| |
| Names | |
|---|---|
|
Preferred IUPAC name
2-Oxopentanedioic acid | |
| Other names
2-Ketoglutaric acid
alpha-Ketoglutaric acid 2-Oxoglutaric acid Oxoglutaric acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.005.756 |
| KEGG | |
| MeSH | alpha-ketoglutaric+acid |
PubChem
CID
|
|
| UNII | |
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
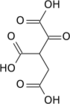
| C5H6O5 | |
| Molar mass | 146.098 g·mol−1 |
| Melting point | 115 °C (239 °F; 388 K) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
α-Ketoglutaric acid (2-oxoglutaric acid) is a keto acid.
Its carboxylate, α-ketoglutarate (also called 2-oxoglutarate), is an important biological compound. It is produced by deamination of glutamate, and is an intermediate in the Krebs cycle.
The term " ketoglutaric acid", when not further qualified, almost always refers to the alpha variant. β-Ketoglutaric acid varies only by the position of the ketone functional group, and is much less common.
Functions
Alanine transaminase
The enzyme alanine transaminase (also termed alanine aminotransferase and glutamate pyruvate transaminase) catalyzes, i.e. converts, 1) α-ketoglutarate to L-glutamate and 2) L-alanine to pyruvate. Both of these catalyzed reactions are fully reversible, i.e., run in both the forward and reverse directions. [2]
Krebs cycle
α-Ketoglutarate is a key intermediate in the Krebs cycle, coming after isocitrate and before succinyl CoA. Anaplerotic reactions can replenish the cycle at this juncture by synthesizing α-ketoglutarate from transamination of glutamate, or through action of glutamate dehydrogenase on glutamate.[ citation needed]
Formation of amino acids
Glutamine is synthesized from glutamate by glutamine synthetase, which utilizes adenosine triphosphate to form glutamyl phosphate; this intermediate is attacked by ammonia as a nucleophile giving glutamine and inorganic phosphate. Proline, arginine, and lysine (in some organisms) are other amino acids synthesized as well. [3] These three amino acids derive from glutamate with the addition of further steps or enzymes to facilitate reactions. [4]
Nitrogen transporter
Another function is to combine with nitrogen released in cells, therefore preventing nitrogen overload. [5]
α-Ketoglutarate is one of the most important nitrogen transporters in metabolic pathways. The amino groups of amino acids are attached to it (by transamination) and carried to the liver where the urea cycle takes place. [6]
α-Ketoglutarate is transaminated, along with glutamine, to form the excitatory neurotransmitter glutamate. Glutamate can then be decarboxylated (requiring vitamin B6) into the inhibitory neurotransmitter gamma-aminobutyric acid. [7]
It is reported that high ammonia and/or high nitrogen levels may occur with high protein intake, excessive aluminium exposure, Reye's syndrome, cirrhosis, and urea cycle disorder. [8]
It plays a role in detoxification of ammonia in brain. [9] [10] [11]
Relationship to molecular oxygen
Acting as a co-substrate for α-ketoglutarate-dependent hydroxylase, it also plays important function in oxidation reactions involving molecular oxygen.[ citation needed]
Molecular oxygen (O2) directly oxidizes many compounds to produce useful products in an organism, such as antibiotics, in reactions catalyzed by oxygenases. In many oxygenases, α-ketoglutarate helps the reaction by being oxidized with the main substrate. EGLN1, one of the α-ketoglutarate-dependent oxygenases, is an O2 sensor, informing the organism of the oxygen level in its environment.[ clarification needed]
In combination with molecular oxygen, alpha-ketoglutarate is one of the requirements for the hydroxylation of proline to hydroxyproline in the production of many collagens.[ citation needed]
Antioxidant
α-Ketoglutarate, which is released by several cell types, decreases the levels of hydrogen peroxide, and the α-ketoglutarate was depleted and converted to succinate in cell culture media. [12]
Supplementation
Longevity
Studies have linked α-ketoglutarate with increased lifespan in nematode worms [13] and increased healthspan/lifespan in mice. [14] [15] [16]
Immune regulation
A study showed that in glutamine deprived conditions, α-ketoglutarate promotes naïve CD4+ T cell differentiation into TH1 whilst inhibiting their differentiation into anti-inflammatory Treg cells. [17]
Enzyme cofactor
α-Ketoglutarate has been shown to be a cofactor for demethylases that contain the Jumonji C (JmjC) domain. [18] [19]
Production
α-Ketoglutarate can be produced by:
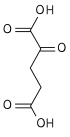
- Oxidative decarboxylation of isocitrate by isocitrate dehydrogenase
- Oxidative deamination of glutamate by glutamate dehydrogenase
- From galacturonic acid by the organism Agrobacterium tumefaciens [20]
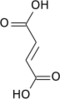
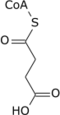
Alpha-ketoglutarate can be used to produce:
Interactive pathway map
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "TCACycle_WP78".
