 | |
| Clinical data | |
|---|---|
| Trade names | Hycamtin, Potactasol |
| AHFS/ Drugs.com | Monograph |
| MedlinePlus | a610007 |
| License data | |
|
Pregnancy category |
|
|
Routes of administration | Intravenous infusion, by mouth ( capsules) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 31.4 % in humans [2] [3] |
| Protein binding | 35% |
| Metabolism | Liver |
| Elimination half-life | 2–3 hours |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.213.372 |
| Chemical and physical data | |
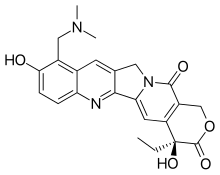
| Formula | C23H23N3O5 |
| Molar mass | 421.453 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Topotecan, sold under the brand name Hycamtin among others, is a chemotherapeutic agent medication that is a topoisomerase inhibitor. It is a synthetic, water-soluble analog of the natural chemical compound camptothecin. It is used in the form of its hydrochloride salt to treat ovarian cancer, lung cancer and other cancer types.
After GlaxoSmithKline received final FDA approval for topotecan on October 15, 2007, it became the first topoisomerase I inhibitor for oral use. [4]
Uses
- Ovarian cancer (FDA May 1996). [5]
- Cervical cancer (FDA June 2006). [6] [7]
- Small cell lung carcinoma (SCLC) (FDA Oct 2007). [8] [9]
Experimental uses
As of 2016, experiments were under way for Neuroblastoma, Brainstem glioma, Ewing's sarcoma and Angelman's syndrome. In addition, topotecan is experimentally treating Non-small cell lung cancer, Colorectal Cancer, Breast cancer, Non-Hodgkin Lymphoma, Endometrial cancer, and Oligodendroglioma. [10]
Angelman's syndrome
Angelman's syndrome is a neuro-genetic disorder characterized by severe developmental delays, seizures, speech impairments and physical impairments. It is an epigenetic disease and other treatments focus on symptoms. It is caused by a deletion or mutation of the maternal allele for the ubiquitin protein ligase E3A. [11] UBE3A is expressed in most body tissues. However, in neurons only the maternal copy of the gene is expressed. UBE3A is located on chromosome 15 and the paternal copy for the gene is genetically imprinted and is silenced by an antisense RNA transcript. The maternal copy control center of the gene is methylated, suppressing transcription in the antisense direction while the paternal copy control center is unmethylated. [12]
Treatment involves unsilencing the paternal allele allowing the normal paternal UBE3A allele to be transcribed. UBE3A, in normal function, adds ubiquitin chains to proteins to target unnecessary or damaged proteins for degradation by the proteasome. [13]
16 topoisomerase inhibitors unsilence paternal UBE3A. Topoisomerases are enzymes that regulate the unwinding of DNA. [14] Of these 16 inhibitors, topotecan was found to induce the strongest upregulation of UBE3A. [15] The enzymes bind to the DNA and cut the phosphate backbone, allowing the DNA to be unwound. Topotecan unsilences the paternal UBE2A allele by reducing the transcription of an antisense transcript. Topotecan inhibits topoisomerase I restoring UBE3A levels to wild-type range in cultured mince neurons. [16]
Transgenic mice with a fluorescently tagged UBE3A were used to test the effectiveness of unsilencing the paternal copy. [12] When tested on mice in vivo, topotecan affected the hippocampus, striatum and cerebral cortex but not the cerebellum unless a higher dose was administered (21.6 micrograms/hour for five days). The study suggested that the topoisomerase inhibitors have the potential to produce a normally functioning UBE3A protein. Most symptoms due to Angelman's syndrome are traditionally treated by speech therapy, physical therapy and occupational therapy. Anti-seizure medication is often prescribed as seizures are a common symptom of Angelman's syndrome. [17] These treatments target only symptoms.
This drug has been administered to cancer patients. It was well tolerated when administered to pediatric and adult patients.
