| |
| |

| |
| Names | |
|---|---|
|
Preferred IUPAC name
(2E)-3-Phenylprop-2-enal | |
Other names
| |
| Identifiers | |
3D model (
JSmol)
|
|
| 3DMet | |
| 1071571 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.111.079 |
| EC Number |
|
| KEGG | |
PubChem
CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C9H8O | |
| Molar mass | 132.16 g/mol |
| Appearance | Yellow oil |
| Odor | Pungent, cinnamon-like |
| Density | 1.0497 g/mL |
| Melting point | −7.5 °C (18.5 °F; 265.6 K) |
| Boiling point | 248 °C (478 °F; 521 K) |
| Slightly soluble | |
| Solubility |
|
| −7.48×10−5 cm3/mol | |
Refractive index (nD)
|
1.6195 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H317, H319, H335 | |
| P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P333+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 71 °C (160 °F; 344 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (
median dose)
|
3400 mg/kg (rat, oral) |
| Related compounds | |
Related compounds
|
Cinnamic acid |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
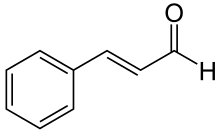
Cinnamaldehyde is an organic compound with the formula or C₆H₅CH=CHCHO. Occurring naturally as predominantly the trans (E) isomer, it gives cinnamon its flavor and odor. [1] It is a phenylpropanoid that is naturally synthesized by the shikimate pathway. [2] This pale yellow, viscous liquid occurs in the bark of cinnamon trees and other species of the genus Cinnamomum. The essential oil of cinnamon bark is about 90% cinnamaldehyde. [3] Cinnamaldehyde decomposes to styrene because of oxidation as a result of bad storage or transport conditions. Styrene especially forms in high humidity and high temperatures. This is the reason why cinnamon contains small amounts of styrene. [4]
Structure and synthesis
Cinnamaldehyde was isolated from cinnamon essential oil in 1834 by Jean-Baptiste Dumas and Eugène-Melchior Péligot [5] and synthesized in the laboratory by the Italian chemist Luigi Chiozza in 1854. [6]
The natural product is trans-cinnamaldehyde. The molecule consists of a benzene ring attached to an unsaturated aldehyde. As such, the molecule can be viewed as a derivative of acrolein. Its color is due to the π → π* transition: increased conjugation in comparison with acrolein shifts this band towards the visible. [7]
Biosynthesis

Cinnamaldehyde occurs widely, and closely related compounds give rise to lignin. All such compounds are biosynthesized starting from phenylalanine, which undergoes conversion. [8]
The biosynthesis of cinnamaldehyde begins with deamination of L-phenylalanine into cinnamic acid by the action of phenylalanine ammonia lyase (PAL). [9] [10] PAL catalyzes this reaction by a non-oxidative deamination. This deamination relies on the MIO prosthetic group of PAL. [11] PAL gives rise to trans-cinnamic acid. In the second step, 4-coumarate–CoA ligase (4CL) converts cinnamic acid to cinnamoyl-CoA by an acid– thiol ligation. [9] 4CL uses ATP to catalyze the formation of cinnamoyl-CoA. [12] 4CL effects this reaction in two steps. [13] 4CL forms a hydroxycinnamate–AMP anhydride, followed by a nucleophile attack on the carbonyl of the acyl adenylate. [14] Finally, Cinnamoyl-CoA is reduced by NADPH catalyzed by CCR (cinnamoyl-CoA reductase) to form cinnamaldehyde. [9] [15]
Preparation
Several methods of laboratory synthesis exist, but cinnamaldehyde is most economically obtained from the steam distillation of the oil of cinnamon bark. The compound can be prepared from related compounds such as cinnamyl alcohol, (the alcohol form of cinnamaldehyde), but the first synthesis from unrelated compounds was the aldol condensation of benzaldehyde and acetaldehyde; this process was patented by Henry Richmond on November 7, 1950. [16]
Applications
As a flavorant