See also
References
- ^ Merck Index, 13th Edition, 5320.
- ^ Yang RZ, Park S, Reagan WJ, Goldstein R, Zhong S, Lawton M, Rajamohan F, Qian K, Liu L, Gong DW (February 2009). "Alanine aminotransferase isoenzymes: molecular cloning and quantitative analysis of tissue expression in rats and serum elevation in liver toxicity". Hepatology (Baltimore, Md.). 49 (2): 598–607. doi: 10.1002/hep.22657. PMC 2917112. PMID 19085960.
- ^ Ledwidge, Richard; Blanchard, John S. (1999). "The Dual Biosynthetic Capability of N-Acetylornithine Aminotransferase in Arginine and Lysine Biosynthesis†". Biochemistry. 38 (10): 3019–3024. doi: 10.1021/bi982574a. PMID 10074354.
- ^ Schousboe, Arne; Scafidi, Susanna; Bak, Lasse K.; Waagepetersen, Helle S.; McKenna, Mary C. (2014). "Glutamate Metabolism in the Brain Focusing on Astrocytes". Glutamate and ATP at the Interface of Metabolism and Signaling in the Brain. Advances in Neurobiology. Vol. 11. pp. 13–30. doi: 10.1007/978-3-319-08894-5_2. ISBN 978-3-319-08893-8. ISSN 2190-5215. PMC 4667713. PMID 25236722.
- ^ Katayama, Kazuhiro (2004-12-01). "Ammonia metabolism and hepatic encephalopathy". Hepatology Research. 30: 73–80. doi: 10.1016/j.hepres.2004.08.013. ISSN 1386-6346. PMID 15607143.
- ^ Li, Tao; Kootstra, Anna B.; Fotheringham, Ian G. (2002-07-01). "Nonproteinogenic α-Amino Acid Preparation Using Equilibrium Shifted Transamination". Organic Process Research & Development. 6 (4): 533–538. doi: 10.1021/op025518x. ISSN 1083-6160.
- ^ Kaneko, T.; Mizuno, N. (August 1994). "Glutamate-synthesizing enzymes in GABAergic neurons of the neocortex: a double immunofluorescence study in the rat". Neuroscience. 61 (4): 839–849. doi: 10.1016/0306-4522(94)90407-3. ISSN 0306-4522. PMID 7838383. S2CID 19178915.
- ^ "Hyperammonemia: What It Is, Causes, Symptoms & Treatment". Cleveland Clinic. Retrieved 2023-11-03.
- ^ "Does infectious fever relieve autistic behavior by releasing glutamine from skeletal muscles as provisional fuel?". Archived from the original on 2014-05-19. Retrieved 2014-05-19.
- ^ Ott, P; Clemmesen, O; Larsen, FS (Jul 2005). "Cerebral metabolic disturbances in the brain during acute liver failure: from hyperammonemia to energy failure and proteolysis". Neurochemistry International. 47 (1–2): 13–8. doi: 10.1016/j.neuint.2005.04.002. PMID 15921824. S2CID 916739.
