 | |
 | |
| Clinical data | |
|---|---|
| AHFS/ Drugs.com | International Drug Names |
| MedlinePlus | a693021 |
|
Routes of administration | Intravenous, intramuscular |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 29% |
| Elimination half-life | 0.65–1.20 hrs |
| Excretion | Mainly kidneys (41–66% within 8 hrs) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.063.506 |
| Chemical and physical data | |
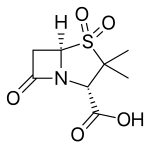
| Formula | C8H11NO5S |
| Molar mass | 233.24 g·mol−1 |
| 3D model ( JSmol) | |
| Melting point | 148 to 151 °C (298 to 304 °F) |
| |
| |
| (verify) | |
Sulbactam is a β-lactamase inhibitor. This drug is given in combination with β-lactam antibiotics to inhibit β-lactamase, an enzyme produced by bacteria that destroys the antibiotics. [1]
It was patented in 1977 and approved for medical use in 1986. [2]
Medical uses
The combination ampicillin/sulbactam (Unasyn) is available in the United States. [3]
The combination cefoperazone/sulbactam (Sulperazon) is available in many countries. [4]
The co-packaged combination sulbactam/durlobactam was approved for medical use in the United States in May 2023. [5]
Mechanism
Sulbactam is primarily used as a suicide inhibitor of β-lactamase, shielding more potent beta-lactams such as ampicillin. [6] Sulbactam itself contains a beta-lactam ring, and has weak antibacterial activity by inhibiting penicillin binding proteins (PBP) 1 and 3, but not 2. [7]
References
- ^ Totir MA, Helfand MS, Carey MP, Sheri A, Buynak JD, Bonomo RA, Carey PR (August 2007). "Sulbactam forms only minimal amounts of irreversible acrylate-enzyme with SHV-1 beta-lactamase". Biochemistry. 46 (31): 8980–8987. doi: 10.1021/bi7006146. PMC 2596720. PMID 17630699.
- ^ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 492. ISBN 9783527607495.
- ^ "Unasyn- ampicillin sodium and sulbactam sodium injection, powder, for solution". DailyMed. U.S. National Library of Medicine. 29 March 2023. Retrieved 25 May 2023.
- ^ "Sulperazon". drugs.com.
- ^ "FDA Approves New Treatment for Pneumonia Caused by Certain Difficult-to-Treat Bacteria". U.S. Food and Drug Administration (Press release). 24 May 2023. Retrieved 24 May 2023.
- ^ Crass RL, Pai MP (February 2019). "Pharmacokinetics and Pharmacodynamics of β-Lactamase Inhibitors". Pharmacotherapy. 39 (2): 182–195. doi: 10.1002/phar.2210. PMID 30589457. S2CID 58567725.
- ^ Penwell WF, Shapiro AB, Giacobbe RA, Gu RF, Gao N, Thresher J, et al. (March 2015). "Molecular mechanisms of sulbactam antibacterial activity and resistance determinants in Acinetobacter baumannii". Antimicrobial Agents and Chemotherapy. 59 (3): 1680–1689. doi: 10.1128/AAC.04808-14. PMC 4325763. PMID 25561334.
Further reading
- Singh GS (January 2004). "Beta-lactams in the new millennium. Part-II: cephems, oxacephems, penams and sulbactam". Mini Reviews in Medicinal Chemistry. 4 (1): 93–109. doi: 10.2174/1389557043487547. PMID 14754446.