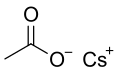
 Structural formula
| |||
 Unit cell of anhydrous caesium acetate.
| |||
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Caesium acetate | |||
| Other names
Cesium acetate
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.020.226 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H3CsO2 | |||
| Molar mass | 191.949 g/mol | ||
| Appearance | colourless, hygroscopic | ||
| Density | 2.423 g/cm3, solid | ||
| Melting point | 194 °C (381 °F; 467 K) | ||
| Boiling point | 945 °C (1,733 °F; 1,218 K) | ||
| 945.1 g/100 g (−2.5 °C) 1345.5 g/100 ml (88.5 °C) | |||
| Structure [2] | |||
| Primitve hexagonal | |||
| P6/m, No. 175 | |||
a = 1488.0 pm, c = 397.65 pm
[2]
| |||
Lattice volume (V)
|
76.542 cm3·mol−1 | ||
Formula units (Z)
|
6 | ||
| Hazards | |||
| Flash point | Non-flammable | ||
| Related compounds | |||
Other
anions
|
Caesium formate | ||
Other
cations
|
Lithium acetate Sodium acetate Potassium acetate Rubidium acetate | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Caesium acetate or cesium acetate is an ionic caesium compound with the molecular formula CH3COOCs. It is a white solid that may be formed by the reaction of caesium hydroxide or caesium carbonate with acetic acid. [3]
Uses
It is used in organic synthesis. One example is in the Perkin synthesis: the formation of unsaturated cinnamic-type acids by the condensation of aromatic aldehydes with fatty acids. Replacement of the commonly used sodium acetate with caesium acetate has been shown to improve yields by up to 10 times. [3] [4]
It is often used to invert secondary alcohols, first by direct SN2 substitution of the hydroxyl group with acetate, which is then converted back to a hydroxyl group. [3]
Caesium acetate is occasionally used instead of caesium formate in petroleum drilling fluids.[ citation needed]
References
- ^ Weast, Robert C., ed. (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. B-91. ISBN 0-8493-0462-8..
- ^ a b Lossin, Adalbert; Meyer, Gerd (1993). "Kristallstruktur von Caesiumacetat, Cs(CH3COO)". Zeitschrift für Anorganische und Allgemeine Chemie. 619 (8): 1462–1464. doi: 10.1002/zaac.19936190823.
- ^ a b c Yode, Ryan (2015), "Cesium Acetate", Encyclopedia of Reagents for Organic Synthesis, John Wiley & Sons, pp. 1–11, doi: 10.1002/047084289x.rn01845, ISBN 978-0-470-84289-8, retrieved 2020-07-21
- ^ Koepp, E.; Vögtle, F. (1987), "Perkin-Synthese mit Cäsiumacetat", Synthesis, 1987 (2): 177–179, doi: 10.1055/s-1987-27880.
Further reading
- Torisawa, Yasuhiro; Okabe, Hiromitsu; Ikegami, Shiro (1984), "Efficient Inversions of Secondary Alcohols using Cesium Acetate and 18-Crown-6", Chem. Lett., 13 (9): 1555–56, doi: 10.1246/cl.1984.1555.

