
Organocobalt chemistry is the chemistry of organometallic compounds containing a carbon to cobalt chemical bond. Organocobalt compounds are involved in several organic reactions and the important biomolecule vitamin B12 has a cobalt-carbon bond. Many organocobalt compounds exhibit useful catalytic properties, the preeminent example being dicobalt octacarbonyl. [1]
Alkyl complexes

Most fundamental are the cobalt complexes with only alkyl ligands. Examples include Co(4-norbornyl)4 and its cation. [3]
Alkylcobalt is represented by vitamin B12 and related enzymes. In methylcobalamin the ligand is a methyl group, which is electrophilic. in vitamin B12, the alkyl ligand is an adenosyl group. Related to vitamin B12 are cobalt porphyrins, dimethylglyoximates, and related complexes of Schiff base ligands. These synthetic compounds also form alkyl derivatives that undergo diverse reactions reminiscent of the biological processes. The weak cobalt(III)-carbon bond in vitamin B12 analogues can be exploited in a type of Cobalt mediated radical polymerization of acrylic and vinyl esters (e.g. vinyl acetate), acrylic acid and acrylonitrile. [4]
Carbonyl complexes
Dicobalt octacarbonyl is produced by the carbonylation of cobalt salts. It and its phosphine derivatives are among the most widely used organocobalt compounds. Heating Co2(CO)8 gives Co4(CO)12. Very elaborate cobalt-carbonyl clusters have been prepared starting from these complexes. Heating cobalt carbonyl with bromoform gives methylidynetricobaltnonacarbonyl. Dicobalt octacarbonyl also reacts with alkynes to give dicobalt hexacarbonyl acetylene complexes with the formula Co2(CO)6(C2R2). Because they can be removed later, the cobalt carbonyl centers function as a protective group for the alkyne. In the Nicholas reaction an alkyne group is also protected and at the same time the alpha-carbon position is activated for nucleophilic substitution.
Cp, allyl, and alkene compounds
Sandwich compounds

Organocobalt compounds are known with alkene, allyl, diene, and Cp ligands. A famous sandwich compound is cobaltocene, a rare example of low-spin Co(II) complex. This 19-electron metallocene is used as a reducing agent and as a source of CpCo. Other sandwich compounds are CoCp(C6Me6) and Co(C6Me6)2, with 20 electrons and 21 electrons, respectively. Reduction of anhydrous cobalt(II) chloride with sodium in the presence of cyclooctadiene gives Co(cyclooctadiene)(cyclooctenyl), a synthetically versatile reagent. [5]
CpCo(CO)2 and derivatives
The half-sandwich compounds of the type CpCoL2 have been well investigated (L = CO, alkene). The complexes CpCo(C2H4)2 and CpCo(cod) catalyze alkyne trimerisation, [6] which has been applied to the synthesis of a variety of complex structures. [7]
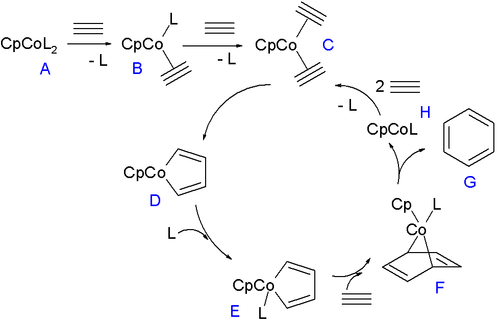
Applications

