1128/AAC.00678-12. PMC 3421593. PMID 22615289. Review. For the original case series, see Soriano A, Miró O, Mensa J (November 2005). "Mitochondrial toxicity associated with linezolid". The New England Journal of Medicine. 353 (21): 2305–6. doi: 10.1056/NEJM200511243532123. PMID 16306535.
- ^ Javaheri M, Khurana RN, O'hearn TM, Lai MM, Sadun AA (January 2007). "Linezolid-induced optic neuropathy: a mitochondrial disorder?". The British Journal of Ophthalmology. 91 (1): 111–5. doi: 10.1136/bjo.2006.102541. PMC 1857552. PMID 17179125.
- ^ McKee EE, Ferguson M, Bentley AT, Marks TA (June 2006). "Inhibition of mammalian mitochondrial protein synthesis by oxazolidinones". Antimicrobial Agents and Chemotherapy. 50 (6): 2042–9. doi: 10.1128/AAC.01411-05. PMC 1479116. PMID 16723564.
- ^ Bishop E, Melvani S, Howden BP, Charles PG, Grayson ML (April 2006). "Good clinical outcomes but high rates of adverse reactions during linezolid therapy for serious infections: a proposed protocol for monitoring therapy in complex patients". Antimicrobial Agents and Chemotherapy. 50 (4): 1599–602. doi: 10.1128/AAC.50.4.1599-1602.2006. PMC 1426936. PMID 16569895.
- ^ King MP, Attardi G (October 1989). "Human cells lacking mtDNA: repopulation with exogenous mitochondria by complementation". Science. 246 (4929): 500–3. Bibcode: 1989Sci...246..500K. doi: 10.1126/science.2814477. PMID 2814477.
- ^ Lawrence KR, Adra M, Gillman PK (June 2006). "Serotonin toxicity associated with the use of linezolid: a review of postmarketing data". Clinical Infectious Diseases. 42 (11): 1578–83. doi: 10.1086/503839. PMID 16652315.
- ^ Huang V, Gortney JS (December 2006). "Risk of serotonin syndrome with concomitant administration of linezolid and serotonin agonists". Pharmacotherapy. 26 (12): 1784–93. doi: 10.1592/phco.26.12.1784. PMID 17125439. S2CID 24150415.
- ^ Waknine Y (5 September 2008). "FDA Safety Changes: Mirena, Zyvox, Orencia". Medscape. Archived from the original on 2 December 2008. Retrieved 6 September 2008. Freely available with registration.
- ^ Stalker DJ, Jungbluth GL (2003). "Clinical pharmacokinetics of linezolid, a novel oxazolidinone antibacterial". Clinical Pharmacokinetics. 42 (13): 1129–40. doi: 10.2165/00003088-200342130-00004. PMID 14531724. S2CID 37436341.
- ^ Renslo AR (May 2010). "Antibacterial oxazolidinones: emerging structure-toxicity relationships". Expert Review of Anti-infective Therapy. 8 (5): 565–74. doi: 10.1586/eri.10.26. PMID 20455685. S2CID 23670767.
- ^ a b Zhanel GG, Love R, Adam H, Golden A, Zelenitsky S, Schweizer F, et al. (February 2015). "Tedizolid: a novel oxazolidinone with potent activity against multidrug-resistant gram-positive pathogens". Drugs. 75 (3): 253–70. doi: 10.1007/s40265-015-0352-7. PMID 25673021. S2CID 3334681.
- ^ Ippolito JA, Kanyo ZF, Wang D, Franceschi FJ, Moore PB, Steitz TA, et al. (June 2008). "Crystal structure of the oxazolidinone antibiotic linezolid bound to the 50S ribosomal subunit". Journal of Medicinal Chemistry. 51 (12): 3353–6. doi: 10.1021/jm800379d. PMID 18494460.
- ^ Wilson DN, Schluenzen F, Harms JM, Starosta AL, Connell SR, Fucini P (September 2008). "The oxazolidinone antibiotics perturb the ribosomal peptidyl-transferase center and effect tRNA positioning". Proceedings of the National Academy of Sciences of the United States of America. 105 (36): 13339–44. Bibcode: 2008PNAS..10513339W. doi: 10.1073/pnas.0804276105. PMC 2533191. PMID 18757750.
- ^ Kalil AC, Murthy MH, Hermsen ED, Neto FK, Sun J, Rupp ME (September 2010). "Linezolid versus vancomycin or teicoplanin for nosocomial pneumonia: a systematic review and meta-analysis". Critical Care Medicine. 38 (9): 1802–8. doi: 10.1097/CCM.0b013e3181eb3b96. PMID 20639754. S2CID 6289226.
