| |
| Names | |
|---|---|
|
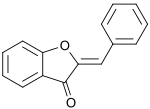
Preferred IUPAC name
2-Benzylidene-1-benzofuran-3(2H)-one | |
| Other names
2-Benzylidenebenzofuran-3(2H)-one
2-Benzylidene-1-benzofuran-3-one | |
| Identifiers | |
| |
3D model (
JSmol)
|
|
| ChemSpider | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C15H10O2 | |
| Molar mass | 222.243 g·mol−1 |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
An aurone is a heterocyclic chemical compound, which is a type of flavonoid. [1] There are two isomers of the molecule, with (E)- and (Z)-configurations. The molecule contains a benzofuran element associated with a benzylidene linked in position 2. In aurone, a chalcone-like group is closed into a 5-membered ring instead of the 6-membered ring more typical of flavonoids.
Aurone derivatives

Aurone forms the core for a family of derivatives which are known collectively as aurones. Aurones are plant flavonoids that provide yellow color to the flowers of some popular ornamental plants, such as snapdragon and cosmos. [2] Aurones including 4'-chloro-2-hydroxyaurone (C15H11O3Cl) and 4'-chloroaurone (C15H9O2Cl) can also be found in the brown alga Spatoglossum variabile. [3]
Most aurones are in a (Z)-configuration, which is the more stable configuration according to Austin Model 1 computation. [3] But there are also some in the (E)-configurations such as (E)-3'-O-β-d-glucopyranosyl-4,5,6,4'-tetrahydroxy-7,2'-dimethoxyaurone, found in Gomphrena agrestis. [4]
Biosynthesis
Aurones are biosynthesized starting from coumaryl-CoA. [5] Aureusidin synthase catalyzes the creation of aurones from chalcones through hydroxylation and oxidative cyclization. [2]
Applications
Some aurone derivatives possess antifungal properties [6] and analogy with flavonoids suggests that aurones could have other biological properties. [7]
Related compound examples
- Aureusidin
- Hispidol (6,4'-dihydroxyaurone) [8]
- Leptosidin
- Sulfuretin (6,3',4'-trihydroxyaurone)
- 4,5,6-Trihydroxyaurone
References
- ^ Nakayama, T (2002). "Enzymology of aurone biosynthesis". Journal of Bioscience and Bioengineering. 94 (6): 487–91. doi: 10.1016/S1389-1723(02)80184-0. PMID 16233339.
- ^ a b Nakayama, T; Sato, T; Fukui, Y; Yonekura-Sakakibara, K; Hayashi, H; Tanaka, Y; Kusumi, T; Nishino, T (2001). "Specificity analysis and mechanism of aurone synthesis catalyzed by aureusidin synthase, a polyphenol oxidase homolog responsible for flower coloration". FEBS Letters. 499 (1–2): 107–11. doi: 10.1016/S0014-5793(01)02529-7. PMID 11418122.
- ^ a b Atta-Ur-Rahman; Choudhary, MI; Hayat, S; Khan, AM; Ahmed, A (2001). "Two new aurones from marine brown alga Spatoglossum variabile". Chemical & Pharmaceutical Bulletin. 49 (1): 105–7. doi: 10.1248/cpb.49.105. PMID 11201212.
- ^ Ferreira, EO; Salvador, MJ; Pral, EM; Alfieri, SC; Ito, IY; Dias, DA (2004). "A new heptasubstituted (E)-aurone glucoside and other aromatic compounds of Gomphrena agrestis with biological activity" (PDF). Zeitschrift für Naturforschung C. 59 (7–8): 499–505. doi: 10.1515/znc-2004-7-808. PMID 15813368. S2CID 15589214.
- ^ Vogt, T. (2010). "Phenylpropanoid Biosynthesis". Molecular Plant. 3: 2–20. doi: 10.1093/mp/ssp106. PMID 20035037.
- ^ Sutton, Caleb L.; Taylor, Zachary E.; Farone, Mary B.; Handy, Scott T. (2017-02-15). "Antifungal activity of substituted aurones". Bioorganic & Medicinal Chemistry Letters. 27 (4): 901–903. doi: 10.1016/j.bmcl.2017.01.012. PMID 28094180.
- ^ Villemin, Didier; Martin, Benoit; Bar, Nathalie (1998). "Application of Microwave in Organic Synthesis. Dry Synthesis of 2-Arylmethylene-3(2)-naphthofuranones". Molecules. 3 (8): 88. doi: 10.3390/30300088.
- ^ Hispidol on metabolomics.jp