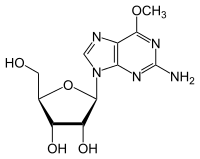
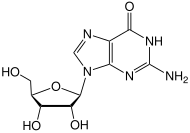
Methylated-DNA--protein-cysteine methyltransferase (MGMT), also known as O6-alkylguanine DNA alkyltransferase AGT, is a protein that in humans is encoded by the MGMT gene. [5] [6] MGMT is crucial for genome stability. It repairs the naturally occurring mutagenic DNA lesion O6-methylguanine back to guanine and prevents mismatch and errors during DNA replication and transcription. Accordingly, loss of MGMT increases the carcinogenic risk in mice after exposure to alkylating agents. [7] The two bacterial isozymes are Ada and Ogt.
Function and mechanism
| 'O6-alkylguanine DNA alkyltransferase' | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 2.1.1.63 | ||||||||
| CAS no. | 77271-19-3 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| |||||||||
Although alkylating mutagens preferentially modify the guanine base at the N7 position, O6-alkyl-guanine is a major carcinogenic lesion in DNA. This DNA adduct is removed by the repair protein O6-alkylguanine DNA alkyltransferase through an SN2 mechanism. This protein is not a true enzyme since it removes the alkyl group from the lesion in a stoichiometric reaction and the active enzyme is not regenerated after it is alkylated (referred to as a suicide enzyme). The methyl-acceptor residue in the protein is a cysteine. [8]
- Demethylation of 6-O-methylguanosine to Guanosine
Clinical significance
In patients with glioblastoma, a severe type of brain tumor, the cancer medicine temozolomide is more effective in those with a methylation of the gene's promoter. [9] Overall, MGMT methylation is associated with prolonged patient survival in clinical prediction models. [10] For testing of the MGMT promoter methylation status in the clinical setting, DNA-based methods such as methylation-specific polymerase chain reaction (MS-PCR) or pyrosequencing are preferred over immunohistochemical or RNA- based assays. [11]
In patients with pituitary tumours, MGMT can predict the clinical and radiological response to treatment with temozolomide. In this context the MGMT status is optimally assessed by immunohistochemistry, with MGMT depleted tumours expected to demonstrate a response. [12] Promotor methylation status (of MGMT) does not predict temozolomide response because, in pituitary tumours, the promotor is almost always unmethylated. [13]
MGMT has also been shown to be a useful tool increasing gene therapy efficiency. By using a two component vector consisting of a transgene of interest and MGMT, in vivo drug selection can be utilized to select for successfully transduced cells. [14]
Mutagens in the environment, [15] in tobacco smoke, [16] food, [17] as well as endogenous metabolic products [18] generate reactive electrophilic species that alkylate or specifically methylate DNA, generating 6-O-methylguanine (m6G).
In 1985 Yarosh summarized the early work that established m6G as the alkylated base in DNA that was the most mutagenic and carcinogenic. [19] In 1994 Rasouli-Nia et al. [20] showed that about one mutation was induced for every eight unrepaired m6Gs in DNA. Mutations can cause progression to cancer by a process of natural selection.[ citation needed]
Expression in cancer
| Cancer type | Frequency of deficiency in cancer | Frequency of deficiency in adjacent field defect |
|---|---|---|
| Cervical [21] | 61% | 39% |
| Colorectal | 40%-90% [22] [23] [24] [25] [26] | 11%-34% [22] [23] |
| Colorectal with microsatellite instability [27] | 70% | 60% |
| Esophageal adenocarcinoma | 71%-79% [28] [29] | 89% [29] |
| Esophageal squamous cell carcinoma | 38%-96% [28] [30] [31] | 65% [31] |
| Glioblastoma due to promoter methylation | 44%-59% [32] [33] | |
| Head and neck squamous cell carcinoma | 54% [34] | |
| Hepatocellular carcinoma (hepatitis C virus associated) [35] | 68% | 65% |
| Larynx | 54%-61% [36] [37] | 38% [37] |
| Stomach | 32%-88% [38] [39] | 17%-78% [38] [39] |
| Thyroid [40] | 87% |
Epigenetic repression
Only a minority of sporadic cancers with a DNA repair deficiency have a mutation in a DNA repair gene. However, a majority of sporadic cancers with a DNA repair deficiency do have one or more epigenetic alterations that reduce or silence DNA repair gene expression. For example, in a study of 113 sequential colorectal cancers, only four had a missense mutation in the DNA repair gene MGMT, while the majority had reduced MGMT expression due to methylation of the MGMT promoter region (an epigenetic alteration). [41]