The most obvious application for cinnamaldehyde is as flavoring in chewing gum, ice cream, candy, e-liquid and beverages; use levels range from 9 to 4,900 parts per million ( ppm) (that is, less than 0.5%). It is also used in some perfumes of natural, sweet, or fruity scents. Almond, apricot, butterscotch, and other aromas may partially employ the compound for their pleasant smells. Cinnamaldehyde can be used as a food adulterant; powdered beechnut husk aromatized with cinnamaldehyde can be marketed as powdered cinnamon. [17] Some breakfast cereals contain as much as 187 ppm cinnamaldehyde. [18]
As an agrichemical
Cinnamaldehyde has been tested as a safe and effective insecticide against mosquito larvae. [19] A concentration of 29 ppm of cinnamaldehyde kills half of Aedes aegypti mosquito larvae in 24 hours. [20] Trans-cinnamaldehyde works as a potent fumigant and practical repellant for adult mosquitos. [21] It also has antibacterial and antifungal properties. [22] [23]
Miscellaneous uses
Cinnamaldehyde is a corrosion inhibitor for steel and other alloys. It is believed to form a protective film on the metal surface. [24]
Derivatives
Numerous derivatives of cinnamaldehyde are commercially useful. Dihydrocinnamyl alcohol (3-phenylpropanol) occurs naturally but is produced by double hydrogenation of cinnamaldehyde. It has the fragrances of hyacinth and lilac. Cinnamyl alcohol similarly occurs naturally and has the odor of lilac but can be also produced starting from cinnamaldehyde. [25] Dihydrocinnamaldehyde is produced by the selective hydrogenation of the alkene subunit. α-Amylcinnamaldehyde and α-hexylcinnamaldehyde are important commercial fragrances, but they are not prepared from cinnamaldehyde. [17] Hydrogenation of cinnamaldehyde, if directed to the alkene, gives hydrocinnamaldehyde.
Toxicology
Cinnamaldehyde is used in agriculture because of its low toxicity, but it is a skin irritant. [26] Cinnamaldehyde may cause allergic contact stomatitis in sensitised individuals, however allergy to the compound is believed to be uncommon. [27]
DNA repair
Cinnamaldehyde is a dietary antimutagen that effectively inhibits both induced and spontaneous mutations. [28] Experimental evidence indicates that cinnamaldehyde induces a type of DNA damage in the bacterium Escherichia coli and in human cells that elicits recombinational DNA repair that then reduces spontaneous mutations. [28] [29] In mice, X-ray induced chromosome aberrations were reduced when cinnamaldehyde was given orally to the mice after X-ray irradiation, [30] perhaps due to cinnamaldehyde stimulated DNA repair.
References
- ^ "Cinnamon". Transport Information Service. Gesamtverband der Deutschen Versicherungswirtschaft e.V. Retrieved 2007-10-23.
- ^ Gutzeit, Herwig (2014). Plant Natural Products: Synthesis, Biological Functions and Practical Applications. Wiley. pp. 19–21. ISBN 978-3-527-33230-4.
- ^ PubChem. "Cinnamaldehyde". pubchem.ncbi.nlm.nih.gov. Retrieved 2019-10-18.
- ^ "High daily intakes of cinnamon: Health risk cannot be ruled out" (PDF). Federal Institute for Risk Assessment (BfR). 18 August 2006. Archived (PDF) from the original on 7 March 2022. Retrieved 20 May 2022.
- ^ Dumas, J.; Péligot, E. (1834). "Recherches de Chimie organique. — Sur l'Huile de Cannelle, l'Acide hippurique et l'Acide sébacique" [Organic chemistry research – On cinnamon oil, hippuric acid and sebacic acid]. Annales de Chimie et de Physique (in French). 57: 305–334.
- ^ Chiozza, L. (1856). "Sur la production artificielle de l'essence de cannelle" [On the artificial production of cinnamon oil]. Comptes Rendus (in French). 42: 222–227.
- ^ Inuzuka, Kozo (1961). "π Electronic structure of cinnamaldehyde". Bulletin of the Chemical Society of Japan. 34 (11): 1557–60. doi: 10.1246/bcsj.34.1557.
- ^ Boerjan, Wout; Ralph, John; Baucher, Marie (2003). "Ligninbiosynthesis". Annual Review of Plant Biology. 54: 519–546. doi: 10.1146/annurev.arplant.54.031902.134938. PMID 14503002.
