(Redirected from
Cesium superoxide)
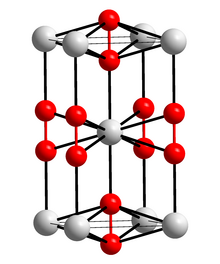 Caesium cations, Cs+ Superoxide anions, O−2 | |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| CsO2 | |
| Molar mass | 164.903 g·mol−1 |
| Appearance | Yellow to orange solid [1] |
| Density | 3.77 g/cm3 [1] |
| Melting point | 600 °C [2] |
| reacts | |
| Related compounds | |
Other
anions
|
|
Other
cations
|
|
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Caesium superoxide is a chemical compound with the chemical formula CsO2. It consists of caesium cations Cs+ and superoxide anions O−2. It is an orange solid.
Preparation
Burning caesium in excess oxygen will produce caesium superoxide. [2]
- Cs + O2 → CsO2
Properties
Caesium superoxide's crystal structure is same as calcium carbide. It contains direct oxygen-oxygen bonding. [2]
It reacts with water to form hydrogen peroxide and caesium hydroxide. [2]
- 2 CsO2 + 2 H2O → O2↑ + H2O2 + 2 CsOH
Heating to approximately 400 °C induces thermal decomposition to caesium peroxide. [3]
The standard enthalpy of formation ΔHf0 of caesium superoxide is −295 kJ/mol. [4]
Caesium superoxide reacts with ozone to form caesium ozonide. [2]
- CsO2 + O3 → CsO3 + O2
References
- ^ a b Caesiumhyperoxid bei webelements.com.
- ^ a b c d e Holleman, Arnold (2007). Lehrbuch der anorganischen Chemie (in German). BerlinNew York: de Gruyter. ISBN 978-3-11-017770-1. OCLC 180963521.
- ^ Berardinelli, S. P.; Kraus, D. L. (1974-01-01). "Thermal decomposition of the higher oxides of cesium in the temperature range 320-500.deg". Inorganic Chemistry. 13 (1). American Chemical Society (ACS): 189–191. doi: 10.1021/ic50131a037. ISSN 0020-1669.
- ^ Holleman, Arnold (2017). Anorganische ChemienBand 1 (in German). Berlin: de Gruyter. ISBN 978-3-11-049585-0. OCLC 968134975.