| |
| Names | |
|---|---|
|
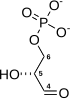
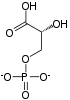
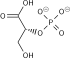
Preferred IUPAC name
(2R)-2-Hydroxy-3-(phosphonooxy)propanoic acid | |
| Identifiers | |
3D model (
JSmol)
|
|
| 3DMet | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| KEGG | |
PubChem
CID
|
|
CompTox Dashboard (
EPA)
|
|
| |
| |
| Properties | |
| C3H7O7P | |
| Molar mass | 186.06 g/mol |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
3-Phosphoglyceric acid (3PG, 3-PGA, or PGA) is the conjugate acid of 3-phosphoglycerate or glycerate 3-phosphate (GP or G3P). [1] This glycerate is a biochemically significant metabolic intermediate in both glycolysis and the Calvin-Benson cycle. The anion is often termed as PGA when referring to the Calvin-Benson cycle. In the Calvin-Benson cycle, 3-phosphoglycerate is typically the product of the spontaneous scission of an unstable 6-carbon intermediate formed upon CO2 fixation. Thus, two equivalents of 3-phosphoglycerate are produced for each molecule of CO2 that is fixed. [2] [3] [4] In glycolysis, 3-phosphoglycerate is an intermediate following the dephosphorylation ( reduction) of 1,3-bisphosphoglycerate. [4]: 14
Glycolysis
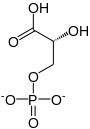
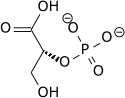
In the glycolytic pathway, 1,3-bisphosphoglycerate is dephosphorylated to form 3-phosphoglyceric acid in a coupled reaction producing two ATP via substrate-level phosphorylation. [5] The single phosphate group left on the 3-PGA molecule then moves from an end carbon to a central carbon, producing 2-phosphoglycerate. [5] [a] This phosphate group relocation is catalyzed by phosphoglycerate mutase, an enzyme that also catalyzes the reverse reaction. [6]
| 1,3-bisphospho-D-glycerate | 3-phosphoglycerate kinase | 3-phospho-D-glycerate | Phosphoglyceromutase | 2-phospho-D-glycerate | ||

|
|
| ||||
| ADP | ATP | |||||
|

| |||||
| ADP | ATP | |||||
| 3-phosphoglycerate kinase | Phosphoglyceromutase | |||||
Compound C00236 at KEGG Pathway Database. Enzyme 2.7.2.3 at KEGG Pathway Database. Compound C00197 at KEGG Pathway Database. Enzyme 5.4.2.1 at KEGG Pathway Database. Compound C00631 at KEGG Pathway Database.
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Calvin-Benson cycle
In the light-independent reactions (also known as the Calvin-Benson cycle), two 3-phosphoglycerate molecules are synthesized. RuBP, a 5-carbon sugar, undergoes carbon fixation, catalyzed by the rubisco enzyme, to become an unstable 6-carbon intermediate. This intermediate is then cleaved into two, separate 3-carbon molecules of 3-PGA. [7] One of the resultant 3-PGA molecules continues through the Calvin-Benson cycle to be regenerated into RuBP while the other is reduced to form one molecule of glyceraldehyde 3-phosphate (G3P) in two steps: the phosphorylation of 3-PGA into 1,3-bisphosphoglyceric acid via the enzyme phosphoglycerate kinase (the reverse of the reaction seen in glycolysis) and the subsequent catalysis by glyceraldehyde 3-phosphate dehydrogenase into G3P. [8] [9] [10] G3P eventually reacts to form the sugars such as glucose or fructose or more complex starches. [4]: 156 [8] [9]
Amino acid synthesis
Glycerate 3-phosphate (formed from 3-phosphoglycerate) is also a precursor for serine, which, in turn, can create cysteine and glycine through the homocysteine cycle. [11] [12] [13]
Measurement
3-phosphoglycerate can be separated and measured using paper chromatography [14] as well as with column chromatography and other chromatographic separation methods. [15] It can be identified using both gas-chromatography and liquid-chromatography mass spectrometry and has been optimized for evaluation using tandem MS techniques. [1] [16] [17]
See also
References
- ^ a b "3-Phosphoglyceric acid (HMDB0000807)". Human Metabolome Database. The Metabolomics Innovation Centre. Retrieved 23 May 2021.
- ^ Berg, J.M.; Tymoczko, J.L.; Stryer, L. (2002). Biochemistry (5th ed.). New York: W.H. Freeman and Company. ISBN 0-7167-3051-0.