- ^ a b c Bang, Hyun-bae; Lee, Yoon-hyeok; Kim, Sun-chang; Sung, Chang-keun; Jeong, Ki-jun (2016-01-19). "Metabolic engineering of Escherichia coli for the production of cinnamaldehyde". Microbial Cell Factories. 15 (1): 16. doi: 10.1186/s12934-016-0415-9. ISSN 1475-2859. PMC 4719340. PMID 26785776.
- ^ Koukol, J.; Conn, E. E. (1961-10-01). "The metabolism of aromatic compounds in higher plants. IV. Purification and properties of the phenylalanine deaminase of Hordeum vulgare". The Journal of Biological Chemistry. 236 (10): 2692–2698. doi: 10.1016/S0021-9258(19)61721-7. ISSN 0021-9258. PMID 14458851.
- ^ Kong, Jian-Qiang (2015-07-20). "Phenylalanine ammonia-lyase, a key component used for phenylpropanoids production by metabolic engineering". RSC Advances. 5 (77): 62587–62603. Bibcode: 2015RSCAd...562587K. doi: 10.1039/C5RA08196C. ISSN 2046-2069.
- ^ Beuerle, Till; Pichersky, Eran (2002-03-15). "Enzymatic Synthesis and Purification of Aromatic Coenzyme A Esters". Analytical Biochemistry. 302 (2): 305–312. doi: 10.1006/abio.2001.5574. PMID 11878812.
- ^ Allina, Sandra M.; Pri-Hadash, Aviva; Theilmann, David A.; Ellis, Brian E.; Douglas, Carl J. (1998-02-01). "4-Coumarate:Coenzyme A Ligase in Hybrid Poplar". Plant Physiology. 116 (2): 743–754. doi: 10.1104/pp.116.2.743. ISSN 0032-0889. PMC 35134. PMID 9489021.
- ^ Li, Zhi; Nair, Satish K. (2015-11-03). "Structural Basis for Specificity and Flexibility in a Plant 4-Coumarate:CoA Ligase". Structure. 23 (11): 2032–2042. doi: 10.1016/j.str.2015.08.012. ISSN 1878-4186. PMID 26412334.
- ^ Wengenmayer, Herta; Ebel, Jurgen; Grisebach, Hans (1976). "Enzymic Synthesis of Lignin Precursors. Purification and Properties of a Cinnamoyl-CoA:NADPH Reductase from Cell Suspension Cultures of Soybean (Glycine max)". European Journal of Biochemistry. 65 (2): 529–536. doi: 10.1111/j.1432-1033.1976.tb10370.x. ISSN 0014-2956. PMID 7454.
- ^ Richmond, H. Preparation of Cinnamaldehyde. US Patent Application 2529186, November 7, 1950.
- ^ a b Fahlbusch, Karl-Georg; Hammerschmidt, Franz-Josef; Panten, Johannes; Pickenhagen, Wilhelm; Schatkowski, Dietmar; Bauer, Kurt; Garbe, Dorothea; Surburg, Horst (2003). "Flavors and Fragrances". Ullmann's Encyclopedia of Industrial Chemistry. doi: 10.1002/14356007.a11_141. ISBN 978-3-527-30673-2.
- ^ Friedman, M.; Kozuekue, N.; Harden, L. A. (2000). "Cinnamaldehyde content in foods determined by gas chromatography-mass spectrometry". Journal of Agricultural and Food Chemistry. 48 (11): 5702–5709. doi: 10.1021/jf000585g. PMID 11087542.
- ^ Dick-Pfaff, Cornelia (July 19, 2004). "Wohlriechender Mückentod" (in German).
-
^ Cheng, Sen-Sung; Liu, Ju-Yun; Tsai, Kun-Hsien; Chen, Wei-June; Chang, Shang-Tzen (2004). "Chemical Composition and Mosquito Larvicidal Activity of Essential Oils from Leaves of Different Cinnamomum osmophloeum Provenances". Journal of Agricultural and Food Chemistry. 52 (14): 4395–4400.
doi:
10.1021/jf0497152.
PMID
15237942.
- "Cinnamon Oil Kills Mosquitoes". ScienceDaily (Press release). July 16, 2004.
