| This is the
talk page for discussing
WikiProject Chemistry and anything related to its purposes and tasks. |
|
|
Archives:
1,
2,
3,
4,
5,
6,
7,
8,
9,
10,
11,
12,
13,
14,
15,
16,
17,
18,
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39,
40,
41,
42,
43,
44,
45,
46,
47,
48,
49,
50,
51,
52,
53,
54Auto-archiving period: 90 days
|
|
| Chemistry Project‑class | ||||||
| |||||||
| Article alerts |
|---|
|
Articles for deletion
Proposed deletions
Categories for discussion
Templates for discussion
Featured article candidates
Good article nominees
Good article reassessments
Requests for comments Requested moves
Articles to be merged
Articles to be split
Articles for creation
|
For Tahoka Formation, how do I write the dot between the water and the rest of the formula? Jo-Jo Eumerus ( talk) 09:51, 28 February 2024 (UTC)
- Don't use chem, it has ugly output, instead use chem2. Chem2 converted the * to a centre-dot · . However I prefer to use • as it is more visible. Graeme Bartlett ( talk) 10:51, 28 February 2024 (UTC)
I propose splitting X-ray diffraction out of X-ray crystallography, discussion started at Talk:X-ray crystallography#Split x-ray diffraction and crystallography. The two are not the same, and there are many areas of XRD where the focus is not on detailed determination of atomic positions. Examples are powder diffraction where comparison is made to known samples, SAXS and many more. There are many areas/pages where it is relevant to say "use XRD" but wrong to say use "X-ray crystallography This would also help to improve the current rambling X-ray crystallography page. Comments to the X-ray crystallography talk page please. Ldm1954 ( talk) 08:40, 14 April 2024 (UTC)
Is there a reason the definition of chemical element seems to vary according to the date you look at the article ? Currently it is defined as a monotypical atomic substance, but a few years ago it was the "species of atom with same atomic number" that was used. I wonder why it's not even mentioned in the article now, the Goldbook cited in the introduction gives the two. frwiki, dewiki, itwiki uses the atomic one, if I'm not mistaken.
The first one seems more complicated and actually a bit dated/historical, the second one is more convenient. The first one is «
simple substance
![]() » on Wikidata and there are several wikis that have articles about that. Maybe we should sort the interwikis and move articles to the right item ?
TomT0m (
talk)
19:26, 22 April 2024 (UTC)
» on Wikidata and there are several wikis that have articles about that. Maybe we should sort the interwikis and move articles to the right item ?
TomT0m (
talk)
19:26, 22 April 2024 (UTC)
Nucleobase is the heterocyclic variable group, nucleoside is nucleobase+sugar, nucleotide is nucleotide+phosphate. Is there a term for the parent category containing all of those? I'm working on organizing some synthetic-biology chemical pages, for which we don't have very many of any one of those three but we have some and they do form a coherent topic set. DMacks ( talk) 16:39, 1 May 2024 (UTC)
- Seems like a very worthy project. It looks like we have a full complement of articles on the main natural nucleosides, although some articles might lack a basic ref to a standard biochem textbook. Is there a good online biochem textbook that is not behind a paywall? It would be appropriate to have articles for each of the main natural nucleotide monophosphates (and eventually all of the diphosphates and triphosphates). I am trying to think of what core info we might want for these articles because otherwise their physical properties are probably comparable and unremarkable. Maybe their tautomers (for the nucleobase articles), base-pairing partners, their susceptibility to modification (N- or O-methylation as in epigenetics). Good luck. -- Smokefoot ( talk) 16:18, 2 May 2024 (UTC)
I am asking for the support of this community to move the following page from drafts to articles: /info/en/?search=Draft_talk:Baltic_Chemistry_Olympiad. That will help with increasing the coverage of chemistry competitions.
Wikipedia covers several major competitions, such as the International Chemistry Olympiad and USNCO. However, there should be more pages to raise awareness. I have written a detailed overview of the Baltic Chemistry Olympiad and Competitions. It is one of the oldest international competitions, which over 30 years generated over 180 problems for training students and popularising chemistry.
Olunet ( talk) 10:04, 11 May 2024 (UTC)
- Wikipedia:Articles for deletion/Baltic Chemistry Olympiad was why it was deleted. Graeme Bartlett ( talk) 10:30, 11 May 2024 (UTC)
I just found ninovium article. I think this article should either be merged with Victor Ninov or with oganesson. Could somebody please take a look? ReyHahn ( talk) 13:26, 15 May 2024 (UTC)
- It is probably better merged with Ninov's article, since the fraud has become a pretty defining event of his career; for oganesson, it would be a history subsection at most.
