Total intravenous anesthesia (TIVA) refers to the intravenous administration of anesthetic agents to induce a temporary loss of sensation or awareness. The first study of TIVA was done in 1872 using chloral hydrate, [1] and the common anesthetic agent propofol was licensed in 1986. TIVA is currently employed in various procedures as an alternative technique of general anesthesia in order to improve post-operative recovery.
TIVA is maintained by intravenous infusion devices and assisted by electroencephalography (EEG) monitoring. These techniques facilitate the use of propofol, etomidate, ketamine, and other intravenous anesthetic agents. During or after TIVA, patients may be subjected to an elevated risk of anesthesia awareness, hyperalgesia and neurotoxicity. [2] Considering these risks, special consideration is given to obese, elderly and pediatric patients.[ citation needed]
History
In the mid-19th century, specific equipment was developed to enable intravenous anesthesia. Francis Rynd developed the hollow needle in 1845, [1] and Charles Gabriel Pravaz developed the syringe in 1853, [1] which allowed drugs to be administered intravenously.
Using this new mode of delivery, many chemical compounds were tested as intravenous anesthetics. This was pioneered by Pierre-Cyprien Oré in 1872, who reported using chloral hydrate as an intravenous anesthetic. [1] However, these early trials were associated with high mortality. [1] Hedonal was later developed in 1909 for general anesthesia, although with limited success due to its long duration of effect. [3] These insufficiencies encouraged the development of paraldehyde by Noel & Souttar, [4] magnesium sulfate by Peck & Meltzer [5] as well as ethanol by Nakagawa [6] as intravenous anesthetic agents.
Propofol (di-isopropyl phenol) was synthesized by Glen and colleagues in the early 1970s, [7] but its first formulations were temporarily withdrawn due to a number of adverse reactions during clinical studies. [1] In 1983, a lipid emulsion formulation of propofol was available, which carried great potential during clinical trials. [8] It was licensed for use in Europe in 1986 and received FDA approval in the US in 1989. [1] Propofol is now used worldwide with a well-defined pharmacological profile for a variety of medical uses.
Medical uses
TIVA is used to induce general anesthesia while avoiding the disadvantages of volatile anesthesia (and traditional inhalation agents). [9] Intravenous anesthetic agents are titrated at safe doses to maintain stage III surgical anesthesia (unconsciousness, amnesia, immobility, and absence of response to noxious stimulation). [10] The use of TIVA is advantageous in cases where volatile anesthesia is of high risk or is impossible, such as cases involving morbidly obese patients. [11] [12] TIVA has also been used for anesthetic delivery at sites of trauma such as serious accidents, disasters and wars. [1]
The overall goals of TIVA include: [13]
- Smooth induction of anesthesia
- Reliable and measurable maintenance of anesthesia
- Rapid emergence out of the effects of infused drugs as soon as the infusion is terminated.