- ^ Nelson, D.L.; Cox, M.M. (2000). Lehninger, Principles of Biochemistry (3rd ed.). New York: Worth Publishing. ISBN 1-57259-153-6.
- ^ a b c Leegood, R.C.; Sharkey, T.D.; von Caemmerer, S., eds. (2000). Photosynthesis: Physiology and Metabolism. Advances in Photosynthesis. Vol. 9. Kluwer Academic Publishers. doi: 10.1007/0-306-48137-5. ISBN 978-0-7923-6143-5. S2CID 266763949.
- ^ a b Rye, Connie; Wise, Robert; Jurukovski, Vladimir; DeSaix, Jean; Choi, Jung; Avissar, Yael (2016). "Glycolysis". Biology. OpenStax College.
- ^ Rose, Z.B.; Dube, S. (1976). "Rates of phosphorylation and dephosphorylation of phosphoglycerate mutase and bisphosphoglycerate synthase". Journal of Biological Chemistry. 251 (16): 4817–4822. doi: 10.1016/S0021-9258(17)33188-5. PMID 8447.
- ^ Andersson, I. (2008). "Catalysis and regulation in Rubisco". Journal of Experimental Botany. 59 (7): 1555–1568. doi: 10.1093/jxb/ern091. PMID 18417482.
- ^ a b Moran, L. (2007). "The Calvin Cycle: Regeneration". Sandwalk. Retrieved 11 May 2021.
- ^ a b Pettersson, G.; Ryde-Pettersson, Ulf (1988). "A mathematical model of the Calvin photosynthesis cycle". European Journal of Biochemistry. 175 (3): 661–672. doi: 10.1111/j.1432-1033.1988.tb14242.x. PMID 3137030.
- ^ Fridlyand, L.E.; Scheibe, R. (1999). "Regulation of the Calvin cycle for CO2 fixation as an example for general control mechanisms in metabolic cycles". Biosystems. 51 (2): 79–93. doi: 10.1016/S0303-2647(99)00017-9. PMID 10482420.
- ^ Igamberdiev, A.U.; Kleczkowski, L.A. (2018). "The Glycerate and Phosphorylated Pathways of Serine Synthesis in Plants: The Branches of Plant Glycolysis Linking Carbon and Nitrogen Metabolism". Frontiers in Plant Science. 9 (318): 318. doi: 10.3389/fpls.2018.00318. PMC 5861185. PMID 29593770.
- ^ Ichihara, A.; Greenberg, D.M. (1955). "Pathway of Serine Formation from Carbohydrate in Rat Liver". PNAS. 41 (9): 605–609. Bibcode: 1955PNAS...41..605I. doi: 10.1073/pnas.41.9.605. JSTOR 89140. PMC 528146. PMID 16589713.
- ^ Hanford, J.; Davies, D.D. (1958). "Formation of Phosphoserine from 3-Phosphoglycerate in Higher Plants". Nature. 182 (4634): 532–533. Bibcode: 1958Natur.182..532H. doi: 10.1038/182532a0. S2CID 4192791.
- ^ Cowgill, R.W.; Pizer, L.I. (1956). "Purification and Some Properties of Phosphorylglyceric Acid Mutase from Rabbit Skeletal Muscle". Journal of Biological Chemistry. 223 (2): 885–895. doi: 10.1016/S0021-9258(18)65087-2. PMID 13385236.
- ^ Hofer, H.W. (1974). "Separation of glycolytic metabolites by column chromatography". Analytical Biochemistry. 61 (1): 54–61. doi: 10.1016/0003-2697(74)90332-7. PMID 4278264.
- ^ Shibayama, J.; Yuzyuk, T.N.; Cox, J.; et al. (2015). "Metabolic Remodeling in Moderate Synchronous versus Dyssynchronous Pacing-Induced Heart Failure: Integrated Metabolomics and Proteomics Study". PLOS ONE. 10 (3): e0118974. Bibcode: 2015PLoSO..1018974S. doi: 10.1371/journal.pone.0118974. PMC 4366225. PMID 25790351.
- ^ Xu, J.; Zhai, Y.; Feng, L. (2019). "An optimized analytical method for cellular targeted quantification of primary metabolites in tricarboxylic acid cycle and glycolysis using gas chromatography-tandem mass spectrometry and its application in three kinds of hepatic cell lines". Journal of Pharmaceutical and Biomedical Analysis. 171: 171–179. doi: 10.1016/j.jpba.2019.04.022. PMID 31005043. S2CID 125170446.
- ^ Note that 3-phosphoglycerate and 2-phosphoglycerate are isomers of one another