- Certainly doubt it merits its own page, especially under the current name - and especially one that introduces it as "a synthetic element", and says things like "ninovium's existence as a valid element remains unsubstantiated, and its characteristics remain uncertain" - which makes me wonder if the page's author understands that the claimed "ninovium" necessarily is oganesson. The whole incident could warrant something like element 118 falsification scandal, I suppose?
- I would say redirect "ninovium" to oganesson#Unconfirmed discovery claims, while integrating the salvageable parts of the existing article into Ninov's? Fishsicles ( talk) 18:28, 7 June 2024 (UTC)
Hi all, I am a new user I was refining Draft:Fausto Calderazzo and I have moved some of the content in the article Migratory insertion. I have also enlarged the article Insertion reaction. Could you please provide some feedback? Thank you in advance.-- Diegoriccio98 ( talk) 13:14, 17 May 2024 (UTC)
- It is valuable to mention the old work on RMn(CO)5 but not much more. Wiki articles rarely describe how experiments were conducted. And we do not explain very much (
WP:NOTTEXTBOOK), Instead our articles focus on facts, the results of experiments, not how they are designed (we want the "baby, not the delivery"). You may consider writing an article about MeMn(CO)5 or its acetyl derivative if you feel the urge to present the details for that work. Migratory insertion is an important step in several catalytic cycles (zero involve Mn), so we want the article to remain general. And a lot of work has been done since the 1960's, some summarized in Hartwig's textbook.--
Smokefoot (
talk)
14:59, 17 May 2024 (UTC)
- The expression "feel the urge" is unkind. I am a student who was studying the history of chemistry in his free time, I found this platform quite "rude" so far despite my goodwill and some recent development add more complication. Some replies I have received so far are too generic to be useful (not yours), so I think I should stop it here. Thank you all and see you maybe in the future.-- Diegoriccio98 ( talk) 15:58, 17 May 2024 (UTC)
Would I be correct in my belief that the phosphate groups in DNA and RNA are chiral at P? Project Osprey ( talk) 19:44, 26 May 2024 (UTC)
- The phosphate substructures would not be chiral. Line bond drawings for the DNA backbone are drawn asymmetrically, with one of these oxygen atoms as part of a P=O double bond, but bond resonance renders the substructure to be symmetrical. I'm sure there are a multitude of ways that the symmetry can be broken. One off the top of my head would be if one of the oxygen atoms, not part of a phosphodiester bond, is a rarer isotope, O17 or O18. ―
Syn
path
00:00, 27 May 2024 (UTC)
- Always fun to jump onto such arcane questions with imperfect knowledge: My understanding is that since DNA/RNA are chiral so the two terminal P-O's are nonequivalent. Every O is nonequivalent. Its asymmetric (not just dissymmetric). This comment is related to the problematic concept of "chiral center" and all that. IMHO. Now about chiral phosphates, that topic was a "thing" several decades ago with the prep of ROP16O17O18O2- by Knowles at Harvard (
https://www.annualreviews.org/content/journals/10.1146/annurev.bi.49.070180.004305). There is another P-chiral Knowles, who got a Nobel for chiral phosphines. --
Smokefoot (
talk)
00:23, 27 May 2024 (UTC)
- Yes, a bit of sloppy language on my part. I meant the phosphate P atoms in typical DNA/RNA polymer are not stereogenic centres. Side note, I was left in a bit of awe when I first learned that you can actually synthesize chiral phosphates reliably to query enzymatic reaction mechanisms. Though, I only recall a MeOP16O18OS2- system. ― Syn path 00:51, 27 May 2024 (UTC)
- Always fun to jump onto such arcane questions with imperfect knowledge: My understanding is that since DNA/RNA are chiral so the two terminal P-O's are nonequivalent. Every O is nonequivalent. Its asymmetric (not just dissymmetric). This comment is related to the problematic concept of "chiral center" and all that. IMHO. Now about chiral phosphates, that topic was a "thing" several decades ago with the prep of ROP16O17O18O2- by Knowles at Harvard (
https://www.annualreviews.org/content/journals/10.1146/annurev.bi.49.070180.004305). There is another P-chiral Knowles, who got a Nobel for chiral phosphines. --
