
| |
| |
| Identifiers | |
|---|---|
3D model (
JSmol)
|
|
| |
| |
| Properties | |
| B6H12 | |
| Molar mass | 76.96 g·mol−1 |
| Appearance | colorless liquid |
| Melting point | −82.3 °C (−116.1 °F; 190.8 K) |
| Boiling point | 85 °C (185 °F; 358 K) (approx) |
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
| |
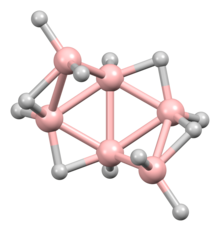
Hexaborane(12) is an inorganic compound with the formula B6H12. It is an obscure member of the boranes. It is a colorless liquid that, like most boron hydrides, is readily hydrolyzed and flammable.
The molecular structure conforms to C2
symmetry group. With the formula BnHn+6, it is classified as an arachno-cluster. As such the boron positions match six of the boron positions in the closo-B8H2−
8.
Preparation
It is typically prepared by the cluster expansion method from B5H−
8, the conjugate base of
pentaborane(9):
[1]
- LiB5H8 + 1/2 B2H6 → LiB6H11
- LiB6H11 + HCl → B6H12 + LiCl
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8. p.172.