
Kavalactones are a class of lactone compounds found in kava roots and Alpinia zerumbet (shell ginger). [1] Some kavalactones are bioactive. [2] [3]
Bioactivity
Kava extract interacts with many pharmaceuticals and herbal medications. In human volunteers, in vivo inhibition includes CYP1A2 [4] and CYP2E1 [5] through use of probe drugs to measure inhibition.
Research
Its anxiolytic and hepatotoxicity activities have been investigated. [6] [7] [8]
The major kavalactones (except for desmethoxyyangonin) potentiate GABAA receptors, which may underlie the anxiolytic and sedative properties of kava. Further, inhibition of the reuptake of norepinephrine and dopamine, binding to the CB1 receptor, [9] inhibition of voltage-gated sodium and calcium channels, and monoamine oxidase B reversible inhibition are additional pharmacological actions that have been reported for kavalactones. [10]
Kavalactone-type compounds may help protect against high glucose induced cell damage. [2]
Toxicity
Several kavalactones (e.g., methysticin and yangonin) affect a group of enzymes involved in metabolism, called the CYP450 system. Hepatotoxicity occurred in a small portion of previously healthy kava users, [7] [11] particularly from extracts, as opposed to whole root powders.
Compounds
At least 18 different kavalactones are known, [1] with methysticin being the first identified. [12] Multiple analogues, such as ethysticin, have also been isolated. [13] Some consist of a substituted α-pyrone as the lactone, while others are partially saturated.
The average elimination half-life of kavalactones typically present in kava root is 9 hr. [14]
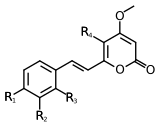
| Name | Structure | R1 | R2 | R3 | R4 |
|---|---|---|---|---|---|
| Yangonin | 1 | -OCH3 | -H | -H | -H |
| 10-methoxyyangonin | 1 | -OCH3 | -H | -OCH3 | -H |
| 11-methoxyyangonin | 1 | -OCH3 | -OCH3 | -H | -H |
| 11-hydroxyyangonin | 1 | -OCH3 | -OH | -H | -H |
| Desmethoxyyangonin | 1 | -H | -H | -H | -H |
| 11-methoxy-12-hydroxydehydrokavain | 1 | -OH | -OCH3 | -H | -H |
| 7,8-dihydroyangonin | 2 | -OCH3 | -H | -H | -H |
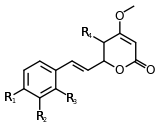
| Kavain | 3 | -H | -H | -H | -H |
| 5-hydroxykavain | 3 | -H | -H | -H | -OH |
| 5,6-dihydroyangonin | 3 | -OCH3 | -H | -H | -H |
| 7,8-dihydrokavain | 4 | -H | -H | -H | -H |
| 5,6,7,8-tetrahydroyangonin | 4 | -OCH3 | -H | -H | -H |
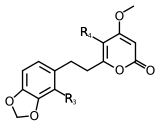
| 5,6-dehydromethysticin | 5 | -O-CH2-O- | -H | -H | |
| Methysticin | 7 | -O-CH2-O- | -H | -H | |
| 7,8-dihydromethysticin | 8 | -O-CH2-O- | -H | -H | |
 |
 |
 |
 |
 |
 |
 |
 |
Biosynthesis
The kavalactone biosynthetic pathway in Piper methysticum was described in 2019. [15]
See also
References
- ^ a b Tadiparthi, Krishnaji; Anand, Pragya (2021). "A Review on Synthetic Approaches towards Kavalactones". Synthesis. 53 (19): 3469–3484. doi: 10.1055/s-0040-1706044. S2CID 236392304.
- ^ a b You, Hualin; He, Min; Pan, Di; Fang, Guanqin; Chen, Yan; Zhang, Xu; Shen, Xiangchun; Zhang, Nenling (2022). "Kavalactones isolated from Alpinia zerumbet (Pers.) Burtt. Et Smith with protective effects against human umbilical vein endothelial cell damage induced by high glucose". Natural Product Research. 36 (22): 5740–5746. doi: 10.1080/14786419.2021.2023866. PMID 34989299. S2CID 245771677.
- ^ James M. Mathews; Amy S. Etheridge; Sherry R. Black (2002). "Inhibition of Human Cytochrome P450 Activities by Kava Extract and Kavalactones". Drug Metabolism and Disposition. 30 (11): 1153–1157. doi: 10.1124/dmd.30.11.1153. PMID 12386118.