MGMT can be epigenetically repressed in a number of ways. [42] When MGMT expression is repressed in cancers, this is often due to methylation of its promoter region. [42] However, expression can also be repressed by di-methylation of lysine 9 of histone 3 [43] or by over-expression of a number of microRNAs including miR-181d, miR-767-3p and miR-603. [42] [44] [45]
MGMT (O-6-methylguanine-DNA methyltransferase) is an important cancer biomarker because it is involved in the repair of DNA damage and is often silenced or inactivated in cancer cells. The loss of MGMT function leads to a higher rate of mutations, promoting the formation and progression of tumors. The presence or absence of MGMT expression in a cancer sample can indicate a patient's response to alkylating chemotherapy, which is a common treatment for certain types of cancer. Hence, MGMT can be used as a prognostic marker to predict the likelihood of treatment response and to guide the selection of appropriate therapies. A number of point-of-care devices are under development to monitor the methylation status of MGMT. [46]
Deficiency in field defects
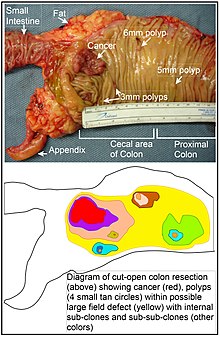
A field defect is an area or "field" of epithelium that has been preconditioned by epigenetic changes and/or mutations so as to predispose it towards development of cancer. A field defect is illustrated in the photo and diagram shown of a colon segment having a colon cancer and four small polyps within the same area as well. As pointed out by Rubin, "The vast majority of studies in cancer research has been done on well-defined tumors in vivo, or on discrete neoplastic foci in vitro. [47] Yet there is evidence that more than 80% of the somatic mutations found in mutator phenotype human colorectal tumors occur before the onset of terminal clonal expansion." [48] Similarly, Vogelstein et al. [49] point out that more than half of somatic mutations identified in tumors occurred in a pre-neoplastic phase (in a field defect), during growth of apparently normal cells.
In the Table above, MGMT deficiencies were noted in the field defects (histologically normal tissues) surrounding most of the cancers. If MGMT is epigenetically reduced or silenced, it would not likely confer a selective advantage upon a stem cell. However, reduced or absent expression of MGMT would cause increased rates of mutation, and one or more of the mutated genes may provide the cell with a selective advantage. The expression-deficient MGMT gene could then be carried along as a selectively neutral or only slightly deleterious passenger (hitch-hiker) gene when the mutated stem cell generates an expanded clone. The continued presence of a clone with an epigenetically repressed MGMT would continue to generate further mutations, some of which could produce a tumor.
Deficiency with exogenous damage
MGMT deficiency alone may not be sufficient to cause progression to cancer. Mice with a homozygous mutation in MGMT did not develop more cancers than wild-type mice when grown without stress. [50] However, stressful treatment of mice with azoxymethane and dextran sulphate caused more than four colonic tumors per MGMT mutant mouse, but less than one tumor per wild-type mouse. [51]
Repression in coordination with other DNA repair genes
In a cancer, multiple DNA repair genes are often found to be simultaneously repressed. [52] In one example, involving MGMT, Jiang et al. [53] conducted a study where they evaluated the mRNA expression of 27 DNA repair genes in 40 astrocytomas compared to normal brain tissues from non-astrocytoma individuals. Among the 27 DNA repair genes evaluated, 13 DNA repair genes, MGMT, NTHL1, OGG1, SMUG1, ERCC1, ERCC2, ERCC3, ERCC4, MLH1, MLH3, RAD50, XRCC4 and XRCC5 were all significantly down-regulated in all three grades (II, III and IV) of astrocytomas. The repression of these 13 genes in lower grade as well as in higher grade astrocytomas suggested that they may be important in early as well as in later stages of astrocytoma. In another example, Kitajima et al. [54] found that immunoreactivity for MGMT and MLH1 expression was closely correlated in 135 specimens of gastric cancer and loss of MGMT and hMLH1 appeared to be synchronously accelerated during tumor progression.
