
| |
| |
| |
| Names | |
|---|---|
|
IUPAC name
silicon(IV) sulfide
| |
| Other names
silicon disulfide
| |
| Identifiers | |
3D model (
JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.935 |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| SiS2 | |
| Molar mass | 92.218 g/mol |
| Appearance | White (samples are sometimes grey or brown) needles. Rotten egg smell in moist air. |
| Density | 1.853 g/cm3 |
| Melting point | 1,090 °C (1,990 °F; 1,360 K) sublimes |
| Decomposes | |
| Structure | |
| Orthorhombic, oI12 | |
| Ibam, No.72 [1] | |
| Tetrahedral | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other
anions
|
silicon dioxide |
Other
cations
|
carbon disulfide germanium disulfide tin(IV) sulfide lead(IV) sulfide |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
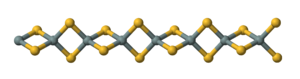
Silicon disulfide is the inorganic compound with the formula Si S2. Like silicon dioxide, this material is polymeric, but it adopts a 1-dimensional structure quite different from the usual forms of SiO2.
Synthesis, structure, and properties
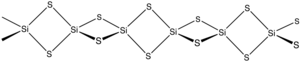
The material is formed by heating silicon and sulfur or by the exchange reaction between SiO2 and Al2S3. The material consists of chains of edge-shared tetrahedra, Si(μ-S)2Si(μS)2, etc. [2]
Like other silicon sulfur-compounds (e.g., bis(trimethylsilyl)sulfide) SiS2 hydrolyzes readily to release H2S. In liquid ammonia it is reported to form the imide Si(NH)2 and NH4SH, [3] but a recent report has identified crystalline (NH4)2[SiS3(NH3)]·2NH3 as a product which contains the tetrahedral thiosilicate anion, SiS3(NH3)2-. [4]
Reaction with ethanol gives the alkoxide tetraethyl orthosilicate and H2S. [3] With bulky tert-butanol, alcoholysis gives tris(tert-butoxy)silanethiol: [5]
- 3 (CH3)3COH + SiS2 → [(CH3)3CO]3SiSH + H2S
Reaction with sodium sulfide, magnesium sulfide and aluminum sulfide give thiosilicates. [3]
SiS2 is claimed to occur in certain interstellar objects. [6]
References
- ^ Weiss, A.; Weiss, A. (1954). "Über Siliciumchalkogenide. VI. Zur Kenntnis der faserigen Siliciumdioxyd-Modifikation". Zeitschrift für Anorganische und Allgemeine Chemie. 276 (1–2): 95–112. doi: 10.1002/zaac.19542760110.
- ^ Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego: Academic Press. ISBN 0-12-352651-5. A printing error in this book states that rSiSi is 214 picometers, when in fact that distance describes rSiS.
- ^ a b c Greenwood, Norman N.; Earnshaw, Alan (1984). Chemistry of the Elements. Oxford: Pergamon Press. p. 359. ISBN 978-0-08-022057-4.
- ^ Meier, Martin; Korber, Nikolaus (2009). "The first thiosilicate from solution: synthesis and crystal structure of (NH4)2[SiS3(NH3)]·2NH3". Dalton Transactions (9): 1506–1508. doi: 10.1039/b818856d. ISSN 1477-9226. PMID 19421590.
- ^ R. Piękoś, W. Wojnowski (1962). "Untersuchungen über die Alkoholyse des SiS2. II. Darstellung von Trialkoxysilanthiolen und Tetraalkoxycyclodisilthianen aus den tertiären Alkoholen". Z. Anorg. Allg. Chem. 318 (3–4): 212–216. doi: 10.1002/zaac.19623180310.
- ^ Goebel, J. H. (1993). "SiS2 in Circumstellar Shells" (PDF). Astronomy and Astrophysics. 278 (1): 226–230. Bibcode: 1993A&A...278..226G.