Propofol-based TIVA significantly improves post-operative recovery profile and comfort, minimizes nausea and vomiting, facilitates rapid recovery, greater hemodynamic stability, preservation of hypoxic pulmonary vasoconstriction, reduction in intracerebral pressure, and reduces the risk of organ toxicity. [14] Despite these advantages, it accounts for a small proportion of general anesthetics due to the relatively expensive cost of preparation and maintenance. [15]
Techniques
Dosing considerations
The doses for intravenous sedative-hypnotic and adjuvant agents vary individually. Pharmacodynamic and pharmacokinetic factors need to be considered for each patient (e.g. patients with impaired kidney or liver function, blood abnormalities and myocardial dysfunction, etc.) [16] There are also risks of adverse effects related to doses such as hypotension and respiratory depression. [17] In terms of adjuvant agents, the co-administration of anesthetic drugs from different classes often produce synergistic hypnotic effects. [18] [19] This is especially common for agents acting on gamma-aminobutyric acid (GABA) receptors that are combined with drugs acting on different types of receptors. [20] [21]
The drug interactions between sedative-hypnotic agents and adjuvant agents suggest that dosing regimens cannot be fixed. [19] Instead, dosing should be based on adjusted body weight or estimated lean body weight, especially for obese patients. It is recommended that drug doses be titrated in brief intervals (around 20 to 60 seconds). [22]
Equipment
The delivery of intravenous anesthetics is dependent on different types of infusion devices. Examples of infusion devices include smart pumps, syringe pumps and target-controlled infusion (TCI) devices. [23]

Smart pumps are commonly used to administer potent anesthetics and various vasoactive drugs such as vasopressors, inotropes, vasodilators, which need to be continuously titrated in the operating room. [24] Smart pumps are advantageous since they administer safe doses with a programmed infusion rate within pre-existing limits based on the institutional standardized medication library. [25]
Syringe pumps are smaller infusion pumps that allow the administration of small amounts of induction agents at a precise rate. [26] The accuracy of syringe pumps is dependent on the selection of syringes during pump programming. Most pumps are able to identify the size of the syringe automatically when the syringe manufacturer's name is input correctly. [27]
Target-controlled infusion (TCI) systems are assisted by computer systems that make use of pharmacokinetic and pharmacodynamic modelling to maintain a target concentration of anesthetic in the brain. [28] [29] TCI requires clinicians to input a target concentration for an anesthetic or other agents, from which the computer calculates the amount of agent required for the input concentration, then the infusion pump delivers the calculated bolus dose. [30] Subsequently, the computer continuously recalculates how much drug is in the system and influences the amount of drug required to maintain the desired concentration at the effect site. [31]
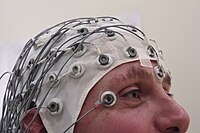
Maintenance
During TIVA, the continuous assessment of heart rate, blood pressure, and state of consciousness is essential when titrating anesthetic agents. [20] [23] Processed electroencephalogram (EEG) monitoring is used to assess anesthetic depth. [32] However, there is 30 seconds of lag time between the subject's state of consciousness and the processed EEG signal. This limits its usefulness during the induction of anesthesia. [20] [21]
Intravenous agents
Propofol, etomidate and ketamine are common intravenous sedative-hypnotic agents for the induction of TIVA. [19] Their highly lipophilic nature allows the rapid onset of anesthesia upon intravenous injection. [17] It also enables penetration through the blood–brain barrier and effective perfusion to the brain. However, the rapid redistribution of these agents from the brain to other muscle and fat tissues causes it to have a short duration of action. Adjuvant agents are typically administered in addition to sedative-hypnotic agents to supplement the induction of TIVA. [17]
Sedative-hypnotic agents
Propofol
Propofol is usually the selected sedative-hypnotic agent to maintain general anesthesia through TIVA because of its rapid onset and offset, beneficial properties and few adverse effects. [33] Its rapid onset of action is due to its high lipid-solubility, rapid redistribution from the brain to other parts of the body, and rapid clearance (20 to 30 mL/kg/minute). [33] Most propofol is conjugated in the liver with pharmacologically inactive metabolites. [33] Although it has a long terminal elimination half-life of 4 to 30 hours, plasma concentrations remain low after the typical induction dose. [33]
Its advantages include “ antiemetic, antipruritic, bronchodilatory, and anticonvulsant properties”, [34] which makes it suitable for patients with kidney or liver insufficiency. [35] Potential adverse effects of propofol include hypotension and respiratory depression caused by inadequate dosing, pain on injection, and risk of contamination. [36]
Etomidate
Etomidate is suitable for patients with hemodynamic instability since it does not compromise blood pressure, cardiac output, or heart rate. [37] [38] Its advantages include anticonvulsant properties and hemodynamic stability. Potential adverse effects include a higher incidence of postoperative nausea and vomiting, transient acute adrenal insufficiency, pain during injection, involuntary myoclonic movements, absence of analgesic effects and mild increases in airway resistance. [37] [39]
Ketamine
Ketamine is suitable for hypotensive patients, or patients with risks of developing hypotension (e.g. those who have hypovolemia, hemorrhage, sepsis or severe cardiovascular compromise). [40] [41] This is because ketamine is associated with increased blood pressure, heart rate and cardiac output. [42] Its advantages include profound analgesic properties, bronchodilation, and the ability to maintain airway reflexes and respiratory drive. [43] It could also be induced via the intramuscular route if TIVA access gets lost. However, its potential adverse effects impact cardiovascular and neurological functions.
