This article may be too technical for most readers to understand. (January 2019) |
| Proteasome endopeptidase complex | |||||||||
|---|---|---|---|---|---|---|---|---|---|
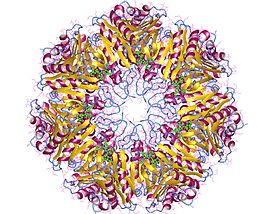 Proteasome core particle, di-heptamer, Archaea | |||||||||
| Identifiers | |||||||||
| EC no. | 3.4.25.1 | ||||||||
| CAS no. | 140879-24-9 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Proteasome endopeptidase complex ( EC 3.4.25.1, ingensin, macropain, multicatalytic endopeptidase complex, prosome, multicatalytic proteinase (complex), MCP, proteasome, large multicatalytic protease, proteasome organelle, alkaline protease, 26S protease, tricorn proteinase, tricorn protease) is an enzyme. [1] [2] [3] [4] This enzyme catalyses the following chemical reaction
- Cleavage of peptide bonds with very broad specificity
This 20-S protein is composed of 28 subunits arranged in four rings of seven.
References
- ^ Seemüller E, Lupas A, Stock D, Löwe J, Huber R, Baumeister W (April 1995). "Proteasome from Thermoplasma acidophilum: a threonine protease". Science. 268 (5210): 579–82. Bibcode: 1995Sci...268..579S. doi: 10.1126/science.7725107. PMID 7725107.
- ^ Coux O, Tanaka K, Goldberg AL (1996). "Structure and functions of the 20S and 26S proteasomes". Annual Review of Biochemistry. 65: 801–47. doi: 10.1146/annurev.bi.65.070196.004101. PMID 8811196.
- ^ Groll M, Ditzel L, Löwe J, Stock D, Bochtler M, Bartunik HD, Huber R (April 1997). "Structure of 20S proteasome from yeast at 2.4 A resolution". Nature. 386 (6624): 463–71. Bibcode: 1997Natur.386..463G. doi: 10.1038/386463a0. PMID 9087403. S2CID 4261663.
- ^ Dick TP, Nussbaum AK, Deeg M, Heinemeyer W, Groll M, Schirle M, Keilholz W, Stevanović S, Wolf DH, Huber R, Rammensee HG, Schild H (October 1998). "Contribution of proteasomal beta-subunits to the cleavage of peptide substrates analyzed with yeast mutants". The Journal of Biological Chemistry. 273 (40): 25637–46. doi: 10.1074/jbc.273.40.25637. PMID 9748229.
External links
- Proteasome+endopeptidase+complex at the U.S. National Library of Medicine Medical Subject Headings (MeSH)