- ^ a b Slatter JG, Stalker DJ, Feenstra KL, Welshman IR, Bruss JB, Sams JP, et al. (August 2001). "Pharmacokinetics, metabolism, and excretion of linezolid following an oral dose of [(14)C]linezolid to healthy human subjects" (PDF). Drug Metabolism and Disposition. 29 (8): 1136–45. PMID 11454733. Archived from the original on 28 August 2021. Retrieved 3 April 2011.
- ^ Sisson TL, Jungbluth GL, Hopkins NK (January 2002). "Age and sex effects on the pharmacokinetics of linezolid". European Journal of Clinical Pharmacology. 57 (11): 793–7. doi: 10.1007/s00228-001-0380-y. PMID 11868801. S2CID 38863420.
- ^ a b Zahedi Bialvaei A, Rahbar M, Yousefi M, Asgharzadeh M, Samadi Kafil H (February 2017). "Linezolid: a promising option in the treatment of Gram-positives". The Journal of Antimicrobial Chemotherapy. 72 (2): 354–364. doi: 10.1093/jac/dkw450. PMID 27999068.
- ^ a b c d e f g Brickner SJ (1996). "Oxazolidinone antibacterial agents". Current Pharmaceutical Design. 2 (2): 175–94. doi: 10.2174/1381612802666220921173820. S2CID 252456453. Detailed review of the discovery and development of the whole oxazolidinone class, including information on synthesis and structure-activity relationships.
- ^ European Medicines Agency (2011). "CHP Assessment Report for Xarelto (EMA/CHMP/301607/2011)" (PDF). Archived (PDF) from the original on 30 January 2012. Retrieved 15 March 2012.
- ^ a b c Xu GY, Zhou Y, Xu MC (2006). "A convenient synthesis of antibacterial linezolid from (S)-glyceraldehyde acetonide" (PDF). Chinese Chemical Letters. 17 (3): 302–4. Archived from the original (PDF) on 7 July 2011.
- ^ a b Kaiser CR, Cunico W, Pinheiro AC, de Oliveira AG, Peralta MA, de Souza MV (2007). "Oxazolidinonas: uma nova classe de compostos no combate à tuberculose" [Oxazolidinones: a new class of compounds against tuberculosis] (PDF). Revista Brasileira de Farmácia (in Portuguese). 88 (2): 83–8. Archived from the original (PDF) on 15 May 2012.
- ^ US patent 5837870, Pearlman BA, Perrault WR, Barbachyn MR, et al., "Process to prepare oxazolidinones", issued 1997-03-28 Retrieved on 13 June 2009.
- ^ Lohray BB, Baskaran S, Rao BS, Reddy BY, Rao IN (June 1999). "A short synthesis of oxazolidinone derivatives linezolid and eperezolid: A new class of antibacterials". Tetrahedron Letters. 40 (26): 4855–6. doi: 10.1016/S0040-4039(99)00893-X.
- ^ Perrault WR, Keeler JB, Snyder WC, et al. (25 June 2008). "Convergent green synthesis of linezolid (Zyvox)" Archived 28 July 2011 at the Wayback Machine, in 12th Annual Green Chemistry and Engineering Conference, 24–26 June 2008, New York, NY. Retrieved on 8 June 2009.
- ^ Tsiodras S, Gold HS, Sakoulas G, et al. (July 2001). "Linezolid resistance in a clinical isolate of Staphylococcus aureus". The Lancet. 358 (9277): 207–8. doi: 10.1016/S0140-6736(01)05410-1. ISSN 0140-6736. PMID 11476839. S2CID 27426801.
- ^ Jones RN, Ross JE, Castanheira M, Mendes RE (December 2008). "United States resistance surveillance results for linezolid (LEADER Program for 2007)". Diagnostic Microbiology and Infectious Disease. 62 (4): 416–26. doi: 10.1016/j.diagmicrobio.2008.10.010. PMID 19022153.
- ^ Jones RN, Kohno S, Ono Y, Ross JE, Yanagihara K (June 2009). "ZAAPS International Surveillance Program (2007) for linezolid resistance: results from 5591 Gram-positive clinical isolates in 23 countries". Diagnostic Microbiology and Infectious Disease. 64 (2): 191–201. doi: 10.1016/j.diagmicrobio.2009.03.001. PMID 19500528.