Smokefoot (
talk)
00:23, 27 May 2024 (UTC)
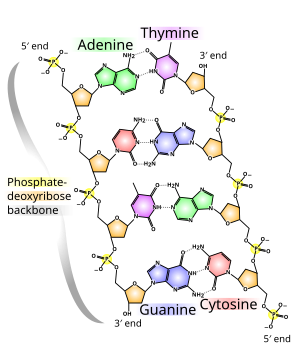
Thank you both. I've added a picture to try and help explain myself. The organophosphate groups here consist of a P bonded to 4 different groups, arraigned in rough tetrahedron O=P(O-)(OR1)(OR2). If that P was a C it would undoubtedly be chiral (obviously allowing for the =O being some other comparable group that satisfies the bond order). Synpath raises the issue of resonance but that isn't really a description of the ground state, I think at most that would allow the molecule to convert between 2 or more forms, all of which would be chiral. I'm also not sure how much phosphate is able to engage in resonance anyway, the bonding around P is exceedingly complex and I barely understand it - but to my mind the process of resonance shares themes with SN2 attack (no change in overall bond number, even at the transition state). Hydrolysis reactions are incredibly slow in phosphates, the uncatalyzed hydrolysis of dimethyl phosphate proceeds at about 1 × 10−15 s−1 [1] that's a half life of thousands of years.
- Maybe others can explain better. I recommend omitting resonance, a bonding concept that just blurs the picture. All atoms in an asymmetric entity are nonequivalent (diastereotopic). Nonequivalence is usually the key question, i.e. every atom and every single bond reacts at a distinct rate (except for coincidences), and every single atom shows a separate NMR resonance. In DNA/RNA the chirality originates in the sugar residue, which dictates the spirality of the helix. If that pentose could be inverted, the helix will reverse.-- Smokefoot ( talk) 14:04, 27 May 2024 (UTC)
- Wrt your statement:
[resonance] would allow the molecule to convert between 2 or more forms, all of which would be chiral.
there is technically no interconversion between forms in resonance. The discreet resonance contributors we draw are just the extremes the molecule could take on, and the reality is closer to the weighted average of these contributors. That's why if you have a 50/50 mix of two resonance contributors of opposite chirality the actual molecule is achiral. - Phosphates do have more complicated bonding patterns but can be thought of as undergoing bond resonance in a similar manner to lighter organic elements, even if it is not an accurate picture of the electronic structure of the molecule. Bond resonance is just an abstract tool for reasoning on molecular chemistry without having to go do quantum calculations, and it works here well enough. Bond length changes for phosphate and variously substituted phosphates are consistent with typical resonance reasoning. For example, the bond lengths to both unsubstituted phosphate oxygen atoms in DNA are ~1.485 Å, while those in PO3−4 are ~1.578 Å (numbers pulled from Protein Data Bank, PDB ID: 3BEP and PDB ID: PO4). ― Syn path 18:56, 27 May 2024 (UTC)
- ^ Wolfenden, Richard; Ridgway, Caroline; Young, Gregory (1 February 1998). "Spontaneous Hydrolysis of Ionized Phosphate Monoesters and Diesters and the Proficiencies of Phosphatases and Phosphodiesterases as Catalysts". Journal of the American Chemical Society. 120 (4): 833–834. doi: 10.1021/ja9733604.
An insource search shows that we currently have 70 cases where Sciencemadness.org has been used in chemistry articles. Some uses (e.g. in Copper(I) phosphide, current citation #6) have that wiki as a source for our Wikipedia article, although Sciencemadness itself usually does not cite any sources and is clearly not reliable by our standards. More worryingly, it seems that many of the instances of links to that website are to books and articles which are copyright. For example, Tetrafluorohydrazine has a citation #3 to John Drury Clark's 1972 book "Ignition" with a link to a scanned copy as a .pdf. I think we need to go through all 70 instances and either remove the cite or, if the cite itself is valid, remove the links to the copyvio. Comments? Mike Turnbull ( talk) 14:29, 28 May 2024 (UTC)
- Support Pygos ( talk) 15:07, 28 May 2024 (UTC)
- I've changed the Tetrafluorohydrazine citation to point to the Google Books preview rather than Sciencemadness.org. I can probably do the same with a lot of these links.