Deficient expression of multiple DNA repair genes are often found in cancers, [52] and may contribute to the thousands of mutations usually found in cancers (see mutation frequencies in cancers).
Interactions
O6-methylguanine-DNA methyltransferase has been shown to interact with estrogen receptor alpha. [55]
See also
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000170430 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000054612 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Tano K, Shiota S, Collier J, Foote RS, Mitra S (January 1990). "Isolation and structural characterization of a cDNA clone encoding the human DNA repair protein for O6-alkylguanine". Proc. Natl. Acad. Sci. U.S.A. 87 (2): 686–90. Bibcode: 1990PNAS...87..686T. doi: 10.1073/pnas.87.2.686. PMC 53330. PMID 2405387.
- ^ Natarajan AT, Vermeulen S, Darroudi F, Valentine MB, Brent TP, Mitra S, Tano K (January 1992). "Chromosomal localization of human O6-methylguanine-DNA methyltransferase (MGMT) gene by in situ hybridization". Mutagenesis. 7 (1): 83–5. doi: 10.1093/mutage/7.1.83. PMID 1635460.
- ^ Shiraishi A, Sakumi K, Sekiguchi M (October 2000). "Increased susceptibility to chemotherapeutic alkylating agents of mice deficient in DNA repair methyltransferase". Carcinogenesis. 21 (10): 1879–83. doi: 10.1093/carcin/21.10.1879. PMID 11023546.
- ^ Kaina B, Christmann M, Naumann S, Roos WP (August 2007). "MGMT: key node in the battle against genotoxicity, carcinogenicity and apoptosis induced by alkylating agents". DNA Repair (Amst.). 6 (8): 1079–99. doi: 10.1016/j.dnarep.2007.03.008. PMID 17485253.
- ^ Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005). "MGMT gene silencing and benefit from temozolomide in glioblastoma". N. Engl. J. Med. 352 (10): 997–1003. doi: 10.1056/NEJMoa043331. PMID 15758010.
- ^ Molenaar RJ, Verbaan D, Lamba S, Zanon C, Jeuken JW, Boots-Sprenger SH, Wesseling P, Hulsebos TJ, Troost D, van Tilborg AA, Leenstra S, Vandertop WP, Bardelli A, van Noorden CJ, Bleeker FE (2014). "The combination of IDH1 mutations and MGMT methylation status predicts survival in glioblastoma better than either IDH1 or MGMT alone". Neuro-Oncology. 16 (9): 1263–73. doi: 10.1093/neuonc/nou005. PMC 4136888. PMID 24510240.
- ^ Preusser, M.; Janzer, Charles R.; Felsberg, J.; Reifenberger, G.; Hamou, M. F.; Diserens, A. C.; Stupp, R.; Gorlia, T.; Marosi, C.; Heinzl, H.; Hainfellner, J. A.; Hegi, M. (Oct 2008). "Anti-O6-methylguanine-methyltransferase (MGMT) immunohistochemistry in glioblastoma multiforme: observer variability and lack of association with patient survival impede its use as clinical biomarker". Brain Pathol. 18 (4): 520–532. doi: 10.1111/j.1750-3639.2008.00153.x. PMC 8095504. PMID 18400046. S2CID 21167901.
- ^ McCormack, A. (2022). "Temozolomide in aggressive pituitary tumours and pituitary carcinomas". Best Practice & Research. Clinical Endocrinology & Metabolism. 36 (6): 101713. doi: 10.1016/j.beem.2022.101713. PMID 36274026. S2CID 252941246.
- ^ Bush, Z. M.; Longtine, J. A.; Cunningham, T.; Schiff, D.; Jane Jr, J. A.; Vance, M. L.; Thorner, M. O.; Laws Jr, E. R.; Lopes, M. B. (2010). "Temozolomide treatment for aggressive pituitary tumors: Correlation of clinical outcome with O(6)-methylguanine methyltransferase (MGMT) promoter methylation and expression". The Journal of Clinical Endocrinology and Metabolism. 95 (11): E280-90. doi: 10.1210/jc.2010-0441. PMC 5393383. PMID 20668043.
- ^ Chang AH, Stephan MT, Lisowski L, Sadelain M (2008). "Erythroid-specific human factor IX delivery from in vivo selected hematopoietic stem cells following nonmyeloablative conditioning in hemophilia B mice". Mol. Ther. 16 (10): 1745–52. doi: 10.1038/mt.2008.161. PMC 2658893. PMID 18682698.
