 | |
| Clinical data | |
|---|---|
| Other names | uracil-6-carboxylic acid |
| AHFS/ Drugs.com | International Drug Names |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard ( EPA) | |
| ECHA InfoCard | 100.000.563 |
| Chemical and physical data | |
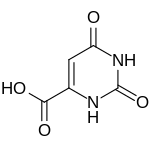
| Formula | C5H4N2O4 |
| Molar mass | 156.097 g·mol−1 |
| 3D model ( JSmol) | |
| |
| |
| | |
Orotic acid ( /ɔːˈrɒtɪk/) [1] is a pyrimidinedione and a carboxylic acid. Historically, it was believed to be part of the vitamin B complex and was called vitamin B13, but it is now known that it is not a vitamin.
The compound is synthesized in the body via a mitochondrial enzyme, dihydroorotate dehydrogenase [2] or a cytoplasmic enzyme of pyrimidine synthesis pathway. It is sometimes used as a mineral carrier in some dietary supplements (to increase their bioavailability), most commonly for lithium orotate.
Synthesis
Dihydroorotate is synthesized to orotic acid by the enzyme dihydroorotate dehydrogenase, where it later combines with phosphoribosyl pyrophosphate (PRPP) to form orotidine-5'-monophosphate (OMP). A distinguishing characteristic of pyrimidine synthesis is that the pyrimidine ring is fully synthesized before being attached to the ribose sugar, whereas purine synthesis happens by building the base directly on the sugar. [3]
Chemistry
Orotic acid is a Bronsted acid and its conjugate base, the orotate anion, is able to bind to metals. Lithium orotate, for example, has been investigated for use in treating alcoholism, [4] [5] and complexes of cobalt, manganese, nickel, and zinc are known. [6] The pentahydrate nickel orotate coordination complex converts into a polymeric trihydrate upon heating in water at 100 °C. [6] [7] [8] Crystals of the trihydrate can be obtained by hydrothermal treatment of nickel(II) acetate and orotic acid. When the reactions are run with bidentate nitrogen ligands such as 2,2'- bipyridine present, other solids can be obtained.
Pathology
A buildup of orotic acid can lead to orotic aciduria and acidemia. [9] It may be a symptom of an increased ammonia load due to a metabolic disorder, such as a urea cycle disorder.
In ornithine transcarbamylase deficiency, an X-linked inherited and the most common urea cycle disorder, excess carbamoyl phosphate is converted into orotic acid. This leads to an increased serum ammonia level, increased serum and urinary orotic acid levels and a decreased serum blood urea nitrogen level. This also leads to an increased urinary orotic acid excretion, because the orotic acid is not being properly utilized and must be eliminated. The hyperammonemia depletes alpha-ketoglutarate leading to the inhibition of the tricarboxylic acid cycle (TCA) decreasing adenosine triphosphate (ATP) production.
Orotic aciduria is a cause of megaloblastic anaemia.
Biochemistry
Orotic acid is a precursor to a RNA base, uracil. [10] The breast milk of smokers has a higher concentration of orotic acid than that of a non smoking woman. It is reasoned that the smoking causes the pyrimidine biosynthesis process in the mother to be altered thus causing the orotic acid concentration to increase. [11]
A modified orotic acid (5-fluoroorotic acid) is toxic to yeast. The mutant yeasts which are resistant to 5-fluoroorotic acid require a supply of uracil. [12]
See also
References
- ^ "Orotic acid". Merriam-Webster.com Dictionary. Retrieved 2023-02-07.
- ^ Rawls J, Knecht W, Diekert K, Lill R, Löffler M (April 2000). "Requirements for the mitochondrial import and localization of dihydroorotate dehydrogenase". European Journal of Biochemistry. 267 (7): 2079–2087. doi: 10.1046/j.1432-1327.2000.01213.x. PMID 10727948.
- ^ Harvey D, Ferrier D, eds. (2008). Biochemistry (PDF) (5th ed.). Lippincott, Williams & Wilkins. p. 302.
- ^ Bach I, Kumberger O, Schmidbaur H (1990). "Orotate complexes. Synthesis and crystal structure of lithium orotate(—I) monohydrate and magnesium bis[orotate(—I)] octahydrate". Chemische Berichte. 123 (12): 2267–2271. doi: 10.1002/cber.19901231207.
- ^ Sartori HE (1986). "Lithium orotate in the treatment of alcoholism and related conditions". Alcohol. 3 (2): 97–100. doi: 10.1016/0741-8329(86)90018-2. PMID 3718672.
- ^ a b Plater MJ, Foreman MR, Skakle JM, Howie RA (April 2002). "Hydrothermal crystallisation of metal (II) orotates (M= nickel, cobalt, manganese or zinc). Effect of 2, 2-bipyridyl, 2, 2-dipyridyl amine, 1-methyl-3-(2-pyridyl) pyrazole, phenanthroline and 2,9-dimethyl-1,10-phenanthroline upon structure". Inorganica Chimica Acta. 332 (1): 135–145. doi: 10.1016/S0020-1693(02)00728-4.
- ^ Plater MJ, Foreman MR, Skakle JM, Howie RA (2002). "CCDC 189770 – Catena-((μ2-1,2,3,6-tetrahydro-2,6-dioxopyrimidine-4-carboxylato-N,O,O')-triaqua-nickel(ii))". Cambridge Crystallographic Data Centre. doi: 10.5517/cc6cgmm.
- ^ Karipides AN, Thomas B (1986). "The structures of tetraaqua(uracil-6-carboxylate)zinc(II) monohydrate (A) and tetraaqua(uracil-6-carboxylato)nickel(II) monohydrate (B)". Acta Crystallographica Section C: Crystal Structure Communications. C42 (12): 1705–1707. doi: 10.1107/S0108270186090856.
- ^ Balasubramaniam S, Duley JA, Christodoulou J (September 2014). "Inborn errors of pyrimidine metabolism: clinical update and therapy". Journal of Inherited Metabolic Disease. 37 (5): 687–698. doi: 10.1007/s10545-014-9742-3. PMID 25030255. S2CID 25297304.
- ^ Ashihara H, Stasolla C, Loukanina N, Thorpe TA (2000). "Purine and pyrimidine metabolism in cultured white spruce (Picea glauca) cells: Metabolic fate of 14C-labeled precursors and activity of key enzymes". Physiologia Plantarum. 108: 25–33. doi: 10.1034/j.1399-3054.2000.108001025.x.
- ^ Karatas F (October 2002). "An investigation of orotic acid levels in the breastmilk of smoking and non-smoking mothers". European Journal of Clinical Nutrition. 56 (10): 958–960. doi: 10.1038/sj.ejcn.1601420. PMID 12373615. S2CID 29181790.
- ^ Adrio JL, Veiga M, Casqueiro J, López M, Fernández C (1993). "Isolation of Phaffia rhodozyma auxotrophic mutants by enrichment methods". The Journal of General and Applied Microbiology. 39 (3): 303–312. doi: 10.2323/jgam.39.303. S2CID 84498320.
Further reading
- Greenbaum SB (1954). "Potential Metabolic Antagonists of Orotic Acid: 6-Uracilsulfonamide and 6-Uracil Methyl Sulfone". Journal of the American Chemical Society. 76 (23): 6052–6054. doi: 10.1021/ja01652a056.
External links
- Orotic+Acid at the U.S. National Library of Medicine Medical Subject Headings (MeSH)