- ^ Hope R, Livermore DM, Brick G, Lillie M, Reynolds R (November 2008). "Non-susceptibility trends among staphylococci from bacteraemias in the UK and Ireland, 2001–06". The Journal of Antimicrobial Chemotherapy. 62 (Suppl 2): ii65–74. doi: 10.1093/jac/dkn353. PMID 18819981.
- ^ Auckland C, Teare L, Cooke F, Kaufmann ME, Warner M, Jones G, et al. (November 2002). "Linezolid-resistant enterococci: report of the first isolates in the United Kingdom". The Journal of Antimicrobial Chemotherapy. 50 (5): 743–6. doi: 10.1093/jac/dkf246. PMID 12407134. S2CID 2468640.
- ^ a b Scheetz MH, Knechtel SA, Malczynski M, Postelnick MJ, Qi C (June 2008). "Increasing Incidence of Linezolid-Intermediate or -Resistant, Vancomycin-Resistant Enterococcus faecium Strains Parallels Increasing Linezolid Consumption". Antimicrobial Agents and Chemotherapy. 52 (6): 2256–9. doi: 10.1128/AAC.00070-08. PMC 2415807. PMID 18391028.
- ^ Schumacher A, Trittler R, Bohnert JA, Kümmerer K, Pagès JM, Kern WV (June 2007). "Intracellular accumulation of linezolid in Escherichia coli, Citrobacter freundii and Enterobacter aerogenes: role of enhanced efflux pump activity and inactivation". Journal of Antimicrobial Chemotherapy. 59 (6): 1261–4. doi: 10.1093/jac/dkl380. PMID 16971414.
- ^ Saager B, Rohde H, Timmerbeil BS, et al. (September 2008). "Molecular characterisation of linezolid resistance in two vancomycin-resistant (VanB) Enterococcus faecium isolates using Pyrosequencing". European Journal of Clinical Microbiology & Infectious Diseases. 27 (9): 873–8. doi: 10.1007/s10096-008-0514-6. ISSN 0934-9723. PMID 18421487. S2CID 1085422.
- ^ Besier S, Ludwig A, Zander J, Brade V, Wichelhaus TA (April 2008). "Linezolid Resistance in Staphylococcus aureus: Gene Dosage Effect, Stability, Fitness Costs, and Cross-Resistances". Antimicrobial Agents and Chemotherapy. 52 (4): 1570–2. doi: 10.1128/AAC.01098-07. PMC 2292563. PMID 18212098.
- ^ Feng J, Lupien A, Gingras H, et al. (May 2009). "Genome sequencing of linezolid-resistant Streptococcus pneumoniae mutants reveals novel mechanisms of resistance". Genome Research. 19 (7): 1214–23. doi: 10.1101/gr.089342.108. ISSN 1088-9051. PMC 2704432. PMID 19351617.
- ^ Lincopan N, de Almeida LM, Elmor de Araújo MR, Mamizuka EM (April 2009). "Linezolid resistance in Staphylococcus epidermidis associated with a G2603T mutation in the 23S rRNA gene". International Journal of Antimicrobial Agents. 34 (3): 281–2. doi: 10.1016/j.ijantimicag.2009.02.023. ISSN 0924-8579. PMID 19376688.
- ^ Liakopoulos A, Neocleous C, Klapsa D, et al. (July 2009). "A T2504A mutation in the 23S rRNA gene responsible for high-level resistance to linezolid of Staphylococcus epidermidis". Journal of Antimicrobial Chemotherapy. 64 (1): 206–7. doi: 10.1093/jac/dkp167. PMID 19429927.
- ^ Slee AM, Wuonola MA, McRipley RJ, et al. (November 1987). "Oxazolidinones, a new class of synthetic antibacterial agents: in vitro and in vivo activities of DuP 105 and DuP 721" (PDF). Antimicrobial Agents and Chemotherapy. 31 (11): 1791–7. doi: 10.1128/AAC.31.11.1791. PMC 175041. PMID 3435127. Archived (PDF) from the original on 11 June 2011.
- ^ Ford CW, Zurenko GE, Barbachyn MR (August 2001). "The discovery of linezolid, the first oxazolidinone antibacterial agent". Current Drug Targets. Infectious Disorders. 1 (2): 181–99. doi: 10.2174/1568005014606099. PMID 12455414.
- ^ "Drug Approval Package: Zyvox". FDA Center for Drug Evaluation and Research. 20 November 2001. Archived from the original on 10 January 2008. Retrieved 17 January 2009. Comprehensive review of the FDA approval process. Includes detailed reviews of the chemistry and pharmacology of linezolid, correspondence between the FDA and Pharmacia & Upjohn, and administrative documents.