- ^ Bartsch H, Montesano R (1984). "Relevance of nitrosamines to human cancer". Carcinogenesis. 5 (11): 1381–93. doi: 10.1093/carcin/5.11.1381. PMID 6386215.
- ^ Christmann M, Kaina B (2012). "O(6)-methylguanine-DNA methyltransferase (MGMT): impact on cancer risk in response to tobacco smoke". Mutat. Res. 736 (1–2): 64–74. doi: 10.1016/j.mrfmmm.2011.06.004. PMID 21708177.
- ^ Fahrer J, Kaina B (2013). "O6-methylguanine-DNA methyltransferase in the defense against N-nitroso compounds and colorectal cancer". Carcinogenesis. 34 (11): 2435–42. doi: 10.1093/carcin/bgt275. PMID 23929436.
- ^ De Bont R, van Larebeke N (2004). "Endogenous DNA damage in humans: a review of quantitative data". Mutagenesis. 19 (3): 169–85. doi: 10.1093/mutage/geh025. PMID 15123782.
- ^ Yarosh DB (1985). "The role of O6-methylguanine-DNA methyltransferase in cell survival, mutagenesis and carcinogenesis". Mutat. Res. 145 (1–2): 1–16. doi: 10.1016/0167-8817(85)90034-3. PMID 3883145.
- ^ Rasouli-Nia A, Sibghat-Ullah, Mirzayans R, Paterson MC, Day RS (1994). "On the quantitative relationship between O6-methylguanine residues in genomic DNA and production of sister-chromatid exchanges, mutations and lethal events in a Mer- human tumor cell line". Mutat. Res. 314 (2): 99–113. doi: 10.1016/0921-8777(94)90074-4. PMID 7510369.
- ^ Iliopoulos D, Oikonomou P, Messinis I, Tsezou A (2009). "Correlation of promoter hypermethylation in hTERT, DAPK and MGMT genes with cervical oncogenesis progression". Oncol. Rep. 22 (1): 199–204. doi: 10.3892/or_00000425. PMID 19513524.
- ^ a b Shen L, Kondo Y, Rosner GL, Xiao L, Hernandez NS, Vilaythong J, Houlihan PS, Krouse RS, Prasad AR, Einspahr JG, Buckmeier J, Alberts DS, Hamilton SR, Issa JP (2005). "MGMT promoter methylation and field defect in sporadic colorectal cancer". J. Natl. Cancer Inst. 97 (18): 1330–8. doi: 10.1093/jnci/dji275. PMID 16174854.
- ^ a b Lee KH, Lee JS, Nam JH, Choi C, Lee MC, Park CS, Juhng SW, Lee JH (2011). "Promoter methylation status of hMLH1, hMSH2, and MGMT genes in colorectal cancer associated with adenoma-carcinoma sequence". Langenbecks Arch Surg. 396 (7): 1017–26. doi: 10.1007/s00423-011-0812-9. PMID 21706233. S2CID 8069716.
- ^ Psofaki V, Kalogera C, Tzambouras N, Stephanou D, Tsianos E, Seferiadis K, Kolios G (2010). "Promoter methylation status of hMLH1, MGMT, and CDKN2A/p16 in colorectal adenomas". World J. Gastroenterol. 16 (28): 3553–60. doi: 10.3748/wjg.v16.i28.3553. PMC 2909555. PMID 20653064.
- ^ Amatu A, Sartore-Bianchi A, Moutinho C, Belotti A, Bencardino K, Chirico G, Cassingena A, Rusconi F, Esposito A, Nichelatti M, Esteller M, Siena S (2013). "Promoter CpG island hypermethylation of the DNA repair enzyme MGMT predicts clinical response to dacarbazine in a phase II study for metastatic colorectal cancer". Clin. Cancer Res. 19 (8): 2265–72. doi: 10.1158/1078-0432.CCR-12-3518. PMID 23422094.
- ^ Mokarram P, Zamani M, Kavousipour S, Naghibalhossaini F, Irajie C, Moradi Sarabi M, Hosseini SV (2013). "Different patterns of DNA methylation of the two distinct O6-methylguanine-DNA methyltransferase (O6-MGMT) promoter regions in colorectal cancer". Mol. Biol. Rep. 40 (5): 3851–7. doi: 10.1007/s11033-012-2465-3. PMID 23271133. S2CID 18733871.