- ^ Ma, W.-B.; Feng, J.-T.; Jiang, Z.-L.; Zhang, X. (2014). "Fumigant Activity of 6 Selected Essential Oil Compounds and Combined Effect of Methyl Salicylate And trans-Cinnamaldehyde Against Culex pipiens pallens". Journal of the American Mosquito Control Association. 30 (3): 199–203. doi: 10.2987/14-6412R.1. PMID 25843095. S2CID 36621630.
- ^ Vasconcelos, N. G.; Croda, J.; Simionatto, S. (July 2018). "Antibacterial mechanisms of cinnamon and its constituents: A review". Microbial Pathogenesis. 120: 198–203. doi: 10.1016/j.micpath.2018.04.036. ISSN 1096-1208. PMID 29702210.
- ^ Shreaz, Sheikh; Wani, Waseem A.; Behbehani, Jawad M.; Raja, Vaseem; Irshad, Md; Karched, Maribasappa; Ali, Intzar; Siddiqi, Weqar A.; Hun, Lee Ting (July 2016). "Cinnamaldehyde and its derivatives, a novel class of antifungal agents". Fitoterapia. 112: 116–131. doi: 10.1016/j.fitote.2016.05.016. ISSN 1873-6971. PMID 27259370.
- ^ Cabello, Gema; Funkhouser, Gary P.; Cassidy, Juanita; Kiser, Chad E.; Lane, Jim; Cuesta, Angel (2013-05-01). "CO and trans-cinnamaldehyde as corrosion inhibitors of I825, L80-13Cr and N80 alloys in concentrated HCl solutions at high pressure and temperature". Electrochimica Acta. 97: 1–9. doi: 10.1016/j.electacta.2013.03.011. hdl: 2164/2891. ISSN 0013-4686.
- ^ Zucca, P.; Littarru, M.; Rescigno, A.; Sanjust, E. (2009). "Cofactor recycling for selective enzymatic biotransformation of cinnamaldehyde to cinnamyl alcohol". Bioscience, Biotechnology, and Biochemistry. 73 (5): 1224–1226. doi: 10.1271/bbb.90025. PMID 19420690. S2CID 28741979.
- ^ Olsen, R. V.; Andersen, H. H.; Møller, H. G.; Eskelund, P. W.; Arendt-Nielsen, L (2014). "Somatosensory and vasomotor manifestations of individual and combined stimulation of TRPM8 and TRPA1 using topical L-menthol and trans-cinnamaldehyde in healthy volunteers". European Journal of Pain. 18 (9): 1333–42. doi: 10.1002/j.1532-2149.2014.494.x. PMID 24664788. S2CID 34286049.
- ^ Isaac-Renton, Megan; Li, Monica Kayi; Parsons, Laurie M. (May 2015). "Cinnamon spice and everything not nice: many features of intraoral allergy to cinnamic aldehyde". Dermatitis: Contact, Atopic, Occupational, Drug. 26 (3): 116–121. doi: 10.1097/DER.0000000000000112. ISSN 2162-5220. PMID 25984687.
- ^ a b Shaughnessy, D. T.; Schaaper, R. M.; Umbach, D. M.; Demarini, D. M. (2006). "Inhibition of spontaneous mutagenesis by vanillin and cinnamaldehyde in Escherichia coli: Dependence on recombinational repair". Mutation Research. 602 (1–2): 54–64. doi: 10.1016/j.mrfmmm.2006.08.006. PMC 2099251. PMID 16999979.
- ^ King, A. A.; Shaughnessy, D. T.; Mure, K.; Leszczynska, J.; Ward, W. O.; Umbach, D. M.; Xu, Z.; Ducharme, D.; Taylor, J. A.; Demarini, D. M.; Klein, C. B. (2007). "Antimutagenicity of cinnamaldehyde and vanillin in human cells: Global gene expression and possible role of DNA damage and repair". Mutation Research. 616 (1–2): 60–69. doi: 10.1016/j.mrfmmm.2006.11.022. PMC 1955325. PMID 17178418.
- ^ Sasaki, Y. F.; Ohta, T.; Imanishi, H.; Watanabe, M.; Matsumoto, K.; Kato, T.; Shirasu, Y. (1990). "Suppressing effects of vanillin, cinnamaldehyde, and anisaldehyde on chromosome aberrations induced by X-rays in mice". Mutation Research. 243 (4): 299–302. doi: 10.1016/0165-7992(90)90146-b. PMID 2325694.