Mechanism of action
Topotecan is a semi-synthetic derivative of camptothecin. Camptothecin is a natural product extracted from the bark of the tree Camptotheca acuminata. Topoisomerase-I is a nuclear enzyme that relieves torsional strain in DNA by opening single strand breaks. [18] Once topoisomerase-I creates a single strand break, the DNA can rotate in front of the advancing replication fork. In physiological environments, topotecan is in equilibrium with its inactive carboxylate form. [19] Topotecan's active lactone form intercalates between DNA bases in the topoisomerase-I cleavage complex. [20] The binding of topotecan in the cleavage complex prevents topoisomerase-I from religating the nicked DNA strand after relieving the strain. [20] This intercalation therefore traps the topoisomerase-I in the cleavage complex bound to the DNA. [20] When the replication-fork collides with the trapped topoisomerase-I, DNA damage occurs. [20] The unbroken DNA strand breaks and mammalian cells cannot efficiently repair these double strand breaks. [21] The accumulation of trapped topoisomerase-I complexes is a known response to apoptotic stimuli. [22] This disruption prevents DNA replication and ultimately leads to cell death. This process leads to breaks in the DNA strand resulting in apoptosis. Administration of topotecan down-regulates its target, topoisomerase-I; therefore, it is dosed to maximize efficacy and minimize related toxicity. [19] Topotecan is often given in combination with paclitaxel as first line treatment for extensive-stage small-cell lung cancer. [19]
Side effects
- Myelosuppression, specifically neutropenia, leukopenia, anemia, and thrombocytopenia
- Diarrhea, nausea, vomiting, stomatitis, and constipation
- Increased susceptibility to infections
- Asthenia [19]
Generic versions
Two generic versions have been approved in the European Union. In Nov 2010 the US FDA approved a generic version. [23] [24]
References
- ^ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- ^ Léger F, Loos WJ, Bugat R, Mathijssen RH, Goffinet M, Verweij J, et al. (December 2004). "Mechanism-based models for topotecan-induced neutropenia". Clinical Pharmacology and Therapeutics. 76 (6): 567–78. doi: 10.1016/j.clpt.2004.08.008. PMID 15592328. S2CID 37527965.
- ^ Gelderblom H, Loos WJ, Sissung TM, Burger H, Nooter K, Soepenberg O, et al. (July 2004). "Effect of ABCG2 genotype and mRNA expression on the bioavailability of topotecan". Journal of Clinical Oncology. 22 (14 suppl): 2015. doi: 10.1200/jco.2004.22.90140.2015. PMID 28015603.
- ^ Delgado, Justine L.; Hsieh, Chao-Ming; Chan, Nei-Li; Hiasa, Hiroshi (2018-01-31). "Topoisomerases as anticancer targets". Biochemical Journal. 475 (2): 373–398. doi: 10.1042/BCJ20160583. ISSN 0264-6021. PMC 6110615. PMID 29363591.
- ^ "FDA Approves Ovaerian Cancer Drug". U.S. Food and Drug Administration. 29 May 1996. Archived from the original on January 19, 2009.
- ^ "FDA Approval for Topotecan Hydrochloride". National Cancer Institute. Archived from the original on 2015-04-06. Retrieved 2009-02-07.
- ^ "FDA approves topotecan hydrochloride (hycamtin) in combination with cisplatin for the treatment of Stage IVB recurrent or persistent carcinoma of the cervix". FDA/Center for Drug Evaluation and Research. Archived from the original on November 7, 2008. Retrieved February 7, 2009.
- ^ "Oral Topotecan FDA Approved in US for Second Line SCLC". Onc Talk. Archived from the original on June 26, 2009. Retrieved February 7, 2009.
- ^ "GSK Receives Approval for Hycamtin (topotecan) Capsules for theTreatment of Relapsed Small Cell Lung Cancer".
- ^ Haglof K (2006). "Recent developments in the clinical activity of topoisomerase-1 inhibitors". Update on Cancer Therapeutics. 1 (2): 117–145. doi: 10.1016/j.uct.2006.05.010.
