| |
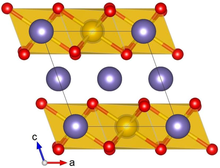
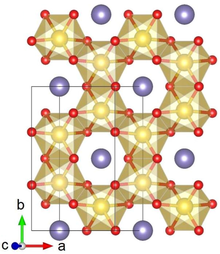 Crystal structure with Pt shown in yellow, Li in purple and O in red
| |
 Scale bar 1 mm
[1]
| |
| Names | |
|---|---|
|
Preferred IUPAC name
Lithium platinate | |
| Identifiers | |
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| Li2PtO3 | |
| Appearance | Yellow crystals |
| Band gap | 2.3 eV [2] |
| Structure | |
| Monoclinic, C2/m [2] | |
a = 5.1836(2) Å, b = 8.9726(3) Å, c = 5.1113(1) Å α = 90°, β = 109.864(2)°, γ = 90°
| |
Formula units (Z)
|
4 |
| Related compounds | |
Other
anions
|
Lithium iridate, lithium ruthenate |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
Lithium platinate, Li2PtO3, is a chemical compound of lithium, platinum and oxygen. It is a semiconductor with a layered honeycomb crystal structure and a band gap of 2.3 eV, and can be prepared by direct calcination of Pt metal and lithium carbonate at ca. 600 °C. [3] Lithium platinate is a potential lithium-ion battery electrode material, [2] [4] though this application is hindered by the high costs of Pt, as compared to the cheaper Li2MnO3 alternative. [5]
References
- ^ Freund, F.; Williams, S. C.; Johnson, R. D.; Coldea, R.; Gegenwart, P.; Jesche, A. (2016). "Single crystal growth from separated educts and its application to lithium transition-metal oxides". Scientific Reports. 6: 35362. arXiv: 1604.04551. Bibcode: 2016NatSR...635362F. doi: 10.1038/srep35362. PMC 5066249. PMID 27748402.
- ^ a b c O'Malley, Matthew J.; Verweij, Henk; Woodward, Patrick M. (2008). "Structure and properties of ordered Li2IrO3 and Li2PtO3". Journal of Solid State Chemistry. 181 (8): 1803. Bibcode: 2008JSSCh.181.1803O. doi: 10.1016/j.jssc.2008.04.005.
- ^ Kasuya, Ryo; Miki, Takeshi; Morikawa, Hisashi; Tai, Yutaka (2013). "Synthesis of alkali metal platinates and their dissolution behavior in hydrochloric acid". Journal of the Ceramic Society of Japan. 121 (1418): 884. doi: 10.2109/jcersj2.121.884.
- ^ Okada, Shigeto; Yamaki, Jun-Ichi; Asakura, Kaoru; Ohtsuka, Hideaki; Arai, Hajime; Tobishima, Shin-Ichi; Sakurai, Yoji (1999). "Cathode characteristics of layered rocksalt oxide, Li2PtO3". Electrochimica Acta. 45 (1–2): 329–334. doi: 10.1016/S0013-4686(99)00214-5.
- ^ Yoshio, Masaki; Brodd, Ralph J.; Kozawa, Akiya (17 July 2010). Lithium-Ion Batteries: Science and Technologies. Springer Science & Business Media. p. 10. ISBN 978-0-387-34445-4.