Potential adverse effects on cardiovascular activities are listed below: [44]
- Increase in myocardial oxygen demand due to a rise in heart rate, blood pressure and cardiac output
- Increase in pulmonary arterial pressure, which could be fatal in patients with ischemic heart disease, systemic or pulmonary hypertension
- Increase in the toxicity of cocaine and tricyclic antidepressants on cardiovascular structures
- Exacerbates hypertension, tachycardia arrhythmia in pheochromocytoma
- Though rare, direct mild myocardial depressant effects
Potential adverse effects on neurological activities are listed below: [44]
- Higher incidence of psychotomimetic effects
- Increase in cerebral blood flow and intracranial pressure, which may increase the cerebral metabolic rate of oxygen
- Unique EEG effects might lead to misinterpretation of processed EEG values
Adjuvant agents
Opioid, lidocaine and midazolam are adjuvant agents frequently administered to minimize pain during the injection of the induction agents. [45] [46] They are also used to lessen the sympathetic stress response, cough reflex during laryngoscopy or intubation, and supplement sedation by synergistic effects. [47] [48] The dose of sedative-hypnotic agents should be reduced due to the synergistic effects when combined with adjuvant agents. [48]
Choice of specific adjuvant agents is dependent upon the patient and procedure-specific factors. [49] Opioid is a commonly administered adjuvant agent as the analgesic component of TIVA. However, when used with propofol, it might exacerbate the adverse hypotensive effects. [48] Other potential adverse effects include respiratory depression, bradycardia, delirium and potential for acute tolerance. [50]
Risks and complications
Accidental awareness during general anesthesia (AAGA)
Patients under TIVA have a higher risk of AAGA. Unlike inhaled anesthetic agents, intravenous agents do not have an indicative end-tidal anesthetic concentration (ETAC) for the monitoring of administered drugs, so the determination of successful delivery is usually left to the anesthetist's clinical judgment. [2]
The high incidence of AAGA with TIVA can be attributed to several factors. Firstly, the target concentration of anesthetic agents required to maintain unresponsiveness is not well understood. [2] Although there have been studies aiming to establish the target concentration of propofol, there is a high degree of variability with the established dosing range. [51] Secondly, intravenous delivery may be impaired by lax monitoring of the intravenous catheter and the insertion site. [2] Thirdly, the use of neuromuscular blockades is a risk factor of AAGA and also hinders communication of distress in the case of accidental awareness. [2]
Opioid-induced hyperalgesia
TIVA techniques which involve the continued administration of opioids (e.g. remifentanil) at high doses can cause opioid-induced hyperalgesia. [52] This may lead to difficult postoperative pain control, as patients with hyperalgesia experience increased chronic pain and require more analgesics following surgery. [51]
Neurotoxicity
Prolonged anesthetic exposure can result in the death of neural cells and defective synaptogenesis, [53] caused by increased expression of neurologically harmful substances. [2] The resulting neurologic injuries may lead to a persistent subtle decline of cognitive abilities, especially in elderly or very young patients. [2] Animal studies suggest that propofol may have similar neurotoxic properties as it is associated with apoptotic degeneration of oligodendrocytes. [2]
Special populations
Obese patients
Obese patients present technical and physiological challenges to TIVA. Physical tasks such as surgical positioning, intravenous insertion and ventilation are complicated by excess fat. [54] Associated physiological and pharmacological changes include higher susceptibility to hypoxemia, decrease in resting metabolic rate and lower cardiac output per kg body weight. [55] The use of dosing models derived from non-obese patients is therefore unsuitable for obese patients. [56]