Recon
rabbit
15:22, 28 May 2024 (UTC)
- Thanks, User:Reconrabbit. I was intending to do similar but was waiting a bit to see if anyone had reasons not to do that. Mike Turnbull ( talk) 15:27, 28 May 2024 (UTC)
- Support replacing those links. A number of dubious sources are cited in our articles, so it is a good idea to root them out. Educational sites include Khan Academy (
https://www.khanacademy.org/science/organic-chemistry) and ChemLibre (
https://chem.libretexts.org/Bookshelves/Organic_Chemistry) seem to present legitimate information, but they probably are not reliable sources. The overarching problem is that most good content sits behind paywalls.--
Smokefoot (
talk)
15:48, 28 May 2024 (UTC)
- That's a good point. Editors here who write chemistry articles should have access to paywalled sources either through their institution or the Wikipedia Library, for example. Provided we summarise such sources accurately, it doesn't matter that the average reader can't themselves easily read the sources. We must try to write quality articles and that often means citing specialist publications. Mike Turnbull ( talk) 17:36, 28 May 2024 (UTC)
- Chemistry LibreTexts is a tricky one out of these; the attribution is terrible, but I have included them as references on some topics because I have had trouble finding online sources that define in basic terms specific concepts like crystal field excitation. Recon rabbit 16:02, 28 May 2024 (UTC)
- In the spirit of gaining some consensus on on-line sources, another one is
American Elements (
https://www.americanelements.com). Dozens or hundreds of chem articles cite this website. Maybe someone can comment on their status as an RS. Like Sigma-Aldrich or other vendors, their physical data are probably acceptable. But they dress up their articles with a narrative, which is not verifiable and is not reviewed in any way. Their narratives are window dressing for the products that they are selling.--
Smokefoot (
talk)
22:49, 28 May 2024 (UTC)
- I support not using American Elements for "use" verification, as we cannot see where their info came from, and when trying to confirm, perhaps only one researcher tried that application. Graeme Bartlett ( talk) 00:01, 29 May 2024 (UTC)
- Contents from American Elements may contain original research. -- Leiem ( talk) 15:08, 29 May 2024 (UTC)
- Sciencenotes.org is another site that shows up infrequently and is written largely by two people with scientific backgrounds but no oversight. Recon rabbit 23:43, 28 May 2024 (UTC)
- I have now replaced or removed all but one reference to Sciencemadness.org.
Graeme Bartlett (
talk)
01:36, 29 May 2024 (UTC)
- Thanks for doing that heavy-lifing!
DMacks (
talk)
02:24, 29 May 2024 (UTC)
- @
Graeme Bartlett:Thanks for removing the direct Sciencemadness.org cites. The insource search at the top of this thread is still giving 62 hits which are copyvios IMO. I'll start trying to replace them, and
User talk:Reconrabbit will probably assist.
Mike Turnbull (
talk)
10:17, 29 May 2024 (UTC)
- Use in
Nicotine and
pyridine is not a copyvio, as works are in public domain.
Graeme Bartlett (
talk)
12:46, 29 May 2024 (UTC)
- OK, I'll check as I go.
Mike Turnbull (
talk)
13:36, 29 May 2024 (UTC)
- There are also several US government publications which are in the public domain. Though I am not sure about LANL publications: are they government or not?
Graeme Bartlett (
talk)
04:48, 1 June 2024 (UTC)
- I think the LANL ones are OK. Part of the website says that's where those documents originally came from. Their library has many books that are clearly PD owing to age but also quite a few I would consider to be copvio as too recent. Anyway, those linked from cites in our articles has been trimmed down to the extent where I'm giving up on the final ones. Mike Turnbull ( talk) 11:04, 1 June 2024 (UTC)
- There are also several US government publications which are in the public domain. Though I am not sure about LANL publications: are they government or not?
Graeme Bartlett (
talk)
04:48, 1 June 2024 (UTC)
- OK, I'll check as I go.
Mike Turnbull (
talk)
13:36, 29 May 2024 (UTC)
- Use in
Nicotine and
pyridine is not a copyvio, as works are in public domain.
Graeme Bartlett (
talk)
12:46, 29 May 2024 (UTC)
- @
Graeme Bartlett:Thanks for removing the direct Sciencemadness.org cites. The insource search at the top of this thread is still giving 62 hits which are copyvios IMO. I'll start trying to replace them, and
User talk:Reconrabbit will probably assist.
Mike Turnbull (
talk)
10:17, 29 May 2024 (UTC)
- Thanks for doing that heavy-lifing!
