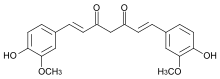
The diarylheptanoids (also known as diphenylheptanoids) are a class of plant secondary metabolites. Diarylheptanoids consist of two aromatic rings ( aryl groups) joined by a seven carbons chain ( heptane) and having various substituents. [1] They can be classified into linear ( curcuminoids) and cyclic diarylheptanoids. The best known member is curcumin, which is isolated from turmeric (Curcuma longa) and is known as food coloring E100. Some other Curcuma species, such as Curcuma comosa also produce diarylheptanoids.
They have been reported from plants in 10 different families, e.g. Betulaceae and Zingiberaceae.
A diarylheptanoid is an intermediate in the biosynthesis of phenylphenalenones in Anigozanthos preissii [2] or Wachendorfia thyrsiflora (Haemodoraceae). [3]
Cyclic diarylheptanoids

Cyclic diarylheptanoids formed from myricanone can be isolated from the bark of Myrica rubra (Myricaceae). [4] Two cyclic diarylheptanoids, named ostryopsitrienol and ostryopsitriol, can be isolated from the stems of endemic Chinese medicinal plant Ostryopsis nobilis (Betulaceae). [5] Acerogenin M can be found in Acer nikoense (Sapindaceae). [6] Jugcathayenoside and (+)- galeon can be found in the root bark of Juglans cathayensis (Juglandaceae). [7]
Health effects
The antioxidant activity of diarylheptanoids isolated from rhizomes of Etlingera elatior (Zingiberaceae) is greater than that of α-tocopherol. [8]
References
- ^ Brahmachari, Goutam (2013-02-20). Chemistry and Pharmacology of Naturally Occurring Bioactive Compounds. CRC Press. pp. 285–. ISBN 9781439891674. Retrieved 5 July 2013.
- ^ Hölscher, Dirk; Schneider, Bernd (1995). "A diarylheptanoid intermediate in the biosynthesis of phenylphenalenones in Anigozanthos preissii". Journal of the Chemical Society, Chemical Communications (5): 525–526. doi: 10.1039/C39950000525.
- ^ Brand, S; Hölscher, D; Schierhorn, A; Svatos, A; Schröder, J; Schneider, B (2006). "A type III polyketide synthase from Wachendorfia thyrsiflora and its role in diarylheptanoid and phenylphenalenone biosynthesis". Planta. 224 (2): 413–428. doi: 10.1007/s00425-006-0228-x. PMID 16496097. S2CID 38508249.
- ^ Akazawa, H; Fujita, Y; Banno, N; Watanabe, K; Kimura, Y; Manosroi, A; Manosroi, J; Akihisa, T (2010). "Three new cyclic diarylheptanoids and other phenolic compounds from the bark of Myrica rubra and their melanogenesis inhibitory and radical scavenging activities". Journal of Oleo Science. 59 (4): 213–221. doi: 10.5650/jos.59.213. PMID 20299768.
- ^ Zhang, Yan-Xia; Xia, Bing; Zhou, Yan; Ding, Li-Sheng; Peng, Shu-Lin (2013). "Two new cyclic diarylheptanoids from the stems of Ostryopsis nobilis". Chinese Chemical Letters. 24 (6): 512–514. doi: 10.1016/j.cclet.2013.03.035. S2CID 95251161. Archived from the original on 2020-05-29. Retrieved 2018-10-27.
- ^ Akihisa, T; Taguchi, Y; Yasukawa, K; Tokuda, H; Akazawa, H; Suzuki, T; Kimura, Y (2006). "Acerogenin M, a cyclic diarylheptanoid, and other phenolic compounds from Acer nikoense and their anti-inflammatory and anti-tumor-promoting effects". Chemical & Pharmaceutical Bulletin. 54 (5): 735–739. doi: 10.1248/cpb.54.735. PMID 16651781.
- ^ Li, Juan; Sun, Jia-Xiang; Yu, Heng-Yi; Chen, Zu-Yu; Zhao, Xiao-Ya; Ruan, Han-Li (2013). "Diarylheptanoids from the root bark of Juglans cathayensis". Chinese Chemical Letters. 24 (6): 521–523. doi: 10.1016/j.cclet.2013.03.050.
- ^ Mohamad, Habsah; Lajis, Nordin H.; Abas, Faridah; Ali, Abdul Manaf; Sukari, Mohamad Aspollah; Kikuzaki, Hiroe; Nakatani, Nobuji (2005). "Antioxidative constituents of Etlingera elatior". Journal of Natural Products. 68 (2): 285–288. doi: 10.1021/np040098l. PMID 15730265.
External links
- Stilbenoid, diarylheptanoid and gingerol biosynthesis pathway at genome.jp
- "Diarylheptanoids". National Center for Biotechnology Information. Retrieved 3 July 2013.