- ^ Hares, P; James, IM; Pearson, RM (May–Jun 1978). "Effect of ornithine alpha ketoglutarate (OAKG) on the response of brain metabolism to hypoxia in the dog". Stroke: A Journal of Cerebral Circulation. 9 (3): 222–4. doi: 10.1161/01.STR.9.3.222. PMID 644619.
- ^ Long, L; Halliwell, B (2011). "Artefacts in cell culture: α-Ketoglutarate can scavenge hydrogen peroxide generated by ascorbate and epigallocatechin gallate in cell culture media". Biochemical and Biophysical Research Communications. 406 (1): 20–24. doi: 10.1016/j.bbrc.2011.01.091. PMID 21281600.
- ^ Chin, RM; Fu, X; Pai, MY; Vergnes, L; Hwang, H; Deng, G; Diep, S; Lomenick, B; Meli, VS; Monsalve, GC; Hu, E; Whelan, SA; Wang, JX; Jung, G; Solis, GM; Fazlollahi, F; Kaweeteerawat, C; Quach, A; Nili, M; Krall, AS; Godwin, HA; Chang, HR; Faull, KF; Guo, F; Jiang, M; Trauger, SA; Saghatelian, A; Braas, D; Christofk, HR; Clarke, CF; Teitell, MA; Petrascheck, M; Reue, K; Jung, ME; Frand, AR; Huang, J (2014). "The metabolite α-ketoglutarate extends lifespan by inhibiting ATP synthase and TOR". Nature. 510 (7505): 397–401. Bibcode: 2014Natur.510..397C. doi: 10.1038/nature13264. PMC 4263271. PMID 24828042.
-
^ Kaiser 1, Jocelyn (2020-09-01).
"Bodybuilding supplement promotes healthy aging and extends life span, at least in mice". Science | AAAS. Retrieved 2020-09-01.
{{ cite web}}: CS1 maint: numeric names: authors list ( link) - ^ "A metabolite produced by the body increases lifespan and dramatically compresses late-life morbidity in mice". BUCK. Retrieved 2020-09-01.
- ^ Shahmirzadi, Azar Asadi; Edgar, Daniel; Liao, Chen-Yu (2020-09-01). "Alpha-Ketoglutarate, an Endogenous Metabolite, Extends Lifespan and Compresses Morbidity in Aging Mice". Cell Metabolism. Retrieved 2020-09-20.
- ^ Klysz, Dorota; Tai, Xuguang (29 September 2015). "Glutamine-dependent α-ketoglutarate production regulates the balance between T helper 1 cell and regulatory T cell generation". Science Signaling. 8 (396): ra97. doi: 10.1126/scisignal.aab2610. PMID 26420908.
- ^ Tsukada, Yu-ichi; Fang, Jia; Erdjument-Bromage, Hediye; Warren, Maria E.; Borchers, Christoph H.; Tempst, Paul; Zhang, Yi (February 2006). "Histone demethylation by a family of JmjC domain-containing proteins". Nature. 439 (7078): 811–816. doi: 10.1038/nature04433. ISSN 1476-4687. PMID 16362057. S2CID 4415889.
- ^ Yamane, Kenichi; Toumazou, Charalambos; Tsukada, Yu-ichi; Erdjument-Bromage, Hediye; Tempst, Paul; Wong, Jiemin; Zhang, Yi (2006-05-05). "JHDM2A, a JmjC-containing H3K9 demethylase, facilitates transcription activation by androgen receptor". Cell. 125 (3): 483–495. doi: 10.1016/j.cell.2006.03.027. ISSN 0092-8674. PMID 16603237.
- ^ Richard, Peter; Hilditch, Satu (2009). "d-Galacturonic acid catabolism in microorganisms and its biotechnological relevance". Applied Microbiology and Biotechnology. 82 (4): 597–604. doi: 10.1007/s00253-009-1870-6. ISSN 0175-7598. PMID 19159926. S2CID 22671282.