DMacks (
talk)
02:24, 29 May 2024 (UTC)
Sources
Ok, maybe we transition to a related topic. If one does not have access to basic textbooks, it is next to impossible to edit technical content. "Crystal field excitation" is not a term encountered very often. Yes, one can imagine what it is, but the phrase is not indexed in usual textbooks (Wiberg, Cotton&Wilk, Shriver). My somewhat obnoxious point is that if one is struggling to find a source for a topic, maybe that topic does not merit an article. -- Smokefoot ( talk) 17:23, 28 May 2024 (UTC)
Renaming the " Monolayer-protected cluster molecules" page
I originally asked about this topic here in Helpdesk. It was suggested that before applying for the move of the article, there should be some discussion about how to name it.
The " Monolayer-protected cluster molecules" page is focused on the crystal structures of these nanoclusters in a list-like manner. The current tittle was generated when the page was accepted but it seems too general or ambiguous. There already exists pages like Nanocluster and Thiolate-protected gold cluster, tittles of which feel very similar, even if the contents serve a different purpose. I would call the page as "Crystallized monolayer-protected clusters" or "List of crystallized monolayer-protected clusters". In the text it was stated that "This article is designed to be a list of known structures of MPCs", therefore the new tittle would reflect more clearly the contents than the current one. What do you suggest? Iridium27 ( talk) 09:28, 30 May 2024 (UTC)
- The reason I used a name like that, is that I think that a more general title is better than a narrower title. Thiolate-protected gold cluster would be a narrower topic. Nanocluster does not imply that there is one layer of ligands, or that it occurs in isolation (could be ions, or as a 3D solid), So I think this article is sufficiently different from those. Currently it is just a list, but it would be fair enough to have more introduction on why they are important, and general facts about them. Graeme Bartlett ( talk) 04:46, 1 June 2024 (UTC)
Sorry for the continuing questions in this area. Are there any reactions of organophosphates that can change the PO4 core (i.e. other than hydrolysis or transesterification)? I can't find any, beyond perhaps carbothermal reduction to elemental P. I find that surprising - it would make it the only functional group I can think of that cannot be converted into different functional group. Project Osprey ( talk) 21:35, 9 June 2024 (UTC)
- phospha-Fries rearrangement might be of interest? Fishsicles ( talk) 22:34, 9 June 2024 (UTC)
- Interesting, and new to me. Thanks --
Project Osprey (
talk)
23:17, 9 June 2024 (UTC)
- Well, phosphate, sulfate, and orthosilicate are indeed pretty dead. A look at the "bible" (Greenwood and Earnshaw) does not help much. In synthetic chemistry, it seems that the negative charge needs to be dealt with before anything happens. Maybe protonation sets the scene for condensation leading ultimately to P4O10 or things like that, which then can be sulfided (by P4S10) and chlorinated. The other way to address the issue is, of course, is biochemistry: organisms have been stuck with these oxyanions for billions of years. A quick glance shows the pyruvate enol phosphates, i.e., a phosphate ester, can be reduced to phosphinic acid level. doi 10.1146/annurev.biochem.78.091707.100215. You never get PH3 or element but organophosphorus compounds. On the silicate front, industry has long wishes to avoid SiO2 down to Si then back up to SiMe4-xClx, but I digress.-- Smokefoot ( talk) 23:21, 9 June 2024 (UTC)
- Interesting, and new to me. Thanks --
Project Osprey (
talk)
23:17, 9 June 2024 (UTC)
- Phosphoenolpyruvate mutase might be something to explore as well. Hopefully there are some nice mechanistic studies on the enzyme reaction. ― Syn path 05:15, 10 June 2024 (UTC)
Categories for Fiveling
There is a dispute between me and the article creator about how broad the categories for this article should be. See Talk:Fiveling#Intractable dispute over categories. – LaundryPizza03 ( d c̄) 22:50, 10 June 2024 (UTC)
- Pro-forma, please note that @ LaundryPizza03 did notify me that he is cross-posting. Ldm1954 ( talk) 02:05, 11 June 2024 (UTC)
Apparently Hindawi (publisher) is a huge operation, occasionally putting out some controversial journals. Here are some that appear to apply to the Chemistry project:
- Advances in Materials Science and Engineering
- Biomed Research International
- Contrast Media & Molecular Imaging
- Disease Markers
- Evidence-Based Complementary and Alternative Medicine
- Journal of Environmental and Public Health
- Journal of Healthcare Engineering
- Journal of Nanomaterials
- Oxidative Medicine and Cellular Longevity
According to Retraction Watch the above journals (and others) have been "delisted" from Web of Science. So, I started to remove some of these references from chemistry articles. Are my removals a good idea?-- Smokefoot ( talk) 04:19, 13 June 2024 (UTC)
- I think it would be a mistake to just remove the citations. Just because some indexer thinks a journal is lower quality, does not mean that an article in it is low quality. We'll probably need to replace by another reference if we take them out. I see usage in some broad topics that would easily be replaceable for important facts. Otherwise if the referenced text is garbage based on a garbage publication, then it should be removed altogether. So we should examine each use. {However I have deleted a chemical referenced to Journal of Nanomaterials from an article I wrote (as substance not well proved to exist) }.