- ^ (Huang et al)
- ^ a b Beaudet 2011.
- ^ Malzac P, Webber H, Moncla A, Graham JM, Kukolich M, Williams C, Pagon RA, Ramsdell LA, Kishino T, Wagstaff J (June 1998). "Mutation analysis of UBE3A in Angelman syndrome patients". American Journal of Human Genetics. 62 (6): 1353–60. doi: 10.1086/301877. PMC 1377156. PMID 9585605.
- ^ (Miller)
- ^ Malpass K (January 2012). "Neurodevelopmental disorders: Unsilencing dormant Ube3a--hope for Angelman syndrome?". Nature Reviews. Neurology. 8 (2): 62. doi: 10.1038/nrneurol.2012.2. PMID 22270025. S2CID 858183.
- ^ Huang HS, Allen JA, Mabb AM, King IF, Miriyala J, Taylor-Blake B, Sciaky N, Dutton JW, Lee HM, Chen X, Jin J, Bridges AS, Zylka MJ, Roth BL, Philpot BD (December 2011). "Topoisomerase inhibitors unsilence the dormant allele of Ube3a in neurons". Nature. 481 (7380): 185–9. doi: 10.1038/nature10726. PMC 3257422. PMID 22190039.
- ^ (Aditi and Williams)
- ^ Pommier Y, Leo E, Zhang H, Marchand C (May 2010). "DNA topoisomerases and their poisoning by anticancer and antibacterial drugs". Chemistry & Biology. 17 (5): 421–33. doi: 10.1016/j.chembiol.2010.04.012. PMC 7316379. PMID 20534341.
- ^ a b c d Cordell G, ed. (2003). The Alkaloids: Chemistry and Biology. California: Academic Press. pp. 1–50. ISBN 978-0080521497.
- ^ a b c d Pommier Y (October 2006). "Topoisomerase I inhibitors: camptothecins and beyond". Nature Reviews. Cancer. 6 (10): 789–802. doi: 10.1038/nrc1977. PMID 16990856. S2CID 25135019.
- ^ Staker BL, Hjerrild K, Feese MD, Behnke CA, Burgin AB, Stewart L (November 2002). "The mechanism of topoisomerase I poisoning by a camptothecin analog". Proceedings of the National Academy of Sciences of the United States of America. 99 (24): 15387–92. Bibcode: 2002PNAS...9915387S. doi: 10.1073/pnas.242259599. PMC 137726. PMID 12426403.
- ^ Bertrand R, Solary E, O'Connor P, Kohn KW, Pommier Y (April 1994). "Induction of a common pathway of apoptosis by staurosporine". Experimental Cell Research. 211 (2): 314–21. doi: 10.1006/excr.1994.1093. PMID 8143779.
- ^ "FDA Rubber-Stamps APP Pharma's Generic Topotecan for Small Cell Lung and Cervical Cancers". 30 Nov 2010. Archived from the original on 30 June 2016. Retrieved 7 December 2010.
- ^ DNA Topoisomerases and Cancer, Yves Pommier Editor, Humana Press 2012
Sources
- Dagli AI, Mueller J, Williams CA (1993-01-01). Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJ, Stephens K, Amemiya A (eds.). Angelman Syndrome. University of Washington, Seattle. PMID 20301323.
- Bailus BJ, Segal DJ (June 2014). "The prospect of molecular therapy for Angelman syndrome and other monogenic neurologic disorders". BMC Neuroscience. 15: 76. doi: 10.1186/1471-2202-15-76. PMC 4069279. PMID 24946931.
- Beaudet AL (December 2011). "Angelman syndrome: Drugs to awaken a paternal gene". Nature. 481 (7380): 150–2. doi: 10.1038/nature10784. PMC 3638729. PMID 22190038.
External links
- "Topotecan". Drug Information Portal. U.S. National Library of Medicine.
- "Topotecan hydrochloride". Drug Information Portal. U.S. National Library of Medicine.
- NCI Drug Dictionary Definition of Topotecan
- Topotecan Hydrochloride