 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Pharmacokinetic data | |
| Excretion | ~ |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
| Formula | C28H37NO8 |
| Molar mass | 515.603 g·mol−1 |
| |
| |
| (verify) | |
Nargenicin (CP-47,444, CS-682) is a 28 carbon macrolide with a fused tricyclic core that has in addition a unique ether bridge. The polyketide antibiotic was isolated from Nocardia argentinensis. [1] Nargenicin is effective towards gram-positive bacteria and been shown to have strong antibacterial activity against Staphylococcus aureus, including strains that are resistant to methicillin. [2] It has also been shown to induce cell differentiation and inhibit cell proliferation in a human myeloid leukemia cell line. [3]
Biosynthesis
The biosynthesis of nargenicin is believed to be closely related to fatty acid biosynthesis to produce a polyketide chain. David E. Cane and colleagues have used feeding experiments to determine that nargenicin is derived from common precursors acetate and propionate. [4] [5] [6]
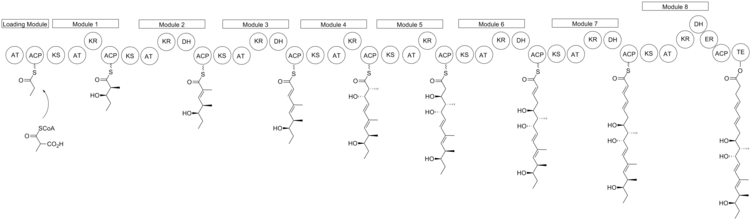
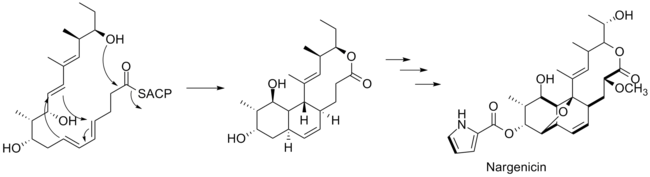
The polyketide chain produced then undergoes a ring closure to form the large lactone ring and a Diels–Alder reaction to form the fused cyclohexane/ cyclohexene rings. The oxygen atoms attached to carbons in positions that do not correspond to polyketides—carbons 8 and 13 (the ether bridge), carbon 2 (the methoxy substituent), and carbon 18 (on the hydroxyethyl chain attached to the lactone ring) are derived from molecular oxygen. [7]
References
- ^ Celmer WD, Chmurny GN, Moppett CE, Ware RS, Watts PT, Whipple EB (1980). "Structure of natural antibiotic CP-47,444". J. Am. Chem. Soc. 102 (12): 4203–4209. doi: 10.1021/ja00532a036.
- ^ Sohng JK, Yamaguchi T, Seong CN, Baik KS, Park SC, Lee HJ, et al. (October 2008). "Production, isolation and biological activity of nargenicin from Nocardia sp. CS682". Archives of Pharmacal Research. 31 (10): 1339–45. doi: 10.1007/s12272-001-2115-0. PMID 18958426. S2CID 20377463.
- ^ Kim SH, Yoo JC, Kim TS (June 2009). "Nargenicin enhances 1,25-dihydroxyvitamin D(3)- and all-trans retinoic acid-induced leukemia cell differentiation via PKCbetaI/MAPK pathways". Biochemical Pharmacology. 77 (11): 1694–701. doi: 10.1016/j.bcp.2009.03.004. PMID 19428323.
- ^ Cane DE, Yang CC (1984). "Biosynthetic Origin of the Carbon Skeleton and Oxygen Atoms of Nargenicin A1". J. Am. Chem. Soc. 106 (3): 784–787. doi: 10.1021/ja00315a052.
- ^ Cane DE, Prabhakaran PC, Tan W, Ott WR (1991). "Macrolide Biosynthesis. 6 Mechanism of Polyketide Chain Elongation". Tetrahedron Lett. 32 (40): 5457–5460. doi: 10.1016/0040-4039(91)80057-D.
- ^ Cane DE, Tan W, Ott WR (1993). "Nargenicin Biosynthesis. Incorporation of Polyketide Chain Elongation Intermediates and Support for a Proposed Intramolecular Diels-Alder Cyclization". J. Am. Chem. Soc. 115 (2): 527–535. doi: 10.1021/ja00055a024.
- ^ Cane DE, Yang CC (March 1985). "Nargenicin biosynthesis: late stage oxidations and absolute configuration". The Journal of Antibiotics. 38 (3): 423–6. doi: 10.7164/antibiotics.38.423. PMID 4008333.