Graeme Bartlett (
talk)
07:32, 13 June 2024 (UTC)
- OK, I undid my edits. -- Smokefoot ( talk) 16:51, 13 June 2024 (UTC)
This tortured IUPAC nomenclature. Unfortunate. Practicing chemists do not use this term. -- Smokefoot ( talk) 16:51, 13 June 2024 (UTC)
- Any time I've seen this out in the wild (I'm sure I have somewhere) it's been potassium ferricyanide which is where it was just moved from. If it really had to be at the IUPAC name, shouldn't it get moved to Tripotassium;iron(3+);hexacyanide? There's a reason the water page isn't called Oxidane.
- Also possibly relevant, Potassium trichloridocuprate(II). Recon rabbit 20:20, 13 June 2024 (UTC)
- Even Potassium hexacyanoferrate(III) was much more commonly used. I have moved the page back to potassium ferricyanide which is more commonly used by about a factor of 1000. Wikipedia policy is to use the most frequently used name rather than official (here closest to IUPAC). Letting @ JWBE: know about reversion of the move. Graeme Bartlett ( talk) 22:48, 13 June 2024 (UTC)
Helium Featured Article review
This discussion may be of interest to the community here. XOR'easter ( talk) 20:35, 17 June 2024 (UTC)
There is a RfC on this topic at Talk:Nonmetal#RfC_on_meaning_of_nonmetal which may be of interest. Is the primary use of the term nonmetal for elements in the periodic table, see discussions in Talk:Nonmetal and also at Talk:Nonmetallic compounds and elements. Editor Sandbh is arguing that this is the case, with some other additions. Editors Johnjbarton, Ldm1954 and YBG have questioned this, and both Johnjbarton and Ldm1954 have questioned the scientific accuracy. Ldm1954 ( talk) 07:46, 18 June 2024 (UTC)
It is nearly impossible to write an overview of a topic without good sources. It can also be a disservice because the topic might be misrepresented. If one does not have access to the major works in an area, ask for help, pray that Google books gives you a glimpse to the good stuff, or forget the writing project because you will be forced to rely on old-timey literature and junky online stuff. Almost all the good on-line content is behind a paywall or is inside of major textbooks and encyclopedias. Some major works are online as pdf's, possibly illegally. March's Organic Chem is one example, Greenwood and Earnshaw for Inorganic (especially Main Group) is another. The major review journals (Chem Soc Rev, Angew, Chem Rev) have some open-access content. Org Syn is open access, but it does not provide sufficient context for an overview. -- Smokefoot ( talk) 13:32, 23 June 2024 (UTC)
- McMurry's Organic Chemistry is now free-access online.
DMacks (
talk)
14:46, 23 June 2024 (UTC)
- Yes, at
Openstax, (at 193 Mb) which also has several other useful textbooks.
Mike Turnbull (
talk)
14:51, 23 June 2024 (UTC)
- .... See also John E. McMurry and the McMurry reaction. Mike Turnbull ( talk) 15:05, 23 June 2024 (UTC)
- Yes, at
Openstax, (at 193 Mb) which also has several other useful textbooks.
Mike Turnbull (
talk)
14:51, 23 June 2024 (UTC)
- All true. One recent welcome trend is for authors of "the good stuff" to make it open access. A recent example is
Peter Atkins making the latest version of Concepts in Physical Chemistry freely available and even downloadable at
doi:10.1039/9781837674244. I've added that link to our article about him and would encourage anyone to use this to update relevant topics. In a related move, IUPAC have licensed their
Gold Book CC BY-SA 4.0 which has allowed
User:Walkerma to include their definitions unchanged into articles such as
adsorption and
Boltzmann constant. Fortunately, many good review articles are available via University subscriptions to journals, which is how some of our student editors will gain access. Others can reach JSTOR via
the Wikipedia library. My pet peeve is that the
American Chemical Society don't seem to provide better access via that or other similar mechanism.