- ^ ANVISA (5 June 2000). "Resolução nº 474, de 5 de junho de 2000" [Resolution number 474, of 5 June 2000] (in Portuguese). National Health Surveillance Agency. Archived from the original on 19 July 2011. Retrieved 19 May 2009.
- ^ Irinoda K, Nomura S, Hashimoto M (October 2002). "[Antimicrobial and clinical effect of linezolid (Zyvox), new class of synthetic antibacterial drug]". Nippon Yakurigaku Zasshi (in Japanese). 120 (4): 245–52. doi: 10.1254/fpj.120.245. ISSN 0015-5691. PMID 12425150.
- ^ a b "Canada Approves Marketing Of Zyvoxam (Linezolid) For Gram Positive Infections" (Press release). 8 May 2001. Archived from the original on 28 August 2008. Retrieved 18 May 2009.
- ^ Karlowsky JA, Kelly LJ, Critchley IA, Jones ME, Thornsberry C, Sahm DF (June 2002). "Determining Linezolid's Baseline In Vitro Activity in Canada Using Gram-Positive Clinical Isolates Collected prior to Its National Release" (PDF). Antimicrobial Agents and Chemotherapy. 46 (6): 1989–92. doi: 10.1128/AAC.46.6.1989-1992.2002. ISSN 0066-4804. PMC 127260. PMID 12019122. Archived (PDF) from the original on 29 September 2011.
- ^ "Pharmacia Corporation Reports 17% Increase In Second-Quarter Earnings-Per-Share Driven By 61% Increase In Pharmaceutical Earnings" (Press release). Pharmacia Corporation. 25 July 2001. Archived from the original on 9 May 2012. Retrieved 19 May 2009.
- ^ Livermore DM, Mushtaq S, Warner M, Woodford N (April 2009). "Activity of oxazolidinone TR-700 against linezolid-susceptible and -resistant staphylococci and enterococci". Journal of Antimicrobial Chemotherapy. 63 (4): 713–5. doi: 10.1093/jac/dkp002. ISSN 0305-7453. PMID 19164418.
- ^ Howe RA, Wootton M, Noel AR, Bowker KE, Walsh TR, MacGowan AP (November 2003). "Activity of AZD2563, a Novel Oxazolidinone, against Staphylococcus aureus Strains with Reduced Susceptibility to Vancomycin or Linezolid" (PDF). Antimicrobial Agents and Chemotherapy. 47 (11): 3651–2. doi: 10.1128/AAC.47.11.3651-3652.2003. ISSN 0066-4804. PMC 253812. PMID 14576139. Archived (PDF) from the original on 29 September 2011.
- ^ Kalia V, Miglani R, Purnapatre KP, et al. (April 2009). "Mode of Action of Ranbezolid against Staphylococci and Structural Modeling Studies of Its Interaction with Ribosomes". Antimicrobial Agents and Chemotherapy. 53 (4): 1427–33. doi: 10.1128/AAC.00887-08. ISSN 0066-4804. PMC 2663096. PMID 19075051.
- ^ "Rx 1741". Rib-X Pharmaceuticals. 2009. Archived from the original on 26 February 2009. Retrieved 17 May 2009.
- ^ "Drug Approval Package: Sivextro (tedizolid phosphate) tablets NDA #205435". U.S. Food and Drug Administration (FDA). 16 July 2014. Archived from the original on 6 July 2020. Retrieved 29 March 2024.
- ^ "Drug Approval Package: Sivextro (tedizolid phosphate) injection NDA #205436". U.S. Food and Drug Administration (FDA). 10 October 2014. Archived from the original on 6 July 2020. Retrieved 29 March 2024.
- ^ a b [ needs update]Grau S, Rubio-Terrés C (April 2008). "Pharmacoeconomics of linezolid". Expert Opinion on Pharmacotherapy. 9 (6): 987–1000. doi: 10.1517/14656566.9.6.987. PMID 18377341. S2CID 3005291.
- ^ "Pfizer agrees record fraud fine". BBC News. 2 September 2009. Archived from the original on 8 September 2009. Retrieved 12 September 2009.
- ^ Harris G (2 September 2009). "Pfizer pays $2.3 billion to settle marketing case". The New York Times. Archived from the original on 22 August 2011. Retrieved 12 September 2009.
- ^ a b "Linezolid". Drugs.com. Archived from the original on 29 December 2018. Retrieved 29 December 2018.