- ^ Svrcek M, Buhard O, Colas C, Coulet F, Dumont S, Massaoudi I, Lamri A, Hamelin R, Cosnes J, Oliveira C, Seruca R, Gaub MP, Legrain M, Collura A, Lascols O, Tiret E, Fléjou JF, Duval A (2010). "Methylation tolerance due to an O6-methylguanine DNA methyltransferase (MGMT) field defect in the colonic mucosa: an initiating step in the development of mismatch repair-deficient colorectal cancers". Gut. 59 (11): 1516–26. doi: 10.1136/gut.2009.194787. PMID 20947886. S2CID 206950452.
- ^ a b Hasina R, Surati M, Kawada I, Arif Q, Carey GB, Kanteti R, Husain AN, Ferguson MK, Vokes EE, Villaflor VM, Salgia R (2013). "O-6-methylguanine-deoxyribonucleic acid methyltransferase methylation enhances response to temozolomide treatment in esophageal cancer". J Carcinog. 12: 20. doi: 10.4103/1477-3163.120632. PMC 3853796. PMID 24319345.
- ^ a b Kuester D, El-Rifai W, Peng D, Ruemmele P, Kroeckel I, Peters B, Moskaluk CA, Stolte M, Mönkemüller K, Meyer F, Schulz HU, Hartmann A, Roessner A, Schneider-Stock R (2009). "Silencing of MGMT expression by promoter hypermethylation in the metaplasia-dysplasia-carcinoma sequence of Barrett's esophagus". Cancer Lett. 275 (1): 117–26. doi: 10.1016/j.canlet.2008.10.009. PMC 4028828. PMID 19027227.
- ^ Ling ZQ, Li P, Ge MH, Hu FJ, Fang XH, Dong ZM, Mao WM (2011). "Aberrant methylation of different DNA repair genes demonstrates distinct prognostic value for esophageal cancer". Dig. Dis. Sci. 56 (10): 2992–3004. doi: 10.1007/s10620-011-1774-z. PMID 21674174. S2CID 22913110.
- ^ a b Su Y, Yin L, Liu R, Sheng J, Yang M, Wang Y, Pan E, Guo W, Pu Y, Zhang J, Liang G (2014). "Promoter methylation status of MGMT, hMSH2, and hMLH1 and its relationship to corresponding protein expression and TP53 mutations in human esophageal squamous cell carcinoma". Med. Oncol. 31 (2): 784. doi: 10.1007/s12032-013-0784-4. PMID 24366688. S2CID 22746140.
- ^ Morandi L, Franceschi E, de Biase D, Marucci G, Tosoni A, Ermani M, Pession A, Tallini G, Brandes A (2010). "Promoter methylation analysis of O6-methylguanine-DNA methyltransferase in glioblastoma: detection by locked nucleic acid based quantitative PCR using an imprinted gene (SNURF) as a reference". BMC Cancer. 10: 48. doi: 10.1186/1471-2407-10-48. PMC 2843669. PMID 20167086.
- ^ Quillien V, Lavenu A, Karayan-Tapon L, Carpentier C, Labussière M, Lesimple T, Chinot O, Wager M, Honnorat J, Saikali S, Fina F, Sanson M, Figarella-Branger D (2012). "Comparative assessment of 5 methods (methylation-specific polymerase chain reaction, MethyLight, pyrosequencing, methylation-sensitive high-resolution melting, and immunohistochemistry) to analyze O6-methylguanine-DNA-methyltranferase in a series of 100 glioblastoma patients". Cancer. 118 (17): 4201–11. doi: 10.1002/cncr.27392. PMID 22294349. S2CID 8145409.
- ^ Koutsimpelas D, Pongsapich W, Heinrich U, Mann S, Mann WJ, Brieger J (2012). "Promoter methylation of MGMT, MLH1 and RASSF1A tumor suppressor genes in head and neck squamous cell carcinoma: pharmacological genome demethylation reduces proliferation of head and neck squamous carcinoma cells". Oncol. Rep. 27 (4): 1135–41. doi: 10.3892/or.2012.1624. PMC 3583513. PMID 22246327.