Dicobalt octacarbonyl is used commercially for hydroformylation of alkenes. A key intermediate is cobalt tetracarbonyl hydride (HCo(CO)4). Processes involving cobalt are practiced commercially mainly for the production of C7-C14 alcohols used for the production of surfactants. [10] [11] Many hydroformylations have switched from cobalt-based processes to rhodium-based processes, despite the great expense of that metal. Replacing H2 by water or an alcohol, the reaction product is a carboxylic acid or an ester. An example of this reaction type is the conversion of butadiene to adipic acid. Cobalt catalysts (together with iron) are relevant in the Fischer–Tropsch process in which it is assumed that organocobalt intermediates form.
Cobalt complexes have been applies to the synthesis of pyridine derivatives starting from alkynes and nitriles.
Aspirational applications
Although really only dicobalt octacarbonyl has achieved commercial success, many reports have appeared promising applications. [12] [13] [14] Often these ventures are motivated by the use of "earth abundant" catalysts. [15]
References
- ^ Omae, Iwao (2007). "Three characteristic reactions of organocobalt compounds in organic synthesis". Applied Organometallic Chemistry. 21 (5): 318–344. doi: 10.1002/aoc.1213.
- ^ B. K. Bower and H. G. Tennent (1972). "Transition metal bicyclo[2.2.1]hept-1-yls". J. Am. Chem. Soc. 94 (7): 2512–2514. doi: 10.1021/ja00762a056.
- ^ Byrne, Erin K.; Theopold, Klaus H. (1987-02-01). "Redox chemistry of tetrakis(1-norbornyl)cobalt. Synthesis and Characterization of a Cobalt(V) Alkyl and Self-Exchange Rate of a Co(III)/Co(IV) Couple". Journal of the American Chemical Society. 109 (4): 1282–1283. doi: 10.1021/ja00238a066. ISSN 0002-7863.
- ^ Antoine, Debuigne; Poli, Rinaldo; Jérôme, Christine; Jérôme, Robert; Detrembleur, Christophe (2009). "Overview of Cobalt-Mediated Radical Polymerization: Roots, State of the Art and Future Prospects" (PDF). Progress in Polymer Science. 34 (3): 211–239. doi: 10.1016/j.progpolymsci.2008.11.003. S2CID 95760628.
- ^ Gosser, L. W.; Cushing, M. A. Jr. (1977). "Π-Cyclooctenyl-π-L,5-Cycloocta-Dienecobalt". π-Cyclooctenyl-π-1,5-cyclooctadienecobalt. Inorganic Syntheses. Vol. 17. pp. 112–15. doi: 10.1002/9780470132487.ch32. ISBN 978-0-470-13248-7.
- ^ Cobalt-Catalyzed Cyclotrimerization of Alkynes: The Answer to the Puzzle of Parallel Reaction Pathways Nicolas Agenet, Vincent Gandon, K. Peter C. Vollhardt, Max Malacria, Corinne Aubert J. Am. Chem. Soc.; 2007; 129(28) pp 8860 - 8871; (Article) doi: 10.1021/ja072208r
- ^ Chebny VJ, Dhar D, Lindeman SV, Rathore R (2006). "Simultaneous Ejection of Six Electrons at a Constant Potential by Hexakis(4-ferrocenylphenyl)benzene". Org. Lett. 8 (22): 5041–5044. doi: 10.1021/ol061904d. PMID 17048838.
- ^ Richard F. Heck; David S. Breslow (1961). "The Reaction of Cobalt Hydrotetracarbonyl with Olefins". Journal of the American Chemical Society. 83 (19): 4023–4027. doi: 10.1021/ja01480a017..
- ^ Jack Halpern (2001). "'Organometallic chemistry at the threshold of a new millennium. Retrospect and prospect". Pure and Applied Chemistry. 73 (2): 209–220. doi: 10.1351/pac200173020209.
- ^ Hebrard, Frédéric; Kalck, Philippe (2009). "Cobalt-Catalyzed Hydroformylation of Alkenes: Generation and Recycling of the Carbonyl Species, and Catalytic Cycle". Chemical Reviews. 109 (9): 4272–4282. doi: 10.1021/cr8002533. PMID 19572688.
- ^ Boy Cornils, Wolfgang A. Herrmann, Chi-Huey Wong, Horst Werner Zanthoff: Catalysis from A to Z: A Concise Encyclopedia, 2408 Seiten, Verlag Wiley-VCH Verlag GmbH & Co. KGaA, (2012), ISBN 3-527-33307-X.
- ^ Liu, Weiping; Sahoo, Basudev; Junge, Kathrin; Beller, Matthias (2018). "Cobalt Complexes as an Emerging Class of Catalysts for Homogeneous Hydrogenations". Accounts of Chemical Research. 51 (8): 1858–1869. doi: 10.1021/acs.accounts.8b00262. PMID 30091891. S2CID 51954703.
- ^ Guo, Jun; Cheng, Zhaoyang; Chen, Jianhui; Chen, Xu; Lu, Zhan (2021). "Iron- and Cobalt-Catalyzed Asymmetric Hydrofunctionalization of Alkenes and Alkynes". Accounts of Chemical Research. 54 (11): 2701–2716. doi: 10.1021/acs.accounts.1c00212. PMID 34011145. S2CID 234792059.
- ^ Biswas, Souvagya; Parsutkar, Mahesh M.; Jing, Stanley M.; Pagar, Vinayak V.; Herbort, James H.; Rajanbabu, T. V. (2021). "A New Paradigm in Enantioselective Cobalt Catalysis: Cationic Cobalt(I) Catalysts for Heterodimerization, Cycloaddition, and Hydrofunctionalization Reactions of Olefins". Accounts of Chemical Research. 54 (24): 4545–4564. doi: 10.1021/acs.accounts.1c00573. PMC 8721816. PMID 34847327.
- ^ Chirik, Paul J. (2015). "Iron- and Cobalt-Catalyzed Alkene Hydrogenation: Catalysis with Both Redox-Active and Strong Field Ligands". Accounts of Chemical Research. 48 (6): 1687–1695. doi: 10.1021/acs.accounts.5b00134. PMID 26042837.