Mike Turnbull (
talk)
14:46, 23 June 2024 (UTC)
- Maybe we should assemble a list with links on our project page?--
Smokefoot (
talk)
15:01, 23 June 2024 (UTC)
- Good idea, which I see you have begun to do. We just need to ensure that no-one adds a WP:ELNEVER to a copyvio version of something. Mike Turnbull ( talk) 15:56, 23 June 2024 (UTC)
- It would be great if
Chemical & Engineering News was easier to access beyond the first "free article"; their online archives are extensive. Could be a proposal for
WP:TWL?
Recon
rabbit
16:40, 23 June 2024 (UTC)
- Its an ACS magazine, hence that problem. However, if you know the URL (from their website) that you want, you can often find it archived at the
Wayback Machine etc. So, for example,
this one . How this squares with
WP:ELNEVER I leave to the lawyers: safest would be to use the information you find to write your Wikipedia entry and cite it but not link it.
Mike Turnbull (
talk)
17:01, 23 June 2024 (UTC)
- I don't imagine it would be different from any other situation where we link to paywalled articles. I did some digging and found a discussion on the External Links noticeboard that is tangentially related: Wikipedia:External_links/Noticeboard/Archive_25#Potential license laundering through ghostarchive.org Recon rabbit 17:52, 23 June 2024 (UTC)
- Its an ACS magazine, hence that problem. However, if you know the URL (from their website) that you want, you can often find it archived at the
Wayback Machine etc. So, for example,
this one . How this squares with
WP:ELNEVER I leave to the lawyers: safest would be to use the information you find to write your Wikipedia entry and cite it but not link it.
Mike Turnbull (
talk)
17:01, 23 June 2024 (UTC)
- Maybe we should assemble a list with links on our project page?--
Smokefoot (
talk)
15:01, 23 June 2024 (UTC)
- I am guilty of sometimes citing refs that I cannot access. The usual scenario is that I'm reading paper 'A' and it states a fact with a reference back to paper 'B', but I cannot access paper 'B', If I can't find another option then sometimes I just accept the fact stated by 'A' and cite 'B' as the source. I've never been certain about this type of by-proxy sourcing. Similarly, I've cited Chem Rev articles when I cannot access them, but only when they are unambiguously relevant (name reactions, for instance). I have no access to pay-walled content beyond the Wikipedia Library, nor any SciFinder etc. I feel that I have become very good at finding sources by other means, but it can still be a challenge to write a new overview. Mostly it's just very slow, I've working on
organophosphate for months and I'm still not done with it. What I will say is that I think it's better then when started. There are lots of pages like that, where if not good, they can at least be made not bad. --
Project Osprey (
talk)
20:32, 23 June 2024 (UTC)
- A plea for help with the short list of good (and legal) open source reference works (which I put on our project page). Should we put this list in our Manual of Style (would it be ignored there?), should we leave it on the project page, should we do something else with this list? --
Smokefoot (
talk)
22:50, 23 June 2024 (UTC)
- Our project page is a good place for it. The list is a bit out of scope for a manual of style. Reacting to Project Osprey's A references B, you must be very careful. You could just reference A. But in a significant number of cases I have found that the A paper misrepresents B, so it is best to make sure what it says before using it as a reference. Also thanks to Smokefoot for recommending Shriver & Atkin's Inorganic Chemistry 5th ed, which can also be found to read on archive.org. It is 14 years old. If this is entirely legal, then it would be worth a link too. Graeme Bartlett ( talk) 10:35, 24 June 2024 (UTC)
- A plea for help with the short list of good (and legal) open source reference works (which I put on our project page). Should we put this list in our Manual of Style (would it be ignored there?), should we leave it on the project page, should we do something else with this list? --
Smokefoot (
talk)
22:50, 23 June 2024 (UTC)
Good article reassessment for Nature
Nature has been nominated for a good article reassessment. If you are interested in the discussion, please participate by adding your comments to the reassessment page. If concerns are not addressed during the review period, the good article status may be removed from the article. Z1720 ( talk) 20:11, 26 June 2024 (UTC)