Even within the obese population, the large variability between individuals limits the accuracy of pharmacokinetic models in predicting and informing anesthetic titration. [54]
Pediatrics
Infants differ from adults in the consideration of pharmacokinetics, pharmacodynamics and side effects. [57] In terms of pharmacokinetics, protein binding, organ function and body composition are significantly different. [58] Pharmacodynamic effects such as the capacity of target organs to respond to drugs are also changed. [57] Based on this knowledge, doses are adjusted to achieve optimal clinical response and avoid toxicity in pediatric patients. [59] Generally, clearance (drug elimination from the body) is greater in children due to the nonlinear scaling between body size and function. [57]
Elderly patients
Aging is associated with an increase in fat and a reduction in lean body mass and total body water. [60] These factors increase the volume of distribution of lipid-soluble drugs, lower their plasma concentration and delay elimination. [61] Aged patients typically have a higher sensitivity to drug action due to a reduction in the initial drug clearance, resulting in higher plasma concentration and hence greater initial drug effect. [61]
References
- ^ a b c d e f g h Sear JW (2017). "When and How Did It All Begin? A Brief History of Intravenous Anesthesia". In Absalom A, Mason K (eds.). Total Intravenous Anesthesia and Target Controlled Infusions. Cham: Springer International Publishing. pp. 3–8. doi: 10.1007/978-3-319-47609-4_1. ISBN 978-3-319-47607-0.
- ^ a b c d e f g h Johnson KB (2017). "Advantages, Disadvantages, and Risks of TIVA/TCI". Total Intravenous Anesthesia and Target Controlled Infusions. Cham: Springer International Publishing. pp. 621–631. doi: 10.1007/978-3-319-47609-4_32. ISBN 978-3-319-47607-0.
- ^ Kissin I, Wright AJ (August 1988). "The introduction of hedonal: a Russian contribution to intravenous anesthesia". Anesthesiology. 69 (2): 242–245. doi: 10.1097/00000542-198808000-00014. PMID 3044190. S2CID 39156836.
- ^ Noel H, Souttar HS (January 1913). "IV. The Anaesthetic Effects of the Intravenous Injection of Paraldehyde". Annals of Surgery. 57 (1): 64–67. doi: 10.1097/00000658-191301000-00004. PMC 1407440. PMID 17862957.
- ^ Peck CH, Meltzer SJ (1916-10-14). "Anesthesia in Human Beings by Intravenous Injection of Magnesium Sulphate". Journal of the American Medical Association. LXVII (16): 1131. doi: 10.1001/jama.1916.02590160009004. ISSN 0002-9955.
- ^ Naragawa K (1921). "Experimentelle Studien über die intravenöse Infusionsnarkose mittels Alkohols". The Tohoku Journal of Experimental Medicine. 2 (1): 81–126. doi: 10.1620/tjem.2.81. ISSN 1349-3329.
- ^ James R, Glen JB (December 1980). "Synthesis, biological evaluation, and preliminary structure-activity considerations of a series of alkylphenols as intravenous anesthetic agents". Journal of Medicinal Chemistry. 23 (12): 1350–1357. doi: 10.1021/jm00186a013. PMID 7452689.
- ^ Cummings GC, Dixon J, Kay NH, Windsor JP, Major E, Morgan M, et al. (December 1984). "Dose requirements of ICI 35,868 (propofol, 'Diprivan') in a new formulation for induction of anaesthesia". Anaesthesia. 39 (12): 1168–1171. doi: 10.1111/j.1365-2044.1984.tb06425.x. PMID 6335003. S2CID 26653276.
- ^ Murray WB (2009-11-01). Provider Needs for Distributed Simulation Education System in Total Intravenous Anesthesia and Target Controlled Infusion (Report). Fort Belvoir, VA. doi: 10.21236/ada542258.
- ^ van den Berg JP, Vereecke HE, Proost JH, Eleveld DJ, Wietasch JK, Absalom AR, Struys MM (January 2017). "Pharmacokinetic and pharmacodynamic interactions in anaesthesia. A review of current knowledge and how it can be used to optimize anaesthetic drug administration". British Journal of Anaesthesia. 118 (1): 44–57. doi: 10.1093/bja/aew312. PMID 28039241.