- ^ Russmann, S; Lauterburg, B; Barguil, Y; Choblet, E; Cabalion, P; Rentsch, K; Wenk, M (2005). "Traditional aqueous kava extracts inhibit cytochrome P450 1A2 in humans: Protective effect against environmental carcinogens?". Clinical Pharmacology & Therapeutics. 77 (5): 453–454. doi: 10.1016/j.clpt.2005.01.021. PMID 15900292. S2CID 36009940.
- ^ Gurley, B; Gardner, S; Hubbard, M; Williams, D; Gentry, W; Khan, I; Shah, A (2005). "In vivo effects of goldenseal, kava kava, black cohosh, and valerian on human cytochrome P450 1A2, 2D6, 2E1, and 3A4/5 phenotypes". Clinical Pharmacology & Therapeutics. 77 (5): 415–426. doi: 10.1016/j.clpt.2005.01.009. PMC 1894911. PMID 15900287.
- ^ Sarris, Jerome; LaPorte, Emma; Schweitzer, Isaac (2011-01-01). "Kava: A Comprehensive Review of Efficacy, Safety, and Psychopharmacology". Australian & New Zealand Journal of Psychiatry. 45 (1): 27–35. doi: 10.3109/00048674.2010.522554. PMID 21073405. S2CID 42935399.
- ^ a b Teschke, R; Lebot, V (2011). "Proposal for a kava quality standardization code". Food and Chemical Toxicology. 49 (10): 2503–16. doi: 10.1016/j.fct.2011.06.075. PMID 21756963.
- ^ Wang, J; Qu, W; Bittenbender, H. C.; Li, Q. X. (2013). "Kavalactone content and chemotype of kava beverages prepared from roots and rhizomes of Isa and Mahakea varieties and extraction efficiency of kavalactones using different solvents". Journal of Food Science and Technology. 52 (2): 1164–1169. doi: 10.1007/s13197-013-1047-2. PMC 4325077. PMID 25694734.
- ^ Ligresti A, Villano R, Allarà M, Ujváry I, Di Marzo V (2012). "Kavalactones and the endocannabinoid system: the plant-derived yangonin is a novel CB₁ receptor ligand". Pharmacol. Res. 66 (2): 163–9. doi: 10.1016/j.phrs.2012.04.003. PMID 22525682.
- ^ Singh YN, Singh NN (2002). "Therapeutic potential of kava in the treatment of anxiety disorders". CNS Drugs. 16 (11): 731–43. doi: 10.2165/00023210-200216110-00002. PMID 12383029. S2CID 34322458.
- ^ Teschke, R; Qiu, S. X.; Xuan, T. D.; Lebot, V (2011). "Kava and kava hepatotoxicity: Requirements for novel experimental, ethnobotanical and clinical studies based on a review of the evidence". Phytotherapy Research. 25 (9): 1263–74. doi: 10.1002/ptr.3464. PMID 21442674. S2CID 19142750.
- ^ Naumov, P.; Dragull, K.; Yoshioka, M.; Tang, C.-S.; Ng, S. W. (2008). "Structural Characterization of Genuine (-)-Pipermethystine, (-)-Epoxypipermethystine, (+)-Dihydromethysticin and Yangonin from the Kava Plant (Piper methysticum)". Natural Product Communications. 3 (8): 1333–1336. doi: 10.1177/1934578X0800300819. S2CID 92030132.
- ^ Shulgin, A. (1973). "The narcotic pepper - the chemistry and pharmacology of Piper methysticum and related species". Bulletin on Narcotics (2): 59–74.
- ^ "Kava (Piper methysticum): Pharmacodynamics/Kinetics". Sigma-Aldrich Co. LLC. 2010.
- ^ Pluskal, Tomáš; Torrens-Spence, Michael P.; Fallon, Timothy R.; De Abreu, Andrea; Shi, Cindy H.; Weng, Jing-Ke (2019-07-22). "The biosynthetic origin of psychoactive kavalactones in kava". Nature Plants. 5 (8). Springer Science and Business Media LLC: 867–878. doi: 10.1038/s41477-019-0474-0. hdl: 1721.1/124692. ISSN 2055-0278. PMID 31332312. S2CID 198139136.