- ^ Zekri AR, Bahnasy AA, Shoeab FE, Mohamed WS, El-Dahshan DH, Ali FT, Sabry GM, Dasgupta N, Daoud SS (2014). "Methylation of multiple genes in hepatitis C virus associated hepatocellular carcinoma". J Adv Res. 5 (1): 27–40. doi: 10.1016/j.jare.2012.11.002. PMC 4294722. PMID 25685469.
- ^ Pierini S, Jordanov SH, Mitkova AV, Chalakov IJ, Melnicharov MB, Kunev KV, Mitev VI, Kaneva RP, Goranova TE (2014). "Promoter hypermethylation of CDKN2A, MGMT, MLH1, and DAPK genes in laryngeal squamous cell carcinoma and their associations with clinical profiles of the patients". Head Neck. 36 (8): 1103–8. doi: 10.1002/hed.23413. PMID 23804521. S2CID 11916790.
- ^ a b Paluszczak J, Misiak P, Wierzbicka M, Woźniak A, Baer-Dubowska W (2011). "Frequent hypermethylation of DAPK, RARbeta, MGMT, RASSF1A and FHIT in laryngeal squamous cell carcinomas and adjacent normal mucosa". Oral Oncol. 47 (2): 104–7. doi: 10.1016/j.oraloncology.2010.11.006. PMID 21147548.
- ^ a b Jin J, Xie L, Xie CH, Zhou YF (2014). "Aberrant DNA methylation of MGMT and hMLH1 genes in prediction of gastric cancer". Genet. Mol. Res. 13 (2): 4140–5. doi: 10.4238/2014.May.30.9. PMID 24938706.
- ^ a b Zou XP, Zhang B, Zhang XQ, Chen M, Cao J, Liu WJ (2009). "Promoter hypermethylation of multiple genes in early gastric adenocarcinoma and precancerous lesions". Hum. Pathol. 40 (11): 1534–42. doi: 10.1016/j.humpath.2009.01.029. PMID 19695681.
- ^ Mokhtar M, Kondo K, Namura T, Ali AH, Fujita Y, Takai C, Takizawa H, Nakagawa Y, Toba H, Kajiura K, Yoshida M, Kawakami G, Sakiyama S, Tangoku A (2014). "Methylation and expression profiles of MGMT gene in thymic epithelial tumors". Lung Cancer. 83 (2): 279–87. doi: 10.1016/j.lungcan.2013.12.004. PMID 24388682.
- ^ Halford S, Rowan A, Sawyer E, Talbot I, Tomlinson I (June 2005). "O(6)-methylguanine methyltransferase in colorectal cancers: detection of mutations, loss of expression, and weak association with G:C>A:T transitions". Gut. 54 (6): 797–802. doi: 10.1136/gut.2004.059535. PMC 1774551. PMID 15888787.
- ^ a b c Cabrini G, Fabbri E, Lo Nigro C, Dechecchi MC, Gambari R (2015). "Regulation of expression of O6-methylguanine-DNA methyltransferase and the treatment of glioblastoma (Review)". Int. J. Oncol. 47 (2): 417–28. doi: 10.3892/ijo.2015.3026. PMC 4501657. PMID 26035292.
- ^ Nakagawachi T, Soejima H, Urano T, Zhao W, Higashimoto K, Satoh Y, Matsukura S, Kudo S, Kitajima Y, Harada H, Furukawa K, Matsuzaki H, Emi M, Nakabeppu Y, Miyazaki K, Sekiguchi M, Mukai T (2003). "Silencing effect of CpG island hypermethylation and histone modifications on O6-methylguanine-DNA methyltransferase (MGMT) gene expression in human cancer". Oncogene. 22 (55): 8835–44. doi: 10.1038/sj.onc.1207183. PMID 14647440.
- ^ Kushwaha D, Ramakrishnan V, Ng K, Steed T, Nguyen T, Futalan D, Akers JC, Sarkaria J, Jiang T, Chowdhury D, Carter BS, Chen CC (2014). "A genome-wide miRNA screen revealed miR-603 as a MGMT-regulating miRNA in glioblastomas". Oncotarget. 5 (12): 4026–39. doi: 10.18632/oncotarget.1974. PMC 4147303. PMID 24994119.
