| |||
| Names | |||
|---|---|---|---|
|
IUPAC name
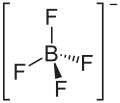
Lithium tetrafluoroborate
| |||
| Other names
Borate(1-), tetrafluoro-, lithium
| |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.034.692 | ||
PubChem
CID
|
|||
| UNII | |||
CompTox Dashboard (
EPA)
|
|||
| |||
| |||
| Properties | |||
| LiBF4 | |||
| Molar mass | 93.746 g/mol | ||
| Appearance | White/grey crystalline solid | ||
| Odor | odorless | ||
| Density | 0.852 g/cm3 solid | ||
| Melting point | 296.5 °C (565.7 °F; 569.6 K) | ||
| Boiling point | decomposes | ||
| Very soluble [1] | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Harmful, causes burns, hygroscopic. | ||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | External MSDS | ||
| Related compounds | |||
Other
anions
|
Tetrafluoroborate, | ||
Related compounds
|
Nitrosyl tetrafluoroborate | ||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
Lithium tetrafluoroborate is an inorganic compound with the formula Li BF4. It is a white crystalline powder. It has been extensively tested for use in commercial secondary batteries, an application that exploits its high solubility in nonpolar solvents. [2]
Applications
Although BF4− has high ionic mobility, solutions of its Li+ salt are less conductive than other less associated salts. [2] As an electrolyte in lithium-ion batteries, LiBF4 offers some advantages relative to the more common LiPF6. It exhibits greater thermal stability [3] and moisture tolerance. [4] For example, LiBF4 can tolerate a moisture content up to 620 ppm at room temperature whereas LiPF6 readily hydrolyzes into toxic POF3 and HF gases, often destroying the battery's electrode materials. Disadvantages of the electrolyte include a relatively low conductivity and difficulties forming a stable solid electrolyte interface with graphite electrodes.
Thermal stability
Because LiBF4 and other alkali-metal salts thermally decompose to evolve boron trifluoride, the salt is commonly used as a convenient source of the chemical at the laboratory scale: [5]
Production
LiBF4 is a byproduct in the industrial synthesis of diborane: [5] [6]
LiBF4 can also be synthesized from LiF and BF3 in an appropriate solvent that is resistant to fluorination by BF3 (e.g. HF, BrF3, or liquified SO2): [5]
- LiF + BF3 → LiBF4
References
- ^ GFS-CHEMICALS Archived 2006-03-16 at the Wayback Machine
- ^ a b Xu, Kang. "Nonaqueous Liquid Electrolytes for Lithium-Based Rechargeable Batteries."Chemical Reviews 2004, volume 104, pp. 4303-418. doi: 10.1021/cr030203g
- ^ S. Zhang; K. Xu; T. Jow (2003). "Low-temperature performance of Li-ion cells with a LiBF4-based electrolyte". Journal of Solid State Electrochemistry. 7 (3): 147–151. doi: 10.1007/s10008-002-0300-9. S2CID 96775286. Retrieved 16 February 2014.
- ^ S. S. Zhang; z K. Xu & T. R. Jow (2002). "Study of LiBF4 as an Electrolyte Salt for a Li-Ion Battery". Journal of the Electrochemical Society. 149 (5): A586–A590. Bibcode: 2002JElS..149A.586Z. doi: 10.1149/1.1466857. Retrieved 16 February 2014.
- ^ a b c Robert Brotherton; Joseph Weber; Clarence Guibert & John Little (2000). "Boron Compounds". Ullmann's Encyclopedia of Industrial Chemistry. p. 10. doi: 10.1002/14356007.a04_309. ISBN 3527306730.
- ^ Brauer, Georg (1963). Handbook of Preparative Inorganic Chemistry Vol. 1, 2nd Ed. New York: Academic Press. p. 773. ISBN 978-0121266011.