 | |
| Clinical data | |
|---|---|
| Other names | TAK-906; ATC-1906 |
| Drug class | Dopamine antagonist |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
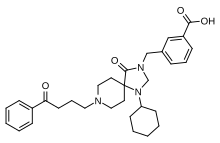
| Formula | C31H39N3O4 |
| Molar mass | 517.670 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
Trazpiroben (developmental code name TAK-906) is a dopamine antagonist drug which was under development for the treatment of gastroparesis. [1] [2] [3] [4] [5] It acts as a peripherally selective dopamine D2 and D3 receptor antagonist. [1] [2] The drug has been found to strongly increase prolactin levels in humans, similarly to other peripherally selective D2 receptor antagonists like domperidone. [5] Clinical development of trazpiroben was discontinued before April 2022. [1] Trazpiroben was originated by Altos Therapeutics and was under development by Takeda Oncology. [1]
References
- ^ a b c d "Trazpiroben - Takeda Oncology - AdisInsight".
- ^ a b Whiting RL, Choppin A, Luehr G, Jasper JR (October 2021). "Preclinical Evaluation of the Effects of Trazpiroben (TAK-906), a Novel, Potent Dopamine D2/D3 Receptor Antagonist for the Management of Gastroparesis". J Pharmacol Exp Ther. 379 (1): 85–95. doi: 10.1124/jpet.121.000698. PMID 34253646. S2CID 235809216.
- ^ Whiting RL, Darpo B, Chen C, Fletcher M, Combs D, Xue H, Stoltz RR (August 2021). "Safety, Pharmacokinetics, and Pharmacodynamics of Trazpiroben (TAK-906), a Novel Selective D2 /D3 Receptor Antagonist: A Phase 1 Randomized, Placebo-Controlled Single- and Multiple-Dose Escalation Study in Healthy Participants". Clin Pharmacol Drug Dev. 10 (8): 927–939. doi: 10.1002/cpdd.906. PMC 8451790. PMID 33462988.
- ^ Kuo B, Scimia C, Dukes G, Zhang W, Gupta S, Chen C, Chuang E, Camilleri M (August 2021). "Randomised clinical trial: safety, pharmacokinetics and pharmacodynamics of trazpiroben (TAK-906), a dopamine D2 /D3 receptor antagonist, in patients with gastroparesis". Aliment Pharmacol Ther. 54 (3): 267–280. doi: 10.1111/apt.16451. PMID 34148244. S2CID 235492049.
- ^ a b Yamaguchi T, Kudou K, Okamoto H, Chen C, Whiting R, Sekino H (December 2021). "Evaluating the Safety, Tolerability, and Disposition of Trazpiroben, a D2 /D3 Receptor Antagonist: Phase I Single- and Multiple-Ascending Dose Studies in Healthy Japanese Participants". Clin Pharmacol Drug Dev. 11 (6): 695–706. doi: 10.1002/cpdd.1057. PMC 9303893. PMID 34967147. S2CID 245567576.
External links