- ^ Zhang W, Zhang J, Hoadley K, Kushwaha D, Ramakrishnan V, Li S, Kang C, You Y, Jiang C, Song SW, Jiang T, Chen CC (2012). "miR-181d: a predictive glioblastoma biomarker that downregulates MGMT expression". Neuro-Oncology. 14 (6): 712–9. doi: 10.1093/neuonc/nos089. PMC 3367855. PMID 22570426.
- ^ Jahin, M., Fenech-Salerno, B., Moser, N., Georgiou, P., Flanagan, J., Toumazou, C., ... & Kalofonou, M. (2021, November). Detection of MGMT methylation status using a Lab-on-Chip compatible isothermal amplification method. In 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC) (pp. 7385-7389). IEEE. doi:10.1109/EMBC46164.2021.9630776.
- ^ Rubin H (March 2011). "Fields and field cancerization: the preneoplastic origins of cancer: asymptomatic hyperplastic fields are precursors of neoplasia, and their progression to tumors can be tracked by saturation density in culture". BioEssays. 33 (3): 224–31. doi: 10.1002/bies.201000067. PMID 21254148. S2CID 44981539.
- ^ Tsao JL, Yatabe Y, Salovaara R, Järvinen HJ, Mecklin JP, Aaltonen LA, Tavaré S, Shibata D (February 2000). "Genetic reconstruction of individual colorectal tumor histories". Proc. Natl. Acad. Sci. U.S.A. 97 (3): 1236–41. Bibcode: 2000PNAS...97.1236T. doi: 10.1073/pnas.97.3.1236. PMC 15581. PMID 10655514.
- ^ Vogelstein B, Papadopoulos N, Velculescu VE, Zhou S, Diaz LA, Kinzler KW (March 2013). "Cancer genome landscapes". Science. 339 (6127): 1546–58. Bibcode: 2013Sci...339.1546V. doi: 10.1126/science.1235122. PMC 3749880. PMID 23539594.
- ^ Meira LB, Calvo JA, Shah D, Klapacz J, Moroski-Erkul CA, Bronson RT, Samson LD (2014). "Repair of endogenous DNA base lesions modulate lifespan in mice". DNA Repair (Amst.). 21: 78–86. doi: 10.1016/j.dnarep.2014.05.012. PMC 4125484. PMID 24994062.
- ^ Wirtz S, Nagel G, Eshkind L, Neurath MF, Samson LD, Kaina B (2010). "Both base excision repair and O6-methylguanine-DNA methyltransferase protect against methylation-induced colon carcinogenesis". Carcinogenesis. 31 (12): 2111–7. doi: 10.1093/carcin/bgq174. PMC 2994278. PMID 20732909.
- ^ a b Bernstein C, Bernstein H (2015). "Epigenetic reduction of DNA repair in progression to gastrointestinal cancer". World J Gastrointest Oncol. 7 (5): 30–46. doi: 10.4251/wjgo.v7.i5.30. PMC 4434036. PMID 25987950.
- ^ Jiang Z, Hu J, Li X, Jiang Y, Zhou W, Lu D (2006). "Expression analyses of 27 DNA repair genes in astrocytoma by TaqMan low-density array". Neurosci. Lett. 409 (2): 112–7. doi: 10.1016/j.neulet.2006.09.038. PMID 17034947. S2CID 54278905.
- ^ Kitajima Y, Miyazaki K, Matsukura S, Tanaka M, Sekiguchi M (2003). "Loss of expression of DNA repair enzymes MGMT, hMLH1, and hMSH2 during tumor progression in gastric cancer". Gastric Cancer. 6 (2): 86–95. doi: 10.1007/s10120-003-0213-z. PMID 12861399.
- ^ Teo AK, Oh HK, Ali RB, Li BF (October 2001). "The modified human DNA repair enzyme O(6)-methylguanine-DNA methyltransferase is a negative regulator of estrogen receptor-mediated transcription upon alkylation DNA damage". Mol. Cell. Biol. 21 (20): 7105–14. doi: 10.1128/MCB.21.20.7105-7114.2001. PMC 99886. PMID 11564893.
Further reading
- Margison GP, Povey AC, Kaina B, Santibáñez Koref MF (2003). "Variability and regulation of O6-alkylguanine-DNA alkyltransferase". Carcinogenesis. 24 (4): 625–35. doi: 10.1093/carcin/bgg005. PMID 12727789.