- ^ De Jong A, Verzilli D, Geniez M, Chanques G, Nocca D, Jaber S (May 2018). "[Why is the morbidly obese patient at high risk of anesthetic complications?]". Presse Médicale (in French). 47 (5): 453–463. doi: 10.1016/j.lpm.2018.01.016. PMID 29609909.
- ^ De Jong A, Rollé A, Souche FR, Yengui O, Verzilli D, Chanques G, et al. (April 2020). "How can I manage anaesthesia in obese patients?". Anaesthesia, Critical Care & Pain Medicine. 39 (2): 229–238. doi: 10.1016/j.accpm.2019.12.009. PMID 32068132. S2CID 211159649.
- ^ Yuill G, Simpson M (2002). "An introduction to total intravenous anaesthesia". BJA CEPD Reviews. 2 (1): 24–26. doi: 10.1093/bjacepd/2.1.24. ISSN 1472-2615.
- ^ Engbers FH (2000). "Total Intravenous Anaesthesia: The Equipment". On the Study and Practice of Intravenous Anaesthesia. Dordrecht: Springer Netherlands. pp. 71–87. doi: 10.1007/978-94-015-9604-6_6. ISBN 978-90-481-5366-4.
- ^ Smith I (2003). "Total intravenous anaesthesia: is it worth the cost?". CNS Drugs. 17 (9): 609–619. doi: 10.2165/00023210-200317090-00001. PMID 12828497. S2CID 53919586.
- ^ Reekers M, Boer F, Vuyk J (2003). "Basic Concepts of Recirculatory Pharmacokinetic Modelling". Advances in Modelling and Clinical Application of Intravenous Anaesthesia. Advances in Experimental Medicine and Biology. Vol. 523. Boston, MA: Springer US. pp. 19–26. doi: 10.1007/978-1-4419-9192-8_2. ISBN 978-1-4613-4830-6. PMID 15088836. S2CID 12457898.
- ^ a b c Garcia P, Whalin MK, Sebel PS (2013). "Intravenous Anesthetics". Pharmacology and Physiology for Anesthesia. Elsevier. pp. 137–158. doi: 10.1016/b978-1-4377-1679-5.00009-0. ISBN 978-1-4377-1679-5.
- ^ Prabhakar A, Lambert T, Kaye RJ, Gaignard SM, Ragusa J, Wheat S, et al. (December 2019). "Adjuvants in clinical regional anesthesia practice: A comprehensive review". Best Practice & Research. Clinical Anaesthesiology. 33 (4): 415–423. doi: 10.1016/j.bpa.2019.06.001. PMID 31791560. S2CID 208611069.
- ^ a b c Hendrickx JF, Eger EI, Sonner JM, Shafer SL (August 2008). "Is synergy the rule? A review of anesthetic interactions producing hypnosis and immobility". Anesthesia and Analgesia. 107 (2): 494–506. doi: 10.1213/ane.0b013e31817b859e. PMID 18633028. S2CID 8125002.
- ^ a b c Bowdle TA (2009). "Can We Prevent Recall during Anesthesia?". Evidence-Based Practice of Anesthesiology. Elsevier. pp. 291–295. doi: 10.1016/b978-1-4160-5996-7.00043-2. ISBN 978-1-4160-5996-7.
- ^ a b Jäntti V, Sloan TB (2008). "EEG and anesthetic effects". Intraoperative Monitoring of Neural Function. Handbook of Clinical Neurophysiology. Vol. 8. Elsevier. pp. 77–93. doi: 10.1016/s1567-4231(07)08004-5. ISBN 978-0-444-51824-8.
- ^ MacCallum CA, Russo EB (March 2018). "Practical considerations in medical cannabis administration and dosing". European Journal of Internal Medicine. 49: 12–19. doi: 10.1016/j.ejim.2018.01.004. PMID 29307505.
- ^ a b Nimmo AF, Absalom AR, Bagshaw O, Biswas A, Cook TM, Costello A, et al. (February 2019). "Guidelines for the safe practice of total intravenous anaesthesia (TIVA): Joint Guidelines from the Association of Anaesthetists and the Society for Intravenous Anaesthesia". Anaesthesia. 74 (2): 211–224. doi: 10.1111/anae.14428. PMID 30378102. S2CID 53107969.
- ^ Ohashi K, Dalleur O, Dykes PC, Bates DW (December 2014). "Benefits and risks of using smart pumps to reduce medication error rates: a systematic review". Drug Safety. 37 (12): 1011–1020. doi: 10.1007/s40264-014-0232-1. PMID 25294653. S2CID 9308973.
- ^ Kan K, Levine WC (2021-01-01). "Infusion Pumps". Anesthesia Equipment: 351–367. doi: 10.1016/B978-0-323-67279-5.00016-9. ISBN 9780323672795. S2CID 243158761.
- ^ Seo KS, Lee K (September 2016). "Smart syringe pumps for drug infusion during dental intravenous sedation". Journal of Dental Anesthesia and Pain Medicine. 16 (3): 165–173. doi: 10.17245/jdapm.2016.16.3.165. PMC 5586553. PMID 28884149.
- ^ Alexovič M, Horstkotte B, Šrámková I, Solich P, Sabo J (2017-01-01). "Automation of dispersive liquid–liquid microextraction and related techniques. Approaches based on flow, batch, flow-batch and in-syringe modes". TrAC Trends in Analytical Chemistry. 86: 39–55. doi: 10.1016/j.trac.2016.10.003. ISSN 0165-9936.
- ^ Absalom AR, Glen JI, Zwart GJ, Schnider TW, Struys MM (January 2016). "Target-Controlled Infusion: A Mature Technology". Anesthesia and Analgesia. 122 (1): 70–78. doi: 10.1213/ane.0000000000001009. PMID 26516798. S2CID 41023659.
- ^ Struys MM, De Smet T, Glen JI, Vereecke HE, Absalom AR, Schnider TW (January 2016). "The History of Target-Controlled Infusion". Anesthesia and Analgesia. 122 (1): 56–69. doi: 10.1213/ane.0000000000001008. PMID 26516804. S2CID 39391491.
- ^ Morton NA (2012-12-18). "Total Intravenous Anesthesia (TIVA) and Target Controlled Infusions (TCI) in Children". Current Anesthesiology Reports. 3 (1): 37–41. doi: 10.1007/s40140-012-0005-2. ISSN 2167-6275.
- ^ Schnider TW, Minto CF, Struys MM, Absalom AR (January 2016). "The Safety of Target-Controlled Infusions". Anesthesia and Analgesia. 122 (1): 79–85. doi: 10.1213/ane.0000000000001005. PMID 26516801. S2CID 25742249.
- ^ "Correspondence". Anaesthesia and Intensive Care. 30 (6): 813–818. 2002. doi: 10.1177/0310057x0203000618. ISSN 0310-057X.
- ^ a b c d McGrenaghan E, Wilson M (2019). "Total intravenous anaesthesia". Anaesthesia & Intensive Care Medicine. 20 (2): 130–135. doi: 10.1016/j.mpaic.2018.12.010. ISSN 1472-0299. S2CID 81449338.
- ^ Shafer SL (1993). "Advances in propofol pharmacokinetics and pharmacodynamics". Journal of Clinical Anesthesia. 5 (6 Suppl 1): 14S–21S. doi: 10.1016/0952-8180(93)90003-w. PMID 8292364.
- ^ Gray PA, Park GR, Cockshott ID, Douglas EJ, Shuker B, Simons PJ (January 1992). "Propofol metabolism in man during the anhepatic and reperfusion phases of liver transplantation". Xenobiotica; the Fate of Foreign Compounds in Biological Systems. 22 (1): 105–114. doi: 10.3109/00498259209053107. PMID 1615701.
- ^ Yeoh CJ, Hwang NC (August 2020). "Volatile Anesthesia Versus Total Intravenous Anesthesia During Cardiopulmonary Bypass: A Narrative Review on the Technical Challenges and Considerations". Journal of Cardiothoracic and Vascular Anesthesia. 34 (8): 2181–2188. doi: 10.1053/j.jvca.2020.03.013. PMID 32360007. S2CID 216248614.
- ^ a b Hulsman N, Hollmann MW, Preckel B (June 2018). "Newer propofol, ketamine, and etomidate derivatives and delivery systems relevant to anesthesia practice". Best Practice & Research. Clinical Anaesthesiology. 32 (2): 213–221. doi: 10.1016/j.bpa.2018.08.002. PMID 30322461. S2CID 53503078.
- ^ Forman SA (March 2011). "Clinical and molecular pharmacology of etomidate". Anesthesiology. 114 (3): 695–707. doi: 10.1097/aln.0b013e3181ff72b5. PMC 3108152. PMID 21263301.
- ^ Hohl CM, Kelly-Smith CH, Yeung TC, Sweet DD, Doyle-Waters MM, Schulzer M (August 2010). "The effect of a bolus dose of etomidate on cortisol levels, mortality, and health services utilization: a systematic review". Annals of Emergency Medicine. 56 (2): 105–13.e5. doi: 10.1016/j.annemergmed.2010.01.030. PMID 20346542.
- ^ Schwenk ES, Viscusi ER, Buvanendran A, Hurley RW, Wasan AD, Narouze S, et al. (July 2018). "Consensus Guidelines on the Use of Intravenous Ketamine Infusions for Acute Pain Management From the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists". Regional Anesthesia and Pain Medicine. 43 (5): 456–466. doi: 10.1097/aap.0000000000000806. PMC 6023582. PMID 29870457.
- ^ Morris C, Perris A, Klein J, Mahoney P (May 2009). "Anaesthesia in haemodynamically compromised emergency patients: does ketamine represent the best choice of induction agent?". Anaesthesia. 64 (5): 532–539. doi: 10.1111/j.1365-2044.2008.05835.x. PMID 19413824. S2CID 16287093.
- ^ Zhou JY, Hamilton P, Macres S, Peña M, Tang S (December 2020). "Update on Ketamine". Advances in Anesthesia. 38: 97–113. doi: 10.1016/j.aan.2020.07.005. PMID 34106842. S2CID 224909438.
- ^ Rascón-Martínez DM, Carrillo-Torres O, Ramos-Nataren RG, Rendón-Jaramillo L (2018). "Advantages of ketamine as a perioperative analgesic". Revista Médica del Hospital General de México. 81 (4): 253–261. doi: 10.1016/j.hgmx.2016.10.007. ISSN 0185-1063.
- ^ a b Hudetz JA, Pagel PS (February 2010). "Neuroprotection by ketamine: a review of the experimental and clinical evidence". Journal of Cardiothoracic and Vascular Anesthesia. 24 (1): 131–142. doi: 10.1053/j.jvca.2009.05.008. PMID 19640746.
- ^ Prabhakar H (28 March 2017). Essentials of neuroanesthesia. ISBN 978-0-12-805299-0. OCLC 959033559.
- ^ Reves JG, Fragen R, Vinik R, Greenblatt D (1985). "Midazolam". Anesthesiology. 62 (3): 310–324. doi: 10.1097/00000542-198503000-00017. ISSN 0003-3022. S2CID 28308031.
- ^ Shaban AA (2016). "Effect of small dose propofol or midazolam to prevent laryngospasm and coughing following oropharyngeal surgeries: Randomized controlled trial". Egyptian Journal of Anaesthesia. 32 (1): 13–19. doi: 10.1016/j.egja.2015.09.008. ISSN 1110-1849.
- ^ a b c Marinella MA (October 1997). "Propofol for sedation in the intensive care unit: essentials for the clinician". Respiratory Medicine. 91 (9): 505–510. doi: 10.1016/s0954-6111(97)90082-2. PMID 9415349.
- ^ Winacoo JN, Maykel JA (February 2009). "Operative anesthesia and pain control". Clinics in Colon and Rectal Surgery. 22 (1): 41–46. doi: 10.1055/s-0029-1202885. PMC 2780232. PMID 20119555.
- ^ Warner LO, Balch DR, Davidson PJ (June 1997). "Is intravenous lidocaine an effective adjuvant for endotracheal intubation in children undergoing induction of anesthesia with halothane-nitrous oxide?". Journal of Clinical Anesthesia. 9 (4): 270–274. doi: 10.1016/s0952-8180(97)00003-2. PMID 9195347.
- ^ a b Reves JG, Glass PS, Lubarsky DA, McEvoy MD, Martinez-Ruiz R (2010). "Intravenous Anesthetics". Miller's Anesthesia. Elsevier. pp. 719–768. doi: 10.1016/b978-0-443-06959-8.00026-1. ISBN 978-0-443-06959-8.
- ^ Hayhurst CJ, Durieux ME (February 2016). "Differential Opioid Tolerance and Opioid-induced Hyperalgesia: A Clinical Reality". Anesthesiology. 124 (2): 483–488. doi: 10.1097/aln.0000000000000963. PMID 26594912.
- ^ Creeley CE, Olney JW (February 2010). "The young: neuroapoptosis induced by anesthetics and what to do about it". Anesthesia and Analgesia. 110 (2): 442–448. doi: 10.1213/ANE.0b013e3181c6b9ca. PMID 19955510. S2CID 14304084.
- ^ a b Sepúlveda V PO, Cortínez LI (2017). "Intravenous Anesthesia in Obese Patients". Total Intravenous Anesthesia and Target Controlled Infusions. Cham: Springer International Publishing. pp. 429–440. doi: 10.1007/978-3-319-47609-4_24. ISBN 978-3-319-47607-0.
- ^ Cheymol G (September 2000). "Effects of obesity on pharmacokinetics implications for drug therapy". Clinical Pharmacokinetics. 39 (3): 215–231. doi: 10.2165/00003088-200039030-00004. PMID 11020136. S2CID 20056863.
- ^ Eleveld DJ, Proost JH, Absalom AR, Struys MM (November 2011). "Obesity and allometric scaling of pharmacokinetics". Clinical Pharmacokinetics. 50 (11): 751–753. doi: 10.2165/11594080-000000000-00000. PMID 21973272. S2CID 41800410.
- ^ a b c Anderson BJ (2017). "Pharmacokinetics and Pharmacodynamics in the Pediatric Patient". Total Intravenous Anesthesia and Target Controlled Infusions. Cham: Springer International Publishing. pp. 441–516. doi: 10.1007/978-3-319-47609-4_25. ISBN 978-3-319-47607-0.
- ^ Kearns GL, Abdel-Rahman SM, Alander SW, Blowey DL, Leeder JS, Kauffman RE (September 2003). "Developmental pharmacology--drug disposition, action, and therapy in infants and children". The New England Journal of Medicine. 349 (12): 1157–1167. doi: 10.1056/nejmra035092. PMID 13679531.
- ^ Anderson BJ, Holford NH (September 2013). "Understanding dosing: children are small adults, neonates are immature children". Archives of Disease in Childhood. 98 (9): 737–744. doi: 10.1136/archdischild-2013-303720. PMID 23832061. S2CID 31723102.
- ^ Hughes VA, Frontera WR, Roubenoff R, Evans WJ, Singh MA (August 2002). "Longitudinal changes in body composition in older men and women: role of body weight change and physical activity". The American Journal of Clinical Nutrition. 76 (2): 473–481. doi: 10.1093/ajcn/76.2.473. PMID 12145025.
- ^ a b Servin FS (2017). "TCI in Special Patients Groups: The Elderly and Obese". Total Intravenous Anesthesia and Target Controlled Infusions. Cham: Springer International Publishing. pp. 571–578. doi: 10.1007/978-3-319-47609-4_29. ISBN 978-3-319-47607-0.