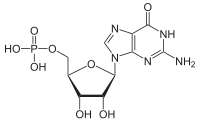
| |

| |
| Names | |
|---|---|
|
IUPAC name
5′-Guanylic acid
| |
|
Systematic IUPAC name
[(2R,3S,4R,5R)-5-(2-Amino-6-oxo-1,6-dihydro-9H-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methyl dihydrogen phosphate | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| Abbreviations | GMP |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.453 |
| E number | E626 (flavour enhancer) |
| MeSH | Guanosine+monophosphate |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| Properties | |
| C10H14N5O8P | |
| Molar mass | 363.223 g·mol−1 |
| Acidity (pKa) | 0.7, 2.4, 6.1, 9.4 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Guanosine monophosphate (GMP), also known as 5′-guanidylic acid or guanylic acid ( conjugate base guanylate), is a nucleotide that is used as a monomer in RNA. It is an ester of phosphoric acid with the nucleoside guanosine. GMP consists of the phosphate group, the pentose sugar ribose, and the nucleobase guanine; hence it is a ribonucleoside monophosphate. Guanosine monophosphate is commercially produced by microbial fermentation. [1]
As an acyl substituent, it takes the form of the prefix guanylyl-.
De novo synthesis
GMP synthesis starts with D-ribose 5′-phosphate, a product of the pentose phosphate pathway. The synthesis proceeds by the gradual formation of the purine ring on carbon-1 of ribose, with CO2, glutamine, glycine, aspartate and one-carbon derivatives of tetrahydrofolate donating various elements towards the building of the ring. [2]
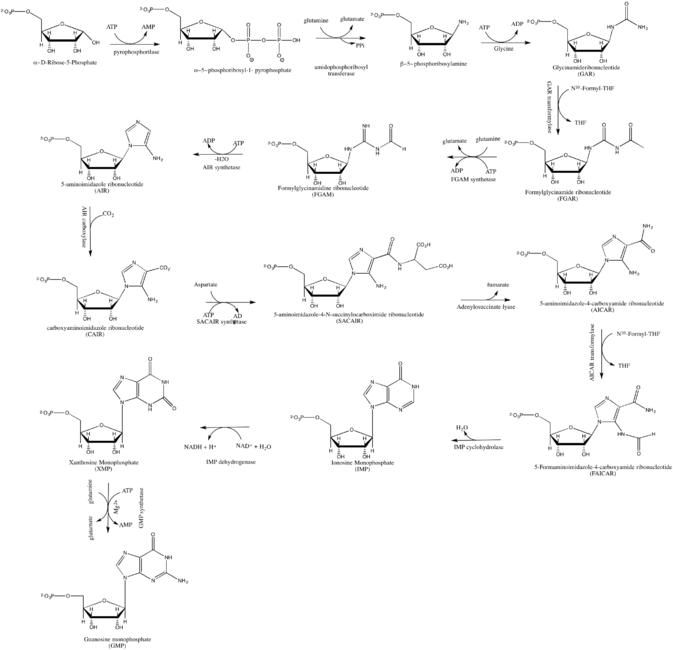
As inhibitor of guanosine monophosphate synthesis in experimental models, the glutamine analogue DON can be used. [3]
cGMP
GMP can also exist as a cyclic structure known as cyclic GMP. Within certain cells the enzyme guanylyl cyclase makes cGMP from GTP.
cGMP plays an important role in mediating hormonal signaling. [2]
Sources
GMP was originally identified as the umami substance in dried shiitake mushroom. The drying process significantly increases GMP content with the breakdown of RNA. It can be found in a number of other mushrooms. [4]
Industrial production is based on fermentation: a bacterium converts sugars into AICA ribonucleotide, which is then converted chemically to GMP. [5] Tapioca starch is a possible sugar source. [6]
Food additive
Guanosine monophosphate is known as E number reference E626. [7] In the form of its salts, such as disodium guanylate ( E627), dipotassium guanylate (E628) and calcium guanylate (E629), are food additives used as flavor enhancers to provide the umami taste. [7] It is often used in synergy with disodium inosinate; the combination is known as disodium 5′-ribonucleotides. Disodium guanylate is often found in instant noodles, potato chips and snacks, savoury rice, tinned vegetables, cured meats, and packet soup.
As it is a fairly expensive additive, it is usually not used independently of glutamic acid or monosodium glutamate (MSG), which also contribute umami. If inosinate and guanylate salts are present in a list of ingredients but MSG does not appear to be, the glutamic acid is likely provided as part of another ingredient, such as a processed soy protein complex (hydrolyzed soy protein), autolyzed yeast, or soy sauce.
See also
References
- ^ "The Vegetarian Resource Group Blog". www.vrg.org. Retrieved 25 April 2018.
- ^ a b Voet, Donald; Voet, Judith G. (2012). Biochemistry. USA: John Wiley & Sons Inc. pp. 1107–1109. ISBN 978-0-470-57095-1.
- ^ Ahluwalia GS et al. Metabolism and action of amino acid analog anti-cancer agents ”, in Pharmac. Ther. (1990) 46: 243-271
- ^ Kurihara, K (2015). "Umami the Fifth Basic Taste: History of Studies on Receptor Mechanisms and Role as a Food Flavor". BioMed Research International. 2015: 189402. doi: 10.1155/2015/189402. PMC 4515277. PMID 26247011.
- ^ Kinoshita, Kazumoto; Shiro, Teruo; Yamazaki, Akihiro; Kumashiro, Izumi; Takenishi, Tadao; Tsunoda, Toshinao (July 1967). "Industrial production of disodium 5?-guanylate". Biotechnology and Bioengineering. 9 (3): 329–342. doi: 10.1002/bit.260090306. S2CID 84216811.
- ^ Conn, Helen (1 February 1992). ""Umami": The Fifth Basic Taste". Nutrition & Food Science. 92 (2): 21–23. doi: 10.1108/EUM0000000000953.
- ^ a b "Additive categories | CEFF". www.ceff.info. Retrieved 2021-11-30.