| |||
| |||
| Names | |||
|---|---|---|---|
|
Preferred IUPAC name
Hydroxy nitrate | |||
| Systematic IUPAC name | |||
| Identifiers | |||
3D model (
JSmol)
|
|||
| ChemSpider | |||
PubChem
CID
|
|||
| |||
| Properties | |||
| HNO4 | |||
| Molar mass | 79.01224 g/mol | ||
| Conjugate base | Peroxynitrate | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |||
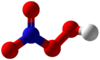
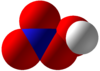
Peroxynitric acid or peroxonitric acid is a
chemical compound with the
formula HNO
4. It is an
oxyacid of nitrogen, after
peroxynitrous acid.
Preparation
Peroxynitrate, the conjugate base of peroxynitric acid, is formed rapidly during decomposition of peroxynitrite in neutral conditions. [4]
Atmospheric chemistry
Peroxynitric acid is formed in the atmosphere, although it is unstable, it is important as a reservoir for NO2 through the reversible radical reaction: [5]
- HO
2NO
2 ⇌ HO•
2 + NO•
2
References
- ^ "Peroxynitric Acid - Compound Summary".
- ^ "peroxynitric acid". PubChem. Retrieved 9 December 2012.
- ^ "125239-87-4". ChemIndex. Retrieved 9 December 2012.
- ^ Miyamoto, S; Ronsein, GE; Corrêa, TC; Martinez, GR; Medeiros, MH; Di Mascio, P (2009). "Direct evidence of singlet molecular oxygen generation from peroxynitrate, a decomposition product of peroxynitrite". Dalton Trans (29): 5720–9. doi: 10.1039/b905560f. PMID 20449086.
- ^ Finlayson-Pitts, Barbara J.; Pitts, James N. (2000), Chemistry of the Upper and Lower Atmosphere, Elsevier, p. 100, doi: 10.1016/b978-012257060-5/50000-9, ISBN 978-0-12-257060-5